Abstract
Respiratory syncytial virus (RSV) has been reported to cause severe morbidity and mortality among cancer patients receiving chemotherapy with or without autologous peripheral blood stem cell transplantation (APBSCT). However, little is known about the natural history of this infection in these patients, and current standard practice, aerosolized ribavirin plus intravenous immunoglobulin (IVIG), is extremely expensive, difficult to use, and not supported by controlled clinical trials. The purpose of this observational study was to determine the frequency, seasonality, morbidity, and mortality of RSV infection in a group of cancer patients receiving cytotoxic chemotherapy with neither ribavirin nor IVIG treatment. During the period of October 3, 1997, through October 14, 1998, 190 cancer patients (median age, 58 years; 71 women) underwent viral nasopharyngeal washing prior to chemotherapy. Multiple myeloma (MM) accounted for most patients (147, 77%). RSV was recovered from cultures taken from 71 patients (37%) throughout the year, although more frequently during fall and winter seasons (P < .001) than spring and summer. Serious respiratory complications developed in 19 (27%) of 71 RSV-positive patients versus 24 (20%) of 119 patients whose RSV cultures were negative (P = .384). The presence of renal failure or increased lactate dehydrogenase (LDH) prior to chemotherapy and the development of mucositis were the only predictive factors for severe respiratory complications. Recovery of RSV from nasopharyngeal washings among cancer patients is common, occurs throughout the year, and does not appear to increase serious morbidity or mortality. RSV infection may not necessarily be a contraindication for APBSCT or an indication for therapy with aerosolized ribavirin and IVIG.
Introduction
Respiratory syncytial virus (RSV) is thought to cause rare but severe community and nosocomial respiratory infections in immunocompromised adults, including cancer patients,1-9 and to occur almost exclusively during winter.10-13
Recommendations for the management of RSV infections in cancer patients include strict infection control measures, delay in the therapy for the underlying disease, and treatment with aerosolized ribavirin plus high-dose intravenous immunoglobulin (IVIG).3-9,14 These strategies are associated with huge costs, toxicity, disruption of patient care activities, and delay in treating the underlying cancer.15,16 Furthermore, these recommendations were based on studies that did not evaluate a homogenous patient population and, most important, failed to include a control group whose respiratory cultures were negative for RSV. We have previously reported on 10 myeloma patients with positive respiratory tract cultures for RSV who successfully underwent autologous peripheral blood stem cell transplantation (APBSCT) without receiving RSV-specific therapy.17
In this prospective observational study, we sought to determine the incidence and seasonality of RSV infection in a homogenous group of cancer patients not receiving RSV-specific therapy. We also determined the type of and risk for complications associated with RSV by including a control group whose respiratory cultures were negative for this virus.
Patients, materials, and methods
Cancer patients receiving cytotoxic chemotherapy, with or without APBSCT or allogeneic bone marrow transplantation (allo-BMT), were evaluated at the University of Arkansas for Medical Sciences in Little Rock. A written informed consent for specimen collection was obtained in keeping with institutional policies. This evaluation occurred over a period of 12 months (October 3, 1997, to October 14, 1998). Nasopharyngeal washings were collected into a sterile cup and transported on wet ice to the virology laboratory, generally within 2 hours. Bronchoalveolar lavage specimens and tissue obtained by open lung biopsy and autopsy were also collected for culture when available, with tissue specimens homogenized prior to culture inoculation. Specimens were inoculated into tubes containing human diploid embryonic lung (MRC-5) fibroblast cells and checked daily during the first week and twice during the second week for the characteristic RSV cytopathic effect (CPE).18,19 Results of the cultures were finalized by day 14 after inoculation. A culture was considered presumptively positive for RSV whenever CPE consistent with RSV was observed, and this was reported as positive if confirmed by an indirect immunofluorescent method (immunofluorescent assay [IFA]) performed on slides of the cell cultures using a monoclonal antibody directed against RSV (Bartels Diagnostics, Issaquah, WA). Criteria for positivity by the IFA method included an appropriate pattern of cytoplasmic immunofluorescent staining in intact cells. All cultures negative for CPE at 14 days were stained by the IFA method at that time and, if positive for this Food and Drug Administration (FDA)–approved IFA, were reported as positive for RSV despite the absence of CPE, as per standard laboratory practice.
All patients received antibacterial, antifungal, and antiviral prophylaxis and therapy according to standards of care.
Upper respiratory tract infections (URTIs) were defined as the presence of rhinorrhea, nasal/sinus congestion, otitis media, pharyngitis, or cough with a clear chest examination and/or chest roentgenogram. Lower respiratory tract infections (LRTIs) included tracheobronchitis (deep, persistent cough associated with widespread fine crepitation, rhonchi, coarse rales, or wheezes on lung auscultation and a clear chest roentgenogram) and pneumonia (acute respiratory symptoms occurring in a patient with a new pulmonary infiltrate and signs and symptoms of infection). Renal failure was defined as having a serum creatinine above 221 μM (2.5 mg/dL), while hypoxemia referred to an arterial blood oxygen tension of less than 60 mm Hg and/or a decrease of the oxygen saturation by pulse oximetry (usually 90% or below) and for which oxygen therapy was initiated. A clinically defined infection was defined as the presence of signs and symptoms of organ infection (eg, pneumonia with fever, cough, shortness of breath, and pulmonary infiltrates) but without the recovery of a specific pathogen. Microbiologically defined infection referred to the presence of the features of a clinically defined infection, together with the identification of the offending pathogen.
A lymphocyte count of below 0.7 × 109/L and a neutrophil count of below 1 × 109/L were defined as lymphopenia and neutropenia, respectively.
The following variables were evaluated: sex, age, underlying disease, stage at diagnosis, disease status prior to chemotherapy (ie, relapse or complete/partial remission), site of care (inpatient or outpatient), presence of comorbidities (ie, amyloidosis, renal failure with/without hemodialysis, others), smoking, preceding therapy including adrenal corticosteroids (with total dose), stem cell or bone marrow transplantation (autologous or allogeneic), conditioning regimen, and number of CD34+ cells infused per kilogram. Baseline laboratory values obtained prior to chemotherapy were also evaluated, including white blood cell count and differential, platelet counts, levels of uninvolved serum immunoglobulins, pulmonary function tests, chest roentgenogram, computed tomography (CT) scan of sinuses, liver and renal function tests, serum albumin, serum lactate dehydrogenase (LDH), and arterial blood oxygen tension for those patients undergoing myeloablative chemotherapy with transplantation. Clinical evaluation included daily examinations.
A composite index of respiratory complications likely to develop as a result of RSV infection was the primary study end point and was defined as the occurrence of any of the following: hypoxemia, tracheobronchitis, pneumonia, transfer to intensive care unit (ICU), intubation, mechanical ventilation, or death within 30 days from initiation of cytotoxic chemotherapy.
Univariate analyses
A univariate analysis of the prognostic effect of each factor was conducted. An analysis of correlations between the evaluated factors was done to identify groups of related factors. Laboratory variables were initially entered as continuous variables. For practical purposes, some continuous variables entered into the model were dichotomized (cutoff points were those of the upper or lower normal limit of the variable). No dichotomized covariates were entered unless the continuous analog had a significant independent prognostic effect.
Multivariate analyses
All variables with a P value .10 or below in the univariate analysis were entered into the model. The RSV status was also forced as a variable, although it was not significant in the univariate analysis.
Results
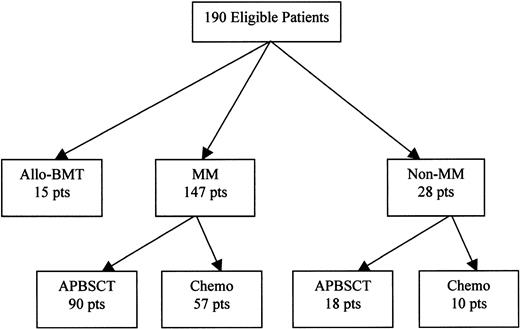
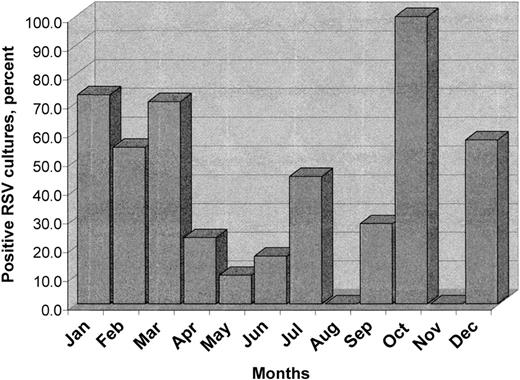
A total of 193 patients underwent nasopharyngeal washings for the detection of respiratory viral pathogens. Three were excluded because of concomitant infection with other respiratory viruses. Of the 190 patients eligible for evaluation, 108 were undergoing APBSCT (90 had multiple myeloma [MM]), 67 were receiving nonmyeloablative chemotherapy without stem cell transplantation (57 had MM), and the remaining 15 were undergoing allo-BMT (Figure 1). The RSV was isolated in 71 (37%) of 190 patients throughout the year, including summer (May to September), although more cases were diagnosed during the fall and winter seasons than spring and summer (59% versus 21%, respectively), P < .001 (Figure 2). Twenty-two (21%) of 105 patients tested during the summer season had RSV-positive cultures, including 12 (44%) of the 27 tested in July.
Distribution of participants according to underlying disease and therapy. MM indicates multiple myeloma; chemo, chemotherapy; APBSCT, autologous stem cell transplantation; allo-BMT, allogeneic bone marrow transplantation; pts, patients.
Distribution of participants according to underlying disease and therapy. MM indicates multiple myeloma; chemo, chemotherapy; APBSCT, autologous stem cell transplantation; allo-BMT, allogeneic bone marrow transplantation; pts, patients.
Monthly rate of recovery of RSV in nasopharyngeal washings from 190 cancer patients. Percentage of RSV-positive cultures for the entire population. RSV was isolated in 71 (37%) of 190 patients throughout the year, including summer (May to September). However, more cases were identified during the fall and winter seasons than spring and summer (59% versus 21%, respectively), P < .001. Twelve (44%) of 27 patients tested in July had RSV-positive cultures.
Monthly rate of recovery of RSV in nasopharyngeal washings from 190 cancer patients. Percentage of RSV-positive cultures for the entire population. RSV was isolated in 71 (37%) of 190 patients throughout the year, including summer (May to September). However, more cases were identified during the fall and winter seasons than spring and summer (59% versus 21%, respectively), P < .001. Twelve (44%) of 27 patients tested in July had RSV-positive cultures.
To avoid the potential confounding variables associated with various underlying diseases and allo-BMT, we first focused our analysis on the 147 patients with MM who had received cytotoxic chemotherapy with or without APBSCT. Patients included 100 men and 47 women, with a median age of 58 years (range, 31 to 82 years). Fifty-six patients (38%) had positive nasopharyngeal cultures for RSV with the same seasonal distribution as the entire group of 190 patients (data not shown).
A significantly higher proportion of myeloma patients undergoing APBSCT were positive for RSV (39 of 90, 43%) than those undergoing standard nonmyeloablative chemotherapy (17 of 57, 30%) (P = .003). Other baseline characteristics by RSV status were not significantly different (Table 1). The conditioning regimen and the number of CD34+ cells infused per kilogram were also comparable among the RSV-positive and RSV-negative patients who underwent APBSCT, P = .216 (data not shown).
Characteristics of 147 myeloma patients according to RSV status
. | RSV-positive . | RSV-negative . | P . |
|---|---|---|---|
| No. of patients | 56 | 91 | NA |
| Median age, y (range) | 57 (31-79) | 58 (31-82) | .380 |
| Male (%) | 37 (66) | 63 (69) | .690 |
| Autologous stem cell transplantation, no. of patients | 39 | 51 | NA |
| Nonmyeloablative chemotherapy, no. of patients | 17 | 40 | .003 |
| Before therapy | |||
| Positive chest x-rays, no. of patients (%)* | 4 (7) | 4 (4) | .480 |
| Renal failure (%) | 16 (29) | 23 (26) | .718 |
| Median dexamethasone, mg (range) | 1 030 (0-3 820) | 965 (0-10 585) | .913 |
| Median cycles of chemotherapy (range) | 3.28 (0-10) | 2.99 (0-16) | .241 |
| Pulmonary function tests | |||
| Median DLCO (range) | 110 (55-169) | 105 (25-174) | .265 |
| Median FEV1 (range) | 85 (40-122) | 88 (51-125) | .271 |
| Median FVC (range) | 86 (42-120) | 88 (52-122) | .351 |
| After therapy | |||
| Neutropenia, below 10 × 109/L | |||
| No. of patients | 53 | 70 | NA |
| Median days (range) | 7 (1-19) | 8 (2-24) | .388 |
| Lymphopenia, below 7 × 109/L | |||
| No. of patients | 54 | 80 | NA |
| Median days (range) | 25 (2-95) | 29 (1-197) | .221 |
| Mucositis | |||
| Grade I (%) | 5 (9) | 10 (11) | .802 |
| Grade II (%) | 12 (21) | 16 (18) | .238 |
| Grade III (%) | 9 (16) | 15 (16) | .507 |
| Grade IV (%) | 8 (14) | 5 (5) | .038 |
| All severe complications (%) | 14 (25) | 15 (17) | .295 |
| Hypoxemia (%) | 13 (23) | 9 (10) | .032 |
| Tracheobronchitis (%) | 6 (11) | 0 | .003 |
| Pneumonia (%) | 6 (11) | 9 (10) | .873 |
| Intubation (%) | 2 (4) | 2 (2) | .623 |
| Transfer to intensive care unit (%) | 5 (9) | 6 (7) | .603 |
| Death within 30 days of therapy (%) | 3 (5) | 2 (2) | .715 |
. | RSV-positive . | RSV-negative . | P . |
|---|---|---|---|
| No. of patients | 56 | 91 | NA |
| Median age, y (range) | 57 (31-79) | 58 (31-82) | .380 |
| Male (%) | 37 (66) | 63 (69) | .690 |
| Autologous stem cell transplantation, no. of patients | 39 | 51 | NA |
| Nonmyeloablative chemotherapy, no. of patients | 17 | 40 | .003 |
| Before therapy | |||
| Positive chest x-rays, no. of patients (%)* | 4 (7) | 4 (4) | .480 |
| Renal failure (%) | 16 (29) | 23 (26) | .718 |
| Median dexamethasone, mg (range) | 1 030 (0-3 820) | 965 (0-10 585) | .913 |
| Median cycles of chemotherapy (range) | 3.28 (0-10) | 2.99 (0-16) | .241 |
| Pulmonary function tests | |||
| Median DLCO (range) | 110 (55-169) | 105 (25-174) | .265 |
| Median FEV1 (range) | 85 (40-122) | 88 (51-125) | .271 |
| Median FVC (range) | 86 (42-120) | 88 (52-122) | .351 |
| After therapy | |||
| Neutropenia, below 10 × 109/L | |||
| No. of patients | 53 | 70 | NA |
| Median days (range) | 7 (1-19) | 8 (2-24) | .388 |
| Lymphopenia, below 7 × 109/L | |||
| No. of patients | 54 | 80 | NA |
| Median days (range) | 25 (2-95) | 29 (1-197) | .221 |
| Mucositis | |||
| Grade I (%) | 5 (9) | 10 (11) | .802 |
| Grade II (%) | 12 (21) | 16 (18) | .238 |
| Grade III (%) | 9 (16) | 15 (16) | .507 |
| Grade IV (%) | 8 (14) | 5 (5) | .038 |
| All severe complications (%) | 14 (25) | 15 (17) | .295 |
| Hypoxemia (%) | 13 (23) | 9 (10) | .032 |
| Tracheobronchitis (%) | 6 (11) | 0 | .003 |
| Pneumonia (%) | 6 (11) | 9 (10) | .873 |
| Intubation (%) | 2 (4) | 2 (2) | .623 |
| Transfer to intensive care unit (%) | 5 (9) | 6 (7) | .603 |
| Death within 30 days of therapy (%) | 3 (5) | 2 (2) | .715 |
Positive chest x-ray: presence of infiltrates. DLCO indicates carbon monoxide diffusing capacity; FEV1, forced expiratory volume in 1 second; FVC, forced vital capacity; and NA, not applicable.
The conditioning regimen for MM patients undergoing APBSCT consisted of high-dose melphalan alone (140 to 200 mg/m2) (71 patients, 78%) or in combination with other agents (20 patients, 22%).20 The chemotherapeutic regimens for MM patients not undergoing transplantation have been previously described.20
Neutropenia was observed among 123 patients (84%) for a median of 7 days (range, 1 to 24 days), and lymphopenia was present among 134 patients (91%) for a median of 27 days (range, 2 to 197 days). None of the patients were treated with ribavirin (aerosolized or intravenous) or IVIG.
Seventeen (12%) of the 147 MM patients had upper respiratory symptoms prior to initiation of chemotherapy: 9 (16%) of 56 among the RSV-positive patients and 8 (9%) of 90 among those who were negative patients, P = .20.
Fourteen (25%) of 56 RSV-positive patients developed severe respiratory complications as defined by our predetermined composite index, compared with 15 (17%) of 91 in the RSV-negative group (P = .295). Although not planned at study initiation, we also performed a post hoc analysis for each component of the composite index. This analysis identified a higher incidence of tracheobronchitis (10% versus 0%, P = .003) and hypoxemia (23% versus 10%, P = .032) in the RSV-positive patients. No differences between the RSV-positive and RSV-negative groups were observed regarding the incidence of pneumonia (11% versus 10%, P = .873), intubation and mechanical ventilation (4% versus 2%, P = .623), transfer to ICU (9% versus 7%, P = .603), or death (5% versus 2%, P = .715) (Table 1).
Because previous reports of RSV-related respiratory complications had focused on patients with respiratory symptoms only, we also performed a post hoc analysis of the rate of respiratory complications in this subset of patients. Of 24 RSV-positive patients with respiratory symptoms, 14 (58%) developed severe complications compared with 15 (65%) of 23 RSV-negative patients (P = .747). Of note, 32 (57%) of 56 RSV-positive patients had no respiratory symptoms.
A total of 134 patients were asymptomatic at baseline (78 APBSCT and 56 chemotherapy). Among the APBSCT group, 8 (25%) of 32 RSV-positive patients developed complications compared with 11 (24%) of 46 RSV-negative patients (P = .1). Among the asymptomatic patients undergoing chemotherapy, 3 (19%) of 16 RSV-positive patients developed complications compared with 5 (13%) of 40 RSV-negative patients, P = .1.
Univariate analysis of baseline factors (prior to chemotherapy) identified the following risk factors for respiratory complications: status of myeloma, P = .099; serum monoclonal protein levels, P = .011; renal failure, P = .0001; hemodialysis, P = .017; concomitant amyloidosis, P = .043; forced expiratory volume1, P = .0008; forced vital capacity, P = .0005; site of care (inpatient or outpatient), P = .018; chest x-ray abnormalities, P = .041; platelet counts, P = .004; and serum LDH, P = .005. Infection with RSV (P = .211) was not found to be a significant factor in this analysis.
Univariate analysis of the posttherapy variables revealed the following significant factors: fever, P = .0001; microbiologically documented infection (other than RSV), P = .054; clinically documented infection (other than RSV), P = .0000; cough, P = .0000; shortness of breath, P = .0000; URTI, P = .0001; and mucositis, P = .010.
Three risk factors for respiratory complications remained significant when multivariate analysis was performed: (1) renal failure at baseline (P = .0004; odds ratio [OR], 10.18; 95% confidence interval [CI], 2.8-36.8); (2) elevated serum LDH at baseline (P = .0008; OR, 9.2; 95% CI, 2.5-33.7); and (3) the development of mucositis (for grade I/II: P = .029; OR, 5.9; 95% CI, 1.2-29.1; for grade III/IV: P = .024; OR, 6.2; 95% CI, 2.5-33.7) (Table 2). Although not significant by univariate analysis (P = .211), RSV status was forced in the multivariate analysis but failed to achieve significance (P = .443).
Multivariate analysis of factors that predicted severe complications in patients with multiple myeloma (n = 147)
Variable . | No. of patients with complication (%); total = 29 . | No. of patients without complication (%); total = 118 . | P . | Odds ratio (95% CI) . |
|---|---|---|---|---|
| Before therapy | ||||
| Renal failure | 17 (59) | 22 (19) | .0004 | 10.2 (2.8-36.8) |
| LDH* | 14 (48) | 21 (18) | .0008 | 9.2 (2.5-33.7) |
| After therapy | ||||
| No mucositis | 6 (21) | 61 (52) | .046 | 1 |
| Mucositis grade I/II | 9 (31) | 34 (29) | .029 | 5.9 (1.2-29) |
| Mucositis grade III/IV | 14 (48) | 23 (20) | .024 | 6.2 (2.5-33.7) |
Variable . | No. of patients with complication (%); total = 29 . | No. of patients without complication (%); total = 118 . | P . | Odds ratio (95% CI) . |
|---|---|---|---|---|
| Before therapy | ||||
| Renal failure | 17 (59) | 22 (19) | .0004 | 10.2 (2.8-36.8) |
| LDH* | 14 (48) | 21 (18) | .0008 | 9.2 (2.5-33.7) |
| After therapy | ||||
| No mucositis | 6 (21) | 61 (52) | .046 | 1 |
| Mucositis grade I/II | 9 (31) | 34 (29) | .029 | 5.9 (1.2-29) |
| Mucositis grade III/IV | 14 (48) | 23 (20) | .024 | 6.2 (2.5-33.7) |
Increased serum LDH (> 190 IU/L).
In addition to the 147 myeloma patients described above, 28 patients had undergone APBSCT for other malignancies (breast and ovarian cancers and lymphoma), and 11 (39%) were RSV-positive. Only 1 (9%) of these 11 patients developed respiratory complications compared with 3 (18%) of 17 patients who were RSV negative.
Of the remaining 15 patients who had undergone allo-BMT, 1 of the 4 RSV-positive patients developed pneumonia compared with 2 of the 11 RSV-negative patients.
Thus, of the entire group of 190 patients, 19 (27%) of 71 RSV-positive patients developed respiratory complications compared with 24 of 119 RSV-negative patients (20%, P = .384).
Multivariate analysis was also applied to the entire group of 190 patients and yielded the same important prognostic variables identified for the 147 patients with myeloma, including baseline renal failure and elevated serum LDH, and development of mucositis (Table 3). Again, RSV status was forced into the multivariate analysis of the 190 patients but was not significant (P = .46; OR, 1.47; 95% CI, 0.53-4.08).
Multivariate analysis of factors that predicted severe complications in the entire population (MM and other cancers) (n = 190)
Variable . | No. of patients with complication (%); total = 36 . | No. of patients without complication (%); total = 154 . | P . | Odds ratio (95% CI) . |
|---|---|---|---|---|
| Before therapy | ||||
| Renal failure | 20 (56) | 26 (17) | .0001 | 8.3 (2.9-23.7) |
| LDH* | 15 (42) | 34 (22) | .017 | 3.4 (1.3-9.5) |
| After therapy | ||||
| No mucositis | 6 (17) | 68 (47) | .029 | 1 |
| Mucositis grade I/II | 13 (37) | 48 (33) | .013 | 5.7 (1.4-22.3) |
| Mucositis grade III/IV | 16 (46) | 30 (21) | .020 | 5.2 (1.3-20.6) |
Variable . | No. of patients with complication (%); total = 36 . | No. of patients without complication (%); total = 154 . | P . | Odds ratio (95% CI) . |
|---|---|---|---|---|
| Before therapy | ||||
| Renal failure | 20 (56) | 26 (17) | .0001 | 8.3 (2.9-23.7) |
| LDH* | 15 (42) | 34 (22) | .017 | 3.4 (1.3-9.5) |
| After therapy | ||||
| No mucositis | 6 (17) | 68 (47) | .029 | 1 |
| Mucositis grade I/II | 13 (37) | 48 (33) | .013 | 5.7 (1.4-22.3) |
| Mucositis grade III/IV | 16 (46) | 30 (21) | .020 | 5.2 (1.3-20.6) |
Increased serum LDH (> 190 IU/L).
An analysis of the drug acquisition cost of RSV-specific therapy was performed assuming 11 days of therapy with 500 mg/kg IVIG every other day plus 6 g aerosolized ribavirin daily, as per Whimbey et al.7 This hypothetical analysis yielded a drug acquisition cost of $20 720 per patient course, for a total of $500 000 for the 24 patients who were RSV-positive and had respiratory symptoms during the 12-month study period. This figure does not include the cost of delivering this combination therapy.
Discussion
This is the first study of the natural history of RSV infection in a homogeneous cancer population (same underlying disease, cytotoxic chemotherapy, and supportive care standards). This is also the first study to include a control group, a key element in determining the contribution of RSV infection to the observed morbidity and mortality in this critically ill group of patients. Several findings emerged from our study.
A significant proportion (37%) of our patients had positive nasopharyngeal cultures for RSV, a rate significantly higher than previously reported in cancer patients. This figure may, in fact, represent an underestimate, given the lower sensitivity of RSV cultures compared with polymerase chain reaction (PCR).21
Most important, the rate of respiratory complications among patients whose nasal cultures yielded RSV did not significantly differ from that of patients who were RSV-negative, although a post hoc analysis did reveal a higher rate of tracheobronchitis and hypoxemia among the former patients. Univariate and multivariate analyses failed to identify positive RSV culture as a risk factor for respiratory complications. Baseline renal failure and elevated serum LDH, and the development of mucositis after therapy, were the only predictors for the development of these complications.
Although more frequent during fall and winter, recovery of RSV from nasopharyngeal secretions occurred throughout the year, including the summer months.
In addition, most RSV-positive patients (57%) had no respiratory tract symptoms, indicating that the virus may colonize the respiratory tract of cancer patients without causing disease.
The recovery of RSV from nasopharyngeal cultures of asymptomatic adults has been described in 3 reports. In a surveillance study conducted between 1961 and 1964, Mufson et al showed a similar rate of recovery of RSV among asymptomatic adults (8 of 761, 1%) and those with respiratory symptoms (3 of 295, 1%).22 More recently, Hall et al described 34 asymptomatic patients with nasopharyngeal cultures yielding RSV,23 and Cubie et al detected RSV nucleic acid in 24% of patients with sudden infant death syndrome (SIDS) and 19% of controls (patients whose death was caused by trauma, congenital heart disease, metabolic disease, and other nonrespiratory illnesses).24
Several studies by the Centers for Disease Control and Prevention (CDC) and others also support our observation that RSV may be recovered from respiratory secretions during summer,24-30 with rates of 13% (32 of 245 patients)29 and 21% (39 of 184).30 In one study, a similar rate of recovery of RSV DNA was observed during the summer and winter seasons (5 of 18, 27%; versus 16 of 58, 27%; respectively).24
Catastrophic complications, including a high death rate among cancer patients receiving chemotherapy with or without stem cell support, have been reported as complications of RSV infection.1-9 Whimbey et al described 8 deaths among 12 autologous BMT patients who developed RSV pneumonia. Death was observed in all 5 patients who did not receive aerosolized ribavirin and IVIG or received this combination within 24 hours from respiratory failure compared with only 3 of 7 deaths in those who received early therapy.7 In another report of 17 patients with leukemia and RSV pneumonia, death was observed in 2 of 10 patients who received early RSV therapy compared with 6 of 7 among the late or no-therapy group.8 Among breast cancer patients undergoing APBSCT, 1 death was observed among the 5 patients who received early RSV therapy, while the only patient who was treated late died.5 By contrast, we have previously reported on 10 consecutive patients with myeloma who underwent APBSCT while having active RSV URTI and without receiving ribavirin. No patient developed LRTI, required transfer to intensive care, or died at a median follow-up of 8 months.17 Similarly, Cole et al described 18 cancer patients with RSV infection, half of whom received RSV-specific therapy. All 9 untreated and 8 of the 9 treated patients survived.31
In addition, the studies that reported severe clinical consequences of RSV infection and good response to RSV-specific therapy included a small number of patients and no control group (patients without RSV infection).1-9 In these studies, the complications observed in the RSV-positive patients were assumed to be caused by this virus. Without a control group, we would have also concluded, erroneously, that the rate of RSV-related respiratory complications was very high in our population (14 of 24, 58%). However, the respiratory complications in this population are multifactorial and include toxicity of the conditioning regimen, hemorrhage, fluid overload, pulmonary embolization, bronchiolitis obliterans, engraftment syndrome, and infections by microorganisms other than RSV.32-38 Indeed, a significant rate of infections with copathogens is not uncommon in cancer patients with RSV infection.1-3,6 Thus, determining the contribution of any of these multiple factors to the observed respiratory complications in a given patient is very difficult even after autopsy examination. Because of the above, the limited RSV-associated morbidity (in this study and others17,31 ), and the recovery of RSV in asymptomatic patients (in this study and others22-24 ), we submit that RSV cultures may simply represent colonization in a sizable proportion of patients. We suspect that the high rate of RSV complications reported by others1-9 may in fact be due to other causes.32-38 The recovery of RSV from the lower respiratory tract may represent, as with other microorganisms (eg, Candida),39-42 contamination from the colonized upper airways.
An increased rate of tracheobronchitis among RSV-positive patients was identified by post hoc analysis. This is not surprising given the propensity of RSV infection to cause tracheobronchitis as a result of development of necrosis, edema, and increased mucus secretion resulting in obstruction.43 Such patients may benefit from a short course of therapy with corticosteroids, as shown in several studies of children with RSV tracheobronchitis44-46 and croup.47-51 Steroid therapy is also supported by the linear dose-response relationship between concentration of steroids and reduction in pathology score in RSV pneumonia in cotton rats.52 Methylprednisolone (1 mg/kg/d) was given to some of our RSV-positive patients with tracheobronchitis with rapid symptomatic relief (anecdotal experience).
Our findings imply that cancer patients whose respiratory cultures yield RSV may not necessarily suffer severe respiratory complications when undergoing chemotherapy, even in the absence of RSV-specific therapy. Delaying chemotherapy in such patients may not be necessary. The presence of signs and symptoms of acute respiratory infection (regardless of RSV culture status) does, however, warrant delaying cytotoxic therapy. In addition, expensive and cumbersome RSV-specific therapy with ribavirin and IVIG was not given to our patients and does not appear to be required. In a recent study, Boeckh et al (M. Boeckh, oral communication, May 1996) utilized aerosolized ribavirin preemptively to prevent progression of RSV URTI to LRTI. A multivariate analysis of 177 allo-BMT patients with RSV infection (60 considered to have RSV disease) revealed that older age and mismatched or matched unrelated transplantation were the only risk factors for progression to LRTI. Aerosolized ribavirin, even when used preemptively, was not a favorable prognostic variable, indicating that RSV may not have caused the observed LRTI and/or that ribavirin was not effective. These results are further supported by a recent meta-analysis of randomized trials that failed to show any benefit from utilizing aerosolized ribavirin among immunocompetent children.53 The high cost of aerosolized ribavirin is further compounded by the need to hospitalize a large number of patients to deliver the recommended 18-hours-a-day therapy as recommended by Whimbey et al.4-9
The implications of our findings for infection control practices are also significant. Infection control measures are most likely to be beneficial at preventing infections caused by uncommon pathogens and that are likely to result in serious clinical consequences (eg, methicillin-resistant Staphylococcus aureus and Mycobacterium tuberculosis).54,55 The high rate of RSV respiratory tract colonization coupled with the low risk of RSV-related complications suggests that strict infection control measures are unlikely to be beneficial. Currently recommended measures include contact precautions for patients with symptoms of URTI until viral cultures are negative, mask and gowns with every contact by health care worker, and dismissal of workers with URTI symptoms until asymptomatic.14,56 These measures are also difficult to implement because of cost and severe nursing shortages. Furthermore, these hospital-based measures will have little effect on the overwhelming majority of RSV infections, because these are typically communityacquired and most cancer patients are cared for in the outpatient setting where these measures are very difficult to implement.
Our findings also imply that patients with baseline renal failure and/or elevated serum LDH and those who develop mucositis may be at high risk for pulmonary complications after cytotoxic chemotherapy. Closer monitoring for the development of respiratory complications in such patients is thus recommended.
The implications of our findings may also be relevant to research in this field. Future studies of RSV infections in cancer patients should always include a control group and be randomized if RSV-specific therapy is being evaluated.
Our study is limited by the small number of allo-BMT recipients. Thus, our observations cannot be extended to this patient population. It is also possible, but unlikely, that a much larger sample size of the non–allo-BMT group might have revealed a higher rate of respiratory complications in those patients whose respiratory cultures yielded RSV.
In conclusion, recovery of RSV from the respiratory tract of cancer patients is common, occurs throughout the year, and is associated with a low rate of attributable respiratory complications. If confirmed by other prospective studies, our findings carry important implications for the management of cancer patients.
Prepublished online as Blood First Edition Paper, October 2, 2003; DOI 10.1182/blood-2003-05-1425.
Supported in part by grant no. CA55819 from the National Cancer Institute, Bethesda, MD.
Presented in part at the 42nd annual meeting of the American Society of Hematology, San Francisco, CA, December 2, 2000.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.
We thank Drs Michael Boeckh, Cecilia Dignani, Paula North, and Gary Wheeler for their insightful comments.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal