Abstract
The tumor necrosis factor (TNF)–like ligand BAFF/BLyS (B-cell activating factor of the TNF family/B-lymphocyte stimulator) is a potent B-cell survival factor, yet its functional relationship with other B-cell surface molecules such as CD19 and CD40 is poorly understood. We found that follicular dendritic cells (FDCs) in human lymph nodes expressed BAFF abundantly. BAFF up-regulated a B cell–specific transcription factor Pax5/BSAP (Pax5/B cell–specific activator protein) activity and its target CD19, a major component of the B-cell coreceptor complex, and synergistically enhanced CD19 phosphorylation by B-cell antigen receptor (BCR). BAFF further enhanced B-cell proliferation, immunoglobulin G (IgG) production, and reactivity to CD154 by BCR/CD19 coligation and interleukin-15 (IL-15). Our results suggest that BAFF may play an important role in FDC–B-cell interactions through the B-cell coreceptor complex and a possibly sequential link between the T cell–independent and –dependent B-cell responses in the germinal centers.
Introduction
The tumor necrosis factor (TNF) superfamily ligand BAFF (B-cell activating factor of the TNF family), also known as BLyS (B-lymphocyte stimulator), TALL-1 (TNF and apoptosis leukocyte-expressed ligand-1), THANK (TNF homolog that activates apoptosis, nuclear factor–κB, and c-Jun NH2-terminal kinase), and zTNF4, is selectively expressed on macrophages, monocytes, and dendritic cells (DCs) regulated by interferon-γ (IFN-γ).1 BAFF acts as a potent B-cell growth factor and costimulator of immunoglobulin (Ig) production.2 BAFF binds to 3 separate receptors, including B-cell maturation antigen (BCMA), transmembrane activator and calcium-modulator and cyclophilin ligand (CAML)–interactor (TACI), and BAFF-R (BAFF receptor). Their expression is restricted mainly to B cells, and BAFF–BAFF-R interactions are required for immature B cells to survive beyond the first transitional stage (T1) in mice.3 BAFF administration in mice can potentiate antibody (Ab) responses and increase the number of plasma cells.4,5 In addition, mice transgenic for BAFF showed increased germinal center (GC) formation and developed autoimmune disorders such as production of autoantibodies and Ig deposition in kidney, considered to be symptoms of human systemic lupus erythematosus (SLE).6 Contrary to this, BAFF-deficient mice have significantly fewer marginal zone and follicular B cells in secondary lymphoid tissue,7 and TACI-Ig–treated mice lack GCs.8 Collectively, these finding suggest that BAFF has a crucial role in the control of GC B-cell development and humoral immune responses. However, the cellular and molecular mechanisms of BAFF functions in GCs are still obscure.
In humoral immune responses, follicular dendritic cell (FDC)–B-cell interactions in GCs play a crucial role in full activation of B cells.9 FDC–B-cell interactions require 2 distinct but synergistic signals. The first signal, delivered through the B-cell antigen receptor (BCR), is provided by antigen (Ag) itself and is responsible for the specificity of the Ab secreted. The second signal is derived from the B-cell coreceptor complex consisting of CD19, CD21 (CR2), and CD81 bound to complement (C) and is not Ag-specific.10 The B-cell coreceptor complex is essential for the rapid trapping of C-coated Ags by B cells and is critical for early protective Ab responses to lethal pathogens that rapidly multiply and quickly overwhelm the immune system.11,12 C3 component and its cleavage products such as C3d are particularly important for the efficient opsonization, lysis, and clearance of bacteria.13 Depletion of C3 in mice reduces T cell–dependent (TD) and some T cell–independent (TI) Ab responses.14 Similar defects of humoral immune responses are found in patients with genetic deficiencies in C3.15 In addition, C3-deficient mice have reduced TD and TI antibody responses.16-19 CD21 is expressed primarily by FDCs and mature B cells, and C3-coated Ags trapped by CD21 on FDCs are also captured by CD21 of the B-cell coreceptor.11 CD21-deficient mice have impaired humoral immunity due to defective maintenance of B cells within GCs,20 which is a similar phenotype to that of C3-deficient mice. Thereby, the binding of CD21 with C3-coated Ags in FDC–B-cell interactions may facilitate the processing and presentation of Ags at low concentrations, particularly in the absence of preformed or natural Abs where Fc receptor interactions may also facilitate immune complex processing.21 Activation of the B-cell coreceptor complex makes B cells more responsive to BCR signals, resulting in positive selection of B cells.11
In the B-cell coreceptor complex, CD19 is thought to functionally link CD21 with BCR following corecognition of C3d-coated Ags and is a key player in its activation.22 CD19 exerts its positive effect on BCR signaling by the phosphorylation-dependent recruitment of Vav and phosphatidylinositol-3 kinase (PI3K), which in turn enhances Ca flux, activation of the extracellular signal-regulated kinase 1/2 (ERK1/2), and ultimately B-cell proliferation.23-25 CD19 is rapidly phosphorylated on Tyr482 and Tyr513 following BCR ligation.26-28 Point mutations in CD19, Y482, and Y513 disrupt CD19 function in B-cell differentiation with reduction of splenic germinal centers, B cells, and class switching in vivo.29 Tyr513 is required for BCR-mediated activation by coupling of PI3K and Src family tyrosine kinases to CD19.30,31 Expression of CD19 is reported to be under the direct control of the B lineage–specific transcription factor Pax5/B cell–specific activator protein (Pax5/BSAP),32 which is implicated in isotype switching and cell proliferation of mature B cells in addition to B-cell development.33 Coligation of CD19 with BCR decreased the signaling threshold for BCR-dependent stimulation and augmented B-cell proliferation.23
However, the functional relationship of BAFF with the above B-cell molecules in coordinating B-cell responses in GCs is not fully clear. We show here that FDCs in GCs of human lymph nodes expressed BAFF and that BAFF stimulation synergistically enhanced the BCR signaling by phosphorylation of CD19. In the presence of interleukin-15 (IL-15), BAFF significantly increased B-cell proliferation and IgG production upon BCR and CD19 coligation. These B cells after BCR/CD19 coligation and BAFF stimulation were highly responsive to CD154 (CD40 ligand) stimulation. These findings suggest that BAFF can mediate FDC–B-cell interactions, facilitate B-cell selection through the B-cell coreceptor complex, and subsequently optimize B-cell responses by T-cell help in GCs.
Materials and methods
Cell preparation and cell cultures
Human peripheral blood mononuclear cells were isolated from pooled healthy donors by Ficoll-Hypaque density-gradient centrifugation. After depletion of monocytes by adherence to the plastic surface of culture dishes, E rosette–negative populations were collected with 5% sheep erythrocytes and Ficoll-Hypaque. Purified B cells were isolated by depletion of non-B cells using a B-cell isolation kit and autoMACS (Miltenyi Biotec, Auburn, CA). The resultant B-cell population was less than 2% CD14+, less than 1% CD3+, less than 2% CD57+, and more than 95% CD20+. CD14+ monocytes were recovered from the B cell–depleted fraction. Monocyte-derived dendritic cells corresponding to CD83+ cells were prepared as reported previously.34 Briefly, monocytes were cultured supplemented with 800 U/mL granulocyte-macrophage colony-stimulating factor (GM-CSF) (PeproTech, Rocky Hill, NJ) and 25 ng/mL IL-4 (PeproTech) for 4 days. The cells were then incubated with GM-CSF and 10 ng/mL TNF-α (PeproTech) for 7 days. Human B-cell lines including Ramos, P32/ISH, Raji, and Daudi cells were purchased from the Human Science Research Resources Bank (Osaka, Japan). Murine fibroblastic L cells expressing human CD32/FcγRII (DT70 cells)35 were used to coligate BCR and CD19 instead of FDCs. All cultures were conducted in RPMI 1640 medium supplemented with 25 mM HEPES (N-2-hydroxyethylpiperazine-N′-2-ethanesulfonic acid), 10% fetal calf serum, 2 mM l-glutamine, 1 mM sodium pyruvate, 5.5 × 10-2 mM 2-mercaptoethanol, 100 U/mL penicillin, and 100 U/mL streptomycin (all from Invitrogen, Carlsbad, CA). Mouse B cells were purified from the spleens of 8-week-old C57BL/6 mice (Japan SLC, Shizuoka, Japan) with the use of a mouse B-cell isolation kit and autoMACS (Miltenyi Biotec). The resultant B-cell population was more than 95% B220+.
Antibodies and reagents
The following Abs were used in this study: BAFF (TALL-1) (Santa Cruz Biotechnology, Santa Cruz, CA); phospho-CD19 (Tyr513), CD19 (Cell Signaling Technology, Beverly, MA); IgM, FLAG M2, β-actin (Sigma-Aldrich, St Louis, MO); Igκ, Igλ, fluorescein isothiocyanate (FITC)–labeled CD40, Pax5 (BD PharMingen, San Diego, CA); control mouse IgG1, CD19 (HD37 clone; DAKO, Glostrup, Denmark); FITC-labeled CD14, CD20, HLA-DR, phycoerythrin (PE)–labeled intercellular adhesion molecule-1 (ICAM-1) (CD54) (eBioscience, San Diego, CA); PE-labeled CD19 (Immunotech, Marseille, France); and PE-labeled CD21 (CALTAG, Burlingame, CA). The following reagents were used: IFN-γ (PeproTech), lipopolysaccharide (LPS) (Sigma-Aldrich), Staphylococcus aureus Cowan I (SAC) (Calbiochem, La Jolla, CA), recombinant human IL-2 (Invitrogen), recombinant human IL-15 (PeproTech), recombinant human BAFF (PeproTech), and recombinant human TACI-Fc chimera protein (TACI-Ig; Genzyme-Techne, Cambridge, MA) consisting of the extracellular domain of TACI and Fc portion of human IgG1.
Establishment of human BAFF transfectant
To construct a BAFF expression vector, a cDNA encoding full-length human BAFF protein (GenBank no. AF132600), subcloned from peripheral blood mononuclear cells (PBMNCs) using a reverse transcription–polymerase chain reaction (RT-PCR) method with linker primers, was inserted into pBluescript SK(+) (Stratagene, La Jolla, CA) at the EcoRI and BamHI sites and finally into the pBCMGSneo expression vector36 at the XhoI and NotI sites (pBCMGS-BAFF). Murine pre-B cells (cell line 300-19) were electroporated with pBCMGS-BAFF, and stable transfectants were selected by G418 treatment. Cells with high-density BAFF were cloned by fluorescence-activated cell sorter (FACS). The CD154 cDNA-transfected cells and the vector alone–transfected cells (mock) have been described previously.37 To coculture with B cells, transfectants were treated with mitomycin C (50 μg/mL; Sigma-Aldrich) for 30 minutes at 37°C.
Flow cytometric analysis
After incubation of arbitrary Ab with 2 μg/106 cells for 20 minutes on ice, cells were washed and resuspended in propidium iodide solution and analyzed using FACSCalibur and associated CellQuest software (both from Becton Dickinson, San Jose, CA). In some experiments, FITC-labeled rabbit antibody to mouse IgG1 (ICN/CAPPEL, Aurora, OH) or FITC-labeled swine antibody to goat IgG (CALTAG) was used as a second Ab. Isotype-matched mouse IgG control was used throughout the studies and always reacted with less than 5% of the cells.
Generation of antihuman BAFF monoclonal antibody
A BALB/c mouse was immunized with human BAFF-expressing transfectant cells 3 times at 10-day intervals. Three days after the final immunization, the splenocytes were fused with NS-1 cells (purchased from Human Science Research Resources Bank), and HAT (hypoxanthine aminopterin thymidine) selection and cloning of hybridomas were performed simultaneously using a hybridoma cloning kit (ClonaCell-HY; StemCell Technologies, Vancouver, BC). Antihuman BAFF designated 1D6 (mouse IgG1, κ) was established and purified from ascitic fluid by using MAbTrap Kit (Amersham Biosciences, Piscataway, NJ).
Immunohistochemical analysis
Biopsied human lymph nodes were fixed in buffered formalin, routinely processed, and embedded in paraffin. After preparation of serial sections, deparaffinization and microwave antigen retrieval using citrate buffer (pH 6.0) were performed. For immunohistochemistry, the sections were stained by the streptavidin-biotin-peroxidase method (Immunotech) using 1D6 monoclonal antibody (mAb), anti-CD21 mAb (2G9; Novocastra, Newcastle, United Kingdom), and anti-FDC mAb (CNA.42; DAKO), accordingly to the instructions provided by the manufacturer. The sections treated with normal mouse serum served as negative controls.
Immunoblot analysis
Phosphate-buffered saline (PBS)–washed cell pellets were resuspended with 0.5% sodium dodecyl sulfate (SDS) solution and boiled for 5 minutes. Proteins (5 to 8 μg) were separated by SDS-polyacrylamide gel electrophoresis (SDS-PAGE), transferred to an Immobilon-P membrane (Millipore, Billerica, MA), blocked with 5% skim milk, and immunoblotted with arbitrary Ab and horseradish peroxidase (HRP)–labeled secondary Ab. Blotting was developed by using the enhanced chemiluminescent substrate (SuperSignal West Pico; Pierce Chemical, Rockford, IL) and visualized with a LumiVision analyzer (Taitec, Tokyo, Japan). The densitometric analysis was performed by using a LumiVision analyzer (Taitec).
Electrophoretic mobility shift assay (EMSA)
We carried out electrophoretic mobility shift assay (EMSA) with Pax5 gel shift oligonucleotides (Santa Cruz) and mutant oligonucleotides (Santa Cruz). These oligonucleotides were 3′ end-labeled with digoxigenin (DIG) by the DIG Oligonucleotide 3′-end Labeling kit (Roche Diagnostics, Penzberg, Germany). Next, 2.5 ng labeled oligonucleotide was incubated at room temperature for 20 minutes with 1 μg poly–deoxy-inosinic-deoxycytidylic acid (dIdC) (Roche Diagnostics) and 5 μg nuclear extracts that were isolated by nuclear extract kit (Active Motif, Carlsbad, CA) in a total volume of 20 μL. DNA probe and protein complexes were resolved by electrophoresis through 5% polyacrylamide gel at 150 V for 45 minutes in 0.5 × Tris–borate–ethylenediaminetetraacetic acid (TBE). To determine the specificity of Pax5 binding activity, a 50-fold excess of unlabeled Pax5 probe was added. For supershift experiments, nuclear extracts were incubated with 2 μL anti-Pax5 antibody for 30 minutes. DNA probes were blotted onto a Hybond-N+ (Amersham Biosciences) and detected using DIG Luminescent Detection kit (Roche Diagnostics) and an ECL-Mini Camera (Amersham Biosciences).
Construction of 3 × FLAG-sBAFF fusion protein
A short version of soluble BAFF (sBAFF/short, amino acid [aa] Gln136-Leu285)38 was amplified using PCR. sBAFFs were resubcloned as HindIII-BamHI fragments into the 3 × FLAG-CMV-9 vector (Sigma-Aldrich). After transfection of the 3 × FLAG-sBAFF vector into COS-7 with cationic liposome reagent (Lipofectamine reagent; Invitrogen), supernatants were collected. Binding of 3 × FLAG-sBAFF to B cells was evaluated by FACS analysis using anti-FLAG M2 mAb (IgG1) and FITC-labeled antimouse IgG1 Ab.
Real-time PCR
Each cDNA sample was analyzed in duplicate by a method for real-time quantitative RT-PCR using the GeneAmp 5700 sequence detection system (Applied Biosystems, Foster City, CA). Quantitative assessment of DNA amplification was detected through the dye SYBR Green according to the manuals provided by the manufacturer for SYBR Green PCR Master Mix (Applied Biosystems). The thermal cycling conditions were 50°C for 2 minutes and 95°C for 10 minutes, followed by 40 cycles of amplification at 95°C for 15 seconds and 60°C for 1 minute for denaturing, annealing-extension, respectively. The following sense and antisense primers were used. TACI: 5′-CAATGTGCATACTTCTGTGAGA-3′ and 5′-AACTTCTCCACTCCGCTGTCTCCTGA-3′; BCMA: 5′-CAACCATTCTTGTCACCACGAA-3′ and 5′-TCTATCTCCGTAGCACTCAAAGCA-3′; BAFF-R: 5′-CAAGGTCATCATTCTGTCTCCG-3′ and 5′-CGGCTCCCTGCTATTGTTGCTCA-3′; and β-actin: 5′-TCACCCACACTGTGCCCATCTACGA-3′ and 5′-CAGCGGAACCGCTCATTGCCAATGG-3′.
Proliferation assay
Purified B cells from human peripheral blood were cultured in a 96-well plate (1 × 105 per well) with mitomycin-treated transfectants expressing BAFF or CD154 (2 × 104 per well) in the presence or absence of SAC and IL-2 (50 U/mL). For coligation experiments, B cells pretreated with anti-BCR mAbs (anti-Igκ and anti-Igλ, 1 μg/mL) and/or anti-CD19 mAb (0.5 μg/mL) were cultured with or without sBAFF (0.1 μg/mL) in a 96-well plate (1 × 105 per well) precoated with mitomycin-treated DT70 cells (2 × 104 per well) in the presence or absence of IL-15 (50 ng/mL). In some experiments, mitomycin-treated CD154 transfectant was also added to the culture (1 × 104 per well). B-cell proliferation was quantitated by pulsing the cells during the last 18 hours of a total of 72 hours of culture with 0.5 μCi (18.5 kBq) per well of [3H]thymidine (Amersham Biosciences).
Detection and quantification of in vitro IgG secretion
Human peripheral blood B cells pretreated with anti-BCR mAbs (anti-Igκ and anti-Igλ, 1 μg/mL) and/or anti-CD19 mAb (0.5 μg/mL) were cultured with or without sBAFF (0.1 μg/mL) in a 96-well plate (1 × 105 per well) precoated with mitomycin-treated DT70 cells (2 × 104 per well) in the presence or absence of IL-15 (50 ng/mL). In some experiments, mitomycin-treated CD154 transfectant was also added to the culture (1 × 104 per well). IgG levels in the supernatants after 10 days of culture were determined by enzyme-linked immunosorbent assay (ELISA) using antihuman Ig (Southern Biotechnology, Birmingham, AL) as the capture Ab and HRP-labeled goat antihuman IgG (Southern Biotechnology) as the detector Ab. After addition of p-nitrophenyl phosphate substrate (Sigma-Aldrich), the amount of IgG present was assessed by spectrophotometric analysis at 490 nm using a model 550 Microplate Reader (Bio-Rad Laboratories, Hercules, CA).
Statistical analysis
All data were expressed as mean ± SD. Differences between groups were examined for statistical significance using the Student t test. A P value less than .05 denoted the presence of a statistically significant difference.
Results
Establishment of antihuman BAFF mAb and its identification of FDCs in GCs
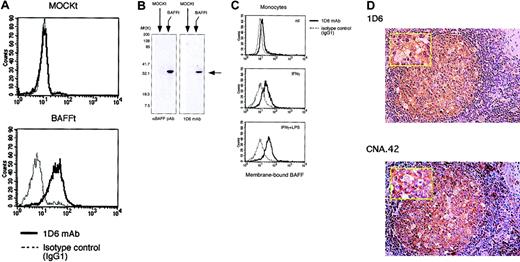
To evaluate the role of BAFF in human B-cell responses, we developed an antihuman BAFF mAb. For this purpose, we first established a transfectant stably expressing human BAFF on the cell surface and analyzed the B-cell costimulatory activity. BAFF cDNA was cloned into a mammalian expression vector pBCMGS-neo from RNA of peripheral blood mononuclear cells by RT-PCR and transfected into murine pre–B-cell line 300-19 cells. Stable expression of human BAFF on the cDNA transfectant but not on mock-transfected cells was verified by functional and immunoblot analyses (Figures 1 and 2A-B). Peripheral blood B cells were cultured with mitomycin-treated transfectants in the presence of suboptimal doses of SAC and IL-2 for BCR stimulation and activation. Flow cytometric analysis showed that addition of BAFF cDNA-transfected cells or CD154 transfectant clearly up-regulated cell surface expression of CD40, ICAM-1, and HLA-DR on B cells compared with that by mock transfectant (Figure 1A). Consistent with the previous report,38 the level of [3H]thymidine incorporation of B cells induced by BAFF cDNA-transfected cells was increased in a dose-dependent manner with levels comparable to those by CD154 transfectants while mock transfectant showed almost the basal level (Figure 1B). Immunoblot analysis using commercially available polyclonal antihuman BAFF Ab revealed that BAFF cDNA-transfected cells expressed the BAFF molecule. These results indicate that BAFF cDNA-transfected cells expressed functional BAFF molecules on the cell surface, which could deliver a B-cell costimulatory signal. This BAFF transfectant was used for further experiments (Figures 3,5). We then immunized a BALB/c mouse with BAFF transfectant and fused the splenocytes with NS-1 myeloma cells. The hybridomas producing antihuman BAFF mAb were screened by specific reactivity to BAFF transfectant. As shown in Figure 2A-B, one mAb, designated 1D6, was obtained, which bound to BAFF transfectant but not to mock transfectant. 1D6 mAb reacted with activated monocytes exposed to IFN-γ, and its expression was further enhanced by the additional treatment with LPS (Figure 2C). As reported previously,39 monocyte-derived DCs also expressed BAFF (data not shown). Interestingly, we found that 1D6-positive cells were mainly localized in GCs of human lymph nodes where the cells with long processes formed a reticular meshwork (Figure 2D). Immunohistochemical analysis using serial tissue sections revealed that the cells stained with 1D6 mAb were also stained with anti-FDC mAb (Figure 2D) and anti-CD21 mAb (data not shown), suggesting that they were FDCs.
Functional characterization of BAFF transfectant. (A) Human peripheral blood B cells (1 × 105 per well) were cultured with or without mitomycin-treated transfected cells (2 × 104 per well) in the presence or absence of SAC (0.0002% [wt/vol]) and IL-2 (50 U/mL). After treatment for 48 hours, the expression level of CD40, ICAM-1, and HLA-DR was analyzed by FACS analysis. (B) B cells (1 × 105 per well) were cocultured in triplicate in 96-well plates with mitomycin-treated transfected cells (2 × 104 per well) in the presence or absence of SAC (0.0002% [wt/vol] or 0.0004% [wt/vol]) and IL-2 (50 U/mL), and [3H]thymidine (0.5 μCi [18.5 kBq] per well). Incorporation of B cells was measured during the last 18 hours of a total of 72 hours. Data are mean ± SD of triplicate wells and are representative of 3 different experiments.
Functional characterization of BAFF transfectant. (A) Human peripheral blood B cells (1 × 105 per well) were cultured with or without mitomycin-treated transfected cells (2 × 104 per well) in the presence or absence of SAC (0.0002% [wt/vol]) and IL-2 (50 U/mL). After treatment for 48 hours, the expression level of CD40, ICAM-1, and HLA-DR was analyzed by FACS analysis. (B) B cells (1 × 105 per well) were cocultured in triplicate in 96-well plates with mitomycin-treated transfected cells (2 × 104 per well) in the presence or absence of SAC (0.0002% [wt/vol] or 0.0004% [wt/vol]) and IL-2 (50 U/mL), and [3H]thymidine (0.5 μCi [18.5 kBq] per well). Incorporation of B cells was measured during the last 18 hours of a total of 72 hours. Data are mean ± SD of triplicate wells and are representative of 3 different experiments.
Characterization of 1D6 mAb and identification of BAFF-positive cells within GCs. (A) FACS analysis of BAFF expression using 1D6 mAb. BAFF transfectants and mock transfectants were stained with 1D6 mAb (solid lines) or isotype-matched control Ab (IgG1) (dashed lines) followed by FITC-conjugated Ab to mouse IgG1. (B) Cell lysates were prepared from BAFF transfectants and mock transfectants and subjected to immunoblotting analysis using 1D6 mAb or antihuman BAFF (TALL-1) polyclonal Ab. (C) Peripheral blood monocytes (CD14+ cells more than 98%) were activated by IFN-γ (1000 U/mL) and LPS (10 ng/mL) for 24 hours. After blocking with normal mouse serum plus control IgG1 mouse mAb, BAFF expression was evaluated by FACS analysis using 1D6 mAb. (D) BAFF-positive cells or FDCs (brown) localized in GCs, as determined by immunohistochemical staining of serial human lymph node sections with 1D6 mAb or CNA.42 mAb, respectively (magnification × 200). (Inset) High magnification (× 400). Nuclei were visualized by hematoxylin staining. Data shown are representative of 3 different experiments.
Characterization of 1D6 mAb and identification of BAFF-positive cells within GCs. (A) FACS analysis of BAFF expression using 1D6 mAb. BAFF transfectants and mock transfectants were stained with 1D6 mAb (solid lines) or isotype-matched control Ab (IgG1) (dashed lines) followed by FITC-conjugated Ab to mouse IgG1. (B) Cell lysates were prepared from BAFF transfectants and mock transfectants and subjected to immunoblotting analysis using 1D6 mAb or antihuman BAFF (TALL-1) polyclonal Ab. (C) Peripheral blood monocytes (CD14+ cells more than 98%) were activated by IFN-γ (1000 U/mL) and LPS (10 ng/mL) for 24 hours. After blocking with normal mouse serum plus control IgG1 mouse mAb, BAFF expression was evaluated by FACS analysis using 1D6 mAb. (D) BAFF-positive cells or FDCs (brown) localized in GCs, as determined by immunohistochemical staining of serial human lymph node sections with 1D6 mAb or CNA.42 mAb, respectively (magnification × 200). (Inset) High magnification (× 400). Nuclei were visualized by hematoxylin staining. Data shown are representative of 3 different experiments.
Up-regulation of Pax5/BSAP and CD19 by BAFF. Human peripheral blood B cells were cultured with or without mitomycin-treated transfectants at a ratio of 10:2 in the presence or absence of SAC (0.0002% [wt/vol]) and IL-2 (50 U/mL). (A) After the treatment for 48 hours, the expression level of CD19, CD20, and CD21 was evaluated by FACS analysis. After treatment for 48 hours in the above conditions (A), cell lysates and nuclear extracts were prepared for immunoblot analysis using anti-Pax5 Ab (B) and EMSA using oligonucleotides containing Pax5 binding site (C), respectively. Lane 1, Pax5 oligonucleotide alone; lane 2, Pax5 mutant oligonucleotide alone; lane 3, B cell alone plus Pax5 oligonucleotides; lane 4, SAC/IL-2–stimulated B cells plus mock transfectants plus Pax5 oligonucleotides; lane 5, SAC/IL-2–stimulated B cells plus CD154 transfectants plus Pax5 oligonucleotides; lane 6, SAC/IL-2–stimulated B cells plus BAFF transfectants plus Pax5 oligonucleotides; lane 7, SAC/IL-2–stimulated B cells plus BAFF transfectants plus Pax5 mutant oligonucleotides; lane 8, SAC/IL-2–stimulated B cells plus BAFF transfectants plus Pax5 oligonucleotides plus competitor; lane 9, SAC/IL-2–stimulated B cells plus BAFF transfectants plus Pax5 oligonucleotides plus anti-Pax5 Ab. NS indicates nonspecific band. Data shown are representative of 3 different experiments.
Up-regulation of Pax5/BSAP and CD19 by BAFF. Human peripheral blood B cells were cultured with or without mitomycin-treated transfectants at a ratio of 10:2 in the presence or absence of SAC (0.0002% [wt/vol]) and IL-2 (50 U/mL). (A) After the treatment for 48 hours, the expression level of CD19, CD20, and CD21 was evaluated by FACS analysis. After treatment for 48 hours in the above conditions (A), cell lysates and nuclear extracts were prepared for immunoblot analysis using anti-Pax5 Ab (B) and EMSA using oligonucleotides containing Pax5 binding site (C), respectively. Lane 1, Pax5 oligonucleotide alone; lane 2, Pax5 mutant oligonucleotide alone; lane 3, B cell alone plus Pax5 oligonucleotides; lane 4, SAC/IL-2–stimulated B cells plus mock transfectants plus Pax5 oligonucleotides; lane 5, SAC/IL-2–stimulated B cells plus CD154 transfectants plus Pax5 oligonucleotides; lane 6, SAC/IL-2–stimulated B cells plus BAFF transfectants plus Pax5 oligonucleotides; lane 7, SAC/IL-2–stimulated B cells plus BAFF transfectants plus Pax5 mutant oligonucleotides; lane 8, SAC/IL-2–stimulated B cells plus BAFF transfectants plus Pax5 oligonucleotides plus competitor; lane 9, SAC/IL-2–stimulated B cells plus BAFF transfectants plus Pax5 oligonucleotides plus anti-Pax5 Ab. NS indicates nonspecific band. Data shown are representative of 3 different experiments.
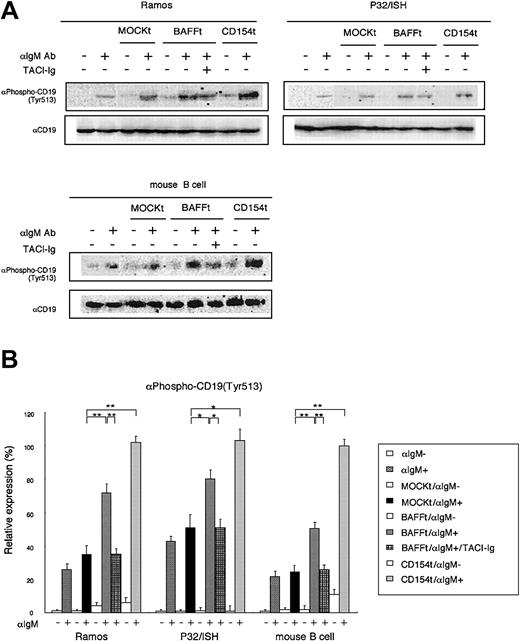
Synergistic effect of BAFF CD19 phosphorylation by BCR. (A) After coculture of human B-cell lines or murine splenic B cells with mitomycin-treated transfectants at a ratio of 10:2 for 3 hours, BCR ligation was induced by antihuman or antimouse IgM Ab (0.5 μg/mL). After 5 minutes, the reaction was terminated and cell lysates were prepared for immunoblot analysis using antiphospho CD19 (Tyr513) Ab. The expression levels of phophorylated CD19 are presented in the histogram in panel B. CD19 was detected as a loading control. For the blocking assay, TACI-Ig fusion protein was used at 0.4 μg/mL. Data shown are representative of 3 different experiments and are presented as mean ± SD; *P < .05; **P < .01.
Synergistic effect of BAFF CD19 phosphorylation by BCR. (A) After coculture of human B-cell lines or murine splenic B cells with mitomycin-treated transfectants at a ratio of 10:2 for 3 hours, BCR ligation was induced by antihuman or antimouse IgM Ab (0.5 μg/mL). After 5 minutes, the reaction was terminated and cell lysates were prepared for immunoblot analysis using antiphospho CD19 (Tyr513) Ab. The expression levels of phophorylated CD19 are presented in the histogram in panel B. CD19 was detected as a loading control. For the blocking assay, TACI-Ig fusion protein was used at 0.4 μg/mL. Data shown are representative of 3 different experiments and are presented as mean ± SD; *P < .05; **P < .01.
Up-regulation of Pax5/BSAP and CD19 by BAFF
The above finding that FDCs in GCs expressed abundant BAFF raised the possibility that BAFF mediates FDC–B-cell interactions in GCs. To test this notion, we examined the effect of BAFF on the expression of CD19 and CD21 on peripheral blood B cells. CD19 and CD21 together compose the B-cell coreceptor complex that plays a crucial role in B-cell responses of FDC–B-cell interactions.10 As shown in Figure 3A, BAFF transfectant exhibited a slight but definite up-regulation of CD19 and CD20 expression, but not CD21 expression, on B cells compared with mock transfectant. Because the expression levels of CD19 and CD20 are directly regulated by a B cell–specific transcription factor Pax5/BSAP,33,40 we next examined the effect of BAFF on Pax5 expression and its DNA binding activity by immunoblot analysis and EMSA. As shown in Figure 3B-C, consistent with up-regulated CD19 and CD20 expression by BAFF transfectant, BAFF transfectant as well as CD154 transfectant clearly increased Pax5 expression and its DNA binding activity in B cells. These results suggest that BAFF induced Pax5 activation and subsequently CD19 and CD20 up-regulation.
Synergistic effect of BAFF on CD19 phosphorylation by BCR
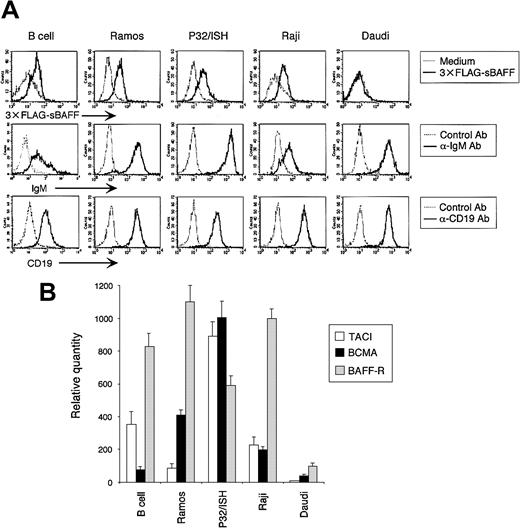
To further confirm the functional relationship between BAFF and the B-cell coreceptor complex, we next examined the effect of BAFF on phosphorylation of CD19 induced by BCR ligation. BCR signaling is known to phosphorylate CD19 on Tyr513, resulting in lowering of the signaling thresholds for B-cell activation, which in turn amplifies BCR signaling.31 No CD19 phosphorylation was detected in human peripheral blood B cells after BCR ligation (data not shown), probably due to the relatively lower expression of BCR, CD19, and receptors for BAFF (Figure 4). We then estimated the expression of these molecules in various human B-cell lines by flow cytometric and real-time RT-PCR analyses. For this purpose, we prepared recombinant soluble BAFF, which contains the receptor binding domain and an N-terminal FLAG tag, and screened for B-cell lines with high affinity to this recombinant soluble BAFF. As shown in Figure 4A, Ramos, P32/ISH, and Raji, but not Daudi, cell lines exhibited a high affinity to soluble BAFF. In accordance with this, the transcription level of 3 receptors for BAFF, including TACI, BCMA and BAFF-R, revealed high expression in these 3 cell lines (Figure 4B). Among these 3 cell lines, Ramos and P32/ISH cell lines showed the strongest expression of BCR and CD19 (Figure 4A). We then evaluated the effect of BAFF on CD19 phosphorylation on Tyr513 following BCR ligation. As shown in Figure 5, BAFF as well as CD154 stimulation synergistically enhanced the phosphorylation level of CD19 in Ramos cells (P < .01) and modestly did so in P32/ISH cells (P < .05) after BCR ligation, which was significantly suppressed by TACI-Fc chimera protein (TACI-Ig). Because 1D6 mAb cannot block the binding of BAFF with its receptors, we used TACI-Ig for blocking experiments. On the other hand, Raji cells failed to induce obvious phosphorylation of CD19 (data not shown), which was reflected by the low level of surface BCR compared with other cell lines (Figure 4A). Daudi cells showed no difference in CD19 phosphorylation level with or without BAFF stimulation (data not shown), probably due to undetectable expression of receptors for BAFF (Figure 4). In addition, we examined the effect of BAFF on CD19 phosphorylation in murine splenic B cells after BCR ligation because it has been reported that BCR-mediated CD19 phosphorylation is well established in murine B cells31 and that human BAFF is active on mouse B cells.1 BAFF stimulation synergistically enhanced BCR-mediated CD19 phosphorylation in murine splenic B cells (P < .01), which was significantly suppressed by TACI-Ig. Taken together, these results indicated that BAFF synergistically amplified CD19 phosphorylation induced by BCR ligation, which is relevant to enhanced B-cell activation mediated by the B-cell coreceptor complex.
BAFF receptor expression in human B cells. (A) Cells were stained with 3 × FLAG-sBAFF supernatant followed by anti-FLAG M2 mAb with FITC-labeled antimouse IgG1 Ab, or anti-IgM Ab followed by FITC-labeled antigoat Ab, or directly PE-labeled anti-CD19 mAb. Expression was analyzed by FACS. (B) The expression of BAFF receptors was evaluated by real-time quantitative RT-PCR for TACI, BCMA, and BAFF-R, as described in Figure 3B. The amount of transcription was normalized to the level of β-actin and designated as a relative quantity. Data shown are representative of 3 different experiments.
BAFF receptor expression in human B cells. (A) Cells were stained with 3 × FLAG-sBAFF supernatant followed by anti-FLAG M2 mAb with FITC-labeled antimouse IgG1 Ab, or anti-IgM Ab followed by FITC-labeled antigoat Ab, or directly PE-labeled anti-CD19 mAb. Expression was analyzed by FACS. (B) The expression of BAFF receptors was evaluated by real-time quantitative RT-PCR for TACI, BCMA, and BAFF-R, as described in Figure 3B. The amount of transcription was normalized to the level of β-actin and designated as a relative quantity. Data shown are representative of 3 different experiments.
B-cell responses induced by BAFF with the B-cell coreceptor complex
The above findings encouraged us to examine the role of BAFF in B-cell responses mediated by the B-cell coreceptor complex in FDC–B-cell interactions. For this purpose, we measured the effect of BAFF on human peripheral blood B-cell proliferation and in vitro IgG production under the conditions of coligation of BCR with CD19 in the presence of IL-15. To mimic FDC–B-cell interactions through the coengagement of BCR and CD19/CD21 with C3-coated Ags, we used murine fibroblastic L cells expressing CD32/FcγRII to cross-link anti-BCR and anti-CD19 mAbs bound to B cells instead of FDCs and used soluble recombinant BAFF instead of BAFF transfectant for increasing cell-cell contacts between peripheral blood B cells and L cells. IL-15 is produced by various Ag-presenting cells and is indispensable for the DC-induced IgG secretion in vitro.39 Under these conditions, BAFF clearly up-regulated CD40 expression on B cells in the presence of BCR/CD19 coligation, which was completely inhibited by TACI-Ig, although BCR/CD19 coligation itself showed less up-regulation of CD40 (Figure 6A). Coligation of BCR/CD19 resulted in an increase of [3H]thymidine incorporation in B cells compared with that induced by anti-BCR or anti-CD19 mAb alone, as reported previously.23 Importantly, BAFF further enhanced B-cell proliferation induced by coligation of BCR/CD19 (P < .05), which was completely inhibited by TACI-Ig (Figure 6B). Consistent with B-cell proliferation, BAFF further enhanced B-cell IgG production in the presence of BCR/CD19 coligation (P < .01), which was also completely inhibited by TACI-Ig (Figure 6C). These results clearly indicated that BAFF can facilitate B-cell responses mediated by the B-cell coreceptor complex, suggesting that BAFF can facilitate B-cell selection by FDCs in GCs.
Effects of BAFF on B-cell responses with coligation of BCR and CD19. Human peripheral blood B cells (1 × 105) pretreated with anti-BCR mAbs (anti-Igκ and anti-Igλ; 1 μg/mL), anti-CD19 mAb (0.5 μg/mL), and/or control mAb (1 μg/mL) were cultured with or without sBAFF (0.1 μg/mL) in triplicate in 96-well plates precoated with mitomycin-treated DT70 cells (2 × 104 per well) in the presence or absence of IL-15 (50 ng/mL) together with or without mitomycin-treated CD154 transfectant (1 × 104 per well). (A) After treatment for 48 hours, the expression level of CD40 was analyzed by FACS analysis. (B) [3H]thymidine (0.5 μCi [18.5 kBq] per well) incorporation of B cells was measured during the last 18 hours of a total of 72 hours. (C) After 10 days, IgG concentration in the supernatant was measured by ELISA. For the blocking assay, TACI-Ig fusion protein was used at 0.4 μg/mL. The values are reported as the mean ± SD of triplicate wells, and data shown are representative of 3 different experiments; *P < .05; **P < .01.
Effects of BAFF on B-cell responses with coligation of BCR and CD19. Human peripheral blood B cells (1 × 105) pretreated with anti-BCR mAbs (anti-Igκ and anti-Igλ; 1 μg/mL), anti-CD19 mAb (0.5 μg/mL), and/or control mAb (1 μg/mL) were cultured with or without sBAFF (0.1 μg/mL) in triplicate in 96-well plates precoated with mitomycin-treated DT70 cells (2 × 104 per well) in the presence or absence of IL-15 (50 ng/mL) together with or without mitomycin-treated CD154 transfectant (1 × 104 per well). (A) After treatment for 48 hours, the expression level of CD40 was analyzed by FACS analysis. (B) [3H]thymidine (0.5 μCi [18.5 kBq] per well) incorporation of B cells was measured during the last 18 hours of a total of 72 hours. (C) After 10 days, IgG concentration in the supernatant was measured by ELISA. For the blocking assay, TACI-Ig fusion protein was used at 0.4 μg/mL. The values are reported as the mean ± SD of triplicate wells, and data shown are representative of 3 different experiments; *P < .05; **P < .01.
Finally, to examine the effect of BAFF on B-cell responses to CD154 after BCR/CD19 coligation, we added the CD154 transfectant to the above culture conditions because results shown in Figure 1A and Figure 6A clearly showed up-regulation of CD40 expression on B cells in response to BAFF. As expected, the CD154 transfectant additively enhanced B-cell proliferation and IgG production induced by BAFF in the presence of BCR/CD19 coligation (P < .05) (Figure 6B-C). These results suggest that BAFF can help the same B cells to prepare for sequential TD B-cell responses after FDC–B-cell interactions.
Discussion
The B-cell zone of primary and secondary follicles contains a network of FDCs. FDCs appear to be specialized to capture Ags in the form of opsonized Ags, such as immune complexes consisting of Ag, Ab, and C, by receptors for Fc and C. Ags are not internalized but remain intact on the FDC surface, where they can be recognized by B cells. Thus, FDCs are potent accessory cells for B cells in GCs, but the molecular basis of their activity is not understood. Several important molecules involved in FDC–B-cell interactions have been identified by blocking the ligands and receptors on FDCs and/or B cells.9 For example, the engagement of CD21 of the B-cell coreceptor complex by C-coated Ags on FDCs delivers a crucial signal through CD19 that markedly augments the stimulation delivered by the binding of Ags to the BCR. Thus, the B-cell coreceptor complex plays a pivotal role in B-cell selection, in which only B cells with higher affinity can be positively selected even in low concentrations of Ags and rewarded with a survival signal. The B-cell receptor complex thus promotes B-cell responses. During their passage through GCs, mature B cells undergo apoptosis if they encounter self-Ags or if they are not positively selected by an antigenic signal.
In this study, we provide several evidences for a new role of BAFF in B-cell responses in GCs through FDC–B-cell interactions. First, we developed a novel antihuman BAFF mAb (1D6). Immunohistochemical analysis using 1D6 mAb demonstrated that FDCs in GCs of human lymph nodes expressed BAFF abundantly (Figure 2). To date, although it has been reported that BAFF is produced by macrophages, monocytes, DCs, and activated neutrophils,2,41 the present study is the first to demonstrate that FDCs in GCs of human lymph nodes express BAFF. This finding prompted us to perform further experiments.
We next showed that BAFF stimulation enhanced the expression of a B cell–specific transcription factor, Pax5/BSAP, and its target molecule CD19 (Figure 3). Our finding about Pax5 expression and its DNA binding activity was in agreement with that reported by other investigators, who reported increased levels of Pax5 following CD154 stimulation.42 The conditional inactivation of Pax5 in late B lymphopoiesis led to a preferential loss of mature B cells in peripheral lymphoid organs and to the absence of recirculating B cells in the bone marrow,43 and this phenotype closely resembles that of BAFF-deficient mice.7
We found that BAFF simulation significantly increased the ability of BCR to phosphorylate CD19 (Figure 5). CD19 is tyrosine phosphorylated when BCR is ligated, and the basis for phosphorylation is the association of CD19 with tyrosine kinases (Lyn and Syk) and other components of the BCR complex consisting of CD22, Ig-α, and Ig-β.44 The tyrosine phosphorylation of CD19 at Tyr513 is required for amplifying BCR-mediated activation by coupling of PI3K, and BLNK links BCR-activated Syk kinase to phosphorylated CD19 Y513 to enhance the BCR signaling. We observed that the magnified capacity of BCR to phosphorylate CD19 after BAFF stimulation was more prominent in Ramos cells than in P32/ISH cells (Figure 5A). A probable explanation for this was presented by a recent study showing that TACI is a negatively regulatory receptor for BAFF signaling.45 Ramos cells displayed barely detectable TACI expression while P32/ISH expressed a relatively high level (Figure 4B), suggesting that TACI signaling might have a suppressive effect on phosphorylation of CD19. At present, the precise molecular mechanisms by which BAFF synergistically augments CD19 phosphorylation by BCR ligation are unknown. However, it is possible that BAFF signaling was integrated at the Lyn/Syk activation pathway, distinct from a signal for the Pax5 activation pathway leading to CD19 and BLNK up-regulation.46 It has been reported that CD40 engagement resulted in increased phosphorylation of Lyn in B cells.47 Alternatively, BAFF might inhibit phosphatase SHP1 activity, which suppresses Syk.44
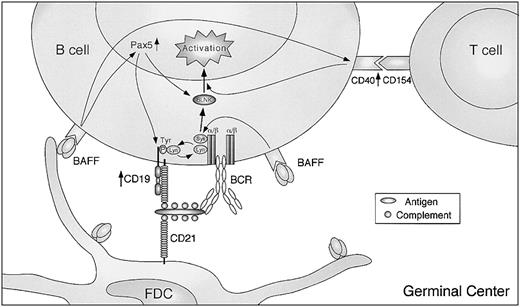
Finally, we demonstrated that when CD19 was coligated directly with BCR in the presence of IL-15, which mimics FDC–B-cell interactions, BAFF stimulation further enhanced B-cell proliferation and caused secretion of IgG (Figure 6B-C). Thus, BAFF can be involved in FDC–B-cell interactions in GCs to potentiate the primary response of B cells to Ags in concert with the B-cell coreceptor complex. Detectable levels of IgG production were not observed in the presence of BCR/CD19 coligation without BAFF stimulation (Figure 6C). Thus, it is possible that BAFF is required for plasma cell differentiation and IgG production in TI immune responses through FDC–B-cell interactions, because B cells proliferated in the same conditions. Furthermore, we demonstrated that after FDC–B-cell interaction induced by BAFF, B cells up-regulated CD40 expression and became susceptible to CD154, which is expressed on activated T cells. Notably, coligation of BCR/CD19 without BAFF failed to up-regulate CD40 (Figure 6A), in which substantial B-cell proliferation was observed. Taken together, these findings suggest that BAFF can play a crucial role in TI B-cell responses in FDC–B-cell interactions through the B-cell coreceptor complex and can transfer its B-cell specificity to TD immune responses (Figure 7). A small number of CD4+ T cells are reported to be present in GCs, so-called GC T cells or follicular B-helper T cells, which express CD154.48 Thus, CD40 engagement by GC T cells may elicit optimal B-cell responses after or simultaneously with FDC–B-cell interactions by BAFF. In addition, enhanced HLA-DR and ICAM-1 expression by BAFF (Figure 1A) would contribute to enhanced Ag presentation and adhesion to T cells. Consistent with this, the TD but not TI IgG response is impaired in CD154-deficient mice,49,50 whereas BAFF deficiency impairs both TD and TI IgG production,7 suggesting that BAFF sequentially links TI and TD B-cell responses.
Proposed scheme of B-cell activation with BAFF in GCs. GC B cells receive signals for activation by FDCs through the BCR and B-cell coreceptor complex. The B-cell responses are further enhanced by BAFF providing CD19 and probable BLNK up-regulation through Pax5 activation and the high ability of BCR to phosphorylate CD19, which in turn amplifies BCR signaling. Moreover, up-regulation of CD40 by BAFF renders the same B cells more susceptible to T-cell help by CD154. See “Discussion” for details.
Proposed scheme of B-cell activation with BAFF in GCs. GC B cells receive signals for activation by FDCs through the BCR and B-cell coreceptor complex. The B-cell responses are further enhanced by BAFF providing CD19 and probable BLNK up-regulation through Pax5 activation and the high ability of BCR to phosphorylate CD19, which in turn amplifies BCR signaling. Moreover, up-regulation of CD40 by BAFF renders the same B cells more susceptible to T-cell help by CD154. See “Discussion” for details.
Interestingly, the transcript for activation-induced cytidine deaminase (AID), which plays a critical role in both Ig somatic hypermutation and class switching in B cells,51 was found to be up-regulated by BAFF stimulation in addition to CD154 stimulation (Litinskiy et al39 and H. H. et al, unpublished data, December 2002). Thus, in addition to TI Ig class switching,39 it is likely that BAFF also produces high-affinity B-cell mutants, suggesting that BAFF is involved in affinity maturation of B cells even under TI conditions.
GCs are the primary sites for affinity maturation coupled with Ag-driven clonal selection (selection of B-cell mutants) and development of B-cell memory and plasma cells. The survival of B-cell mutants in GCs is dependent on their ability to compete for Ag on the surface of FDCs. Only high-affinity mutants are able to compete and retrieve Ags from FDCs and interact with GC helper T cells. In contrast, GC B cells with lower-affinity BCR fail to compete for Ags on FDCs and are consequently eliminated by apoptosis. Thus, the ability of the B-cell coreceptor complex to decrease the signaling thresholds for BCR stimulation in FDC–B-cell interactions plays a quite important role in B-cell selection. The development of SLE in humans is associated with abnormalities of B-cell functions, and an altered expression pattern of CD21 on B cells has been reported.52 Failure of CD21 expression was also found in a human SLE model mouse, MRL/lpr.53 Overexpression of CD19 is associated with autoimmunity in mice,54 and abnormalities in CD19 expression are observed in the human autoimmune disease scleroderma.55 The binding of C-coated self-Ags to CD21, despite low-affinity BCRs, may promote clonal expansion of self-reactive B cells and be involved in the selection of the repertoire of natural antibodies, suggesting a defect in peripheral tolerance of B cells in FDC–B-cell interactions in GCs, where BAFF can further help self-reactive B cells to escape from apoptosis. Indeed, BAFF overexpression increased IgG, IgA, and IgE production and led to an SLE-like syndrome with kidney deposition of IgG and IgA.6,56 Thus, it is most likely that BAFF regulates peripheral tolerance through the B-cell coreceptor complex. This new role for BAFF in FDC–B-cell interactions in concert with the B-cell coreceptor complex suggests that BAFF-induced accumulation of self-reactive B cells might be the result of direct effects on their survival and selection rather than simple expansion of the B-cell compartment, thereby increasing the number of responders. BAFF may therefore play an important role in regulating immunity and tolerance by controlling the development of B cells.
Prepublished online as Blood First Edition Paper, November 20, 2003; DOI 10.1182/blood-2003-08-2694.
Supported in part by grants from the NOVARTIS Foundation (Japan) for the Promotion of Science and from the Promotion and Mutual Aid Corporation for Private Schools of Japan.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.
We thank Dr Hajime Karasuyama for the pBCMGSneo expression vector, Dr Akira Niwa for helpful discussion, and Ms Yoshie Nitta for excellent secretarial assistance. We also thank the Laboratory Animal Research Center and Laboratory of Analytical Instruments, Institute for Medical Science, Dokkyo University School of Medicine, for the use of their facilities and thank the Japanese Red Cross Tochigi Blood Center for leukopaks. We also thank Dr F. G. Issa for critical reading of the manuscript.

![Figure 1. Functional characterization of BAFF transfectant. (A) Human peripheral blood B cells (1 × 105 per well) were cultured with or without mitomycin-treated transfected cells (2 × 104 per well) in the presence or absence of SAC (0.0002% [wt/vol]) and IL-2 (50 U/mL). After treatment for 48 hours, the expression level of CD40, ICAM-1, and HLA-DR was analyzed by FACS analysis. (B) B cells (1 × 105 per well) were cocultured in triplicate in 96-well plates with mitomycin-treated transfected cells (2 × 104 per well) in the presence or absence of SAC (0.0002% [wt/vol] or 0.0004% [wt/vol]) and IL-2 (50 U/mL), and [3H]thymidine (0.5 μCi [18.5 kBq] per well). Incorporation of B cells was measured during the last 18 hours of a total of 72 hours. Data are mean ± SD of triplicate wells and are representative of 3 different experiments.](https://ash.silverchair-cdn.com/ash/content_public/journal/blood/103/6/10.1182_blood-2003-08-2694/6/m_zh80060458260001.jpeg?Expires=1769083395&Signature=Sdgj7qOUPFa4uOM7SwDAn2984KvouOpC2P-hcRWULlXnKxA1I7oEYXkmUBVmI~uczNTMoiFIRPfeggYIiYGKqyziod36q3N3MNxlQvj~mAA2eCErXunl3-s~OQXgUi9LvCyn4QbacWwS0wQgeCEIqKvneNBSiSnZXl46xhisg67zfJKiWpihnuETazgxgBUZWe5RkAFAK01eAwbYQzMJQkKKc~1x53p7DJ3DhiqIOji8n64YO6-mNiJNYjoWoCskGt5iUiPknHxLYXvY5oT-2XP5x0vfGAFa1OhuF8GJzvytg~YT4LIXT7CETMBHOo57K8JJOFiUZ1bh8MULtZ4QNA__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)
![Figure 3. Up-regulation of Pax5/BSAP and CD19 by BAFF. Human peripheral blood B cells were cultured with or without mitomycin-treated transfectants at a ratio of 10:2 in the presence or absence of SAC (0.0002% [wt/vol]) and IL-2 (50 U/mL). (A) After the treatment for 48 hours, the expression level of CD19, CD20, and CD21 was evaluated by FACS analysis. After treatment for 48 hours in the above conditions (A), cell lysates and nuclear extracts were prepared for immunoblot analysis using anti-Pax5 Ab (B) and EMSA using oligonucleotides containing Pax5 binding site (C), respectively. Lane 1, Pax5 oligonucleotide alone; lane 2, Pax5 mutant oligonucleotide alone; lane 3, B cell alone plus Pax5 oligonucleotides; lane 4, SAC/IL-2–stimulated B cells plus mock transfectants plus Pax5 oligonucleotides; lane 5, SAC/IL-2–stimulated B cells plus CD154 transfectants plus Pax5 oligonucleotides; lane 6, SAC/IL-2–stimulated B cells plus BAFF transfectants plus Pax5 oligonucleotides; lane 7, SAC/IL-2–stimulated B cells plus BAFF transfectants plus Pax5 mutant oligonucleotides; lane 8, SAC/IL-2–stimulated B cells plus BAFF transfectants plus Pax5 oligonucleotides plus competitor; lane 9, SAC/IL-2–stimulated B cells plus BAFF transfectants plus Pax5 oligonucleotides plus anti-Pax5 Ab. NS indicates nonspecific band. Data shown are representative of 3 different experiments.](https://ash.silverchair-cdn.com/ash/content_public/journal/blood/103/6/10.1182_blood-2003-08-2694/6/m_zh80060458260003.jpeg?Expires=1769083396&Signature=TZfzPFKb41ZaAeaakf-Zf-y8-yS9xoKKsOkMn9s7qkt3h1t3TvzFVDyT7OXtc6a5yLUz36WwWaHx8FZ10qX70ViWN1S4Q3RcqN-s4BrAo7LyBAp-OqXpI1sy-AY7PVOD-IJFvNiEyafUquQdF3j7je4zvKv2XQKfWYCNgUbqEINgIuj72~3xmxtMllDzZy82ZmMmozcl8qcsZ09m4c19WvWMwRUzw9C2fyJuBmL0ohXvP3lbQI8V5fbVn-iINrEjOSfB4LWxFhlQx1Qp9A0GY8ijyRUnXG4fSzbIarIAHlsfYH0nxuEqRJfFv660ejVqk0KkdsT5i8Hw5MLSH9eF6Q__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)
![Figure 6. Effects of BAFF on B-cell responses with coligation of BCR and CD19. Human peripheral blood B cells (1 × 105) pretreated with anti-BCR mAbs (anti-Igκ and anti-Igλ; 1 μg/mL), anti-CD19 mAb (0.5 μg/mL), and/or control mAb (1 μg/mL) were cultured with or without sBAFF (0.1 μg/mL) in triplicate in 96-well plates precoated with mitomycin-treated DT70 cells (2 × 104 per well) in the presence or absence of IL-15 (50 ng/mL) together with or without mitomycin-treated CD154 transfectant (1 × 104 per well). (A) After treatment for 48 hours, the expression level of CD40 was analyzed by FACS analysis. (B) [3H]thymidine (0.5 μCi [18.5 kBq] per well) incorporation of B cells was measured during the last 18 hours of a total of 72 hours. (C) After 10 days, IgG concentration in the supernatant was measured by ELISA. For the blocking assay, TACI-Ig fusion protein was used at 0.4 μg/mL. The values are reported as the mean ± SD of triplicate wells, and data shown are representative of 3 different experiments; *P < .05; **P < .01.](https://ash.silverchair-cdn.com/ash/content_public/journal/blood/103/6/10.1182_blood-2003-08-2694/6/m_zh80060458260006.jpeg?Expires=1769083396&Signature=gG9kG69JpvlIaPnERniy9fW68piV~wCEHAXuLXDOaoICSFcELtjHa6uaeMJWa9cFW67UMYEmZGmF1Xwwt6bb75Uk3GeTPDClFZvX10VlALinRx-OFfYsD37u~ZPIEbjfs89IWbFAuvACDl~8FW0dUGHKSjvltaFuuBft9uK05yeOUwcyS2x2ppLgkCeevcgwprVNG9RAv8cpTijKk8RfuH5veNj3RZUOx0n-jUO708esdHkQOs3bPo4AbntmAokKxcZmCBMe95asb-tDzpcRSwNKLNmkHnT0TWexfPIBgjMy~1NqJ6Kmw9zU3LWITzXHqIoawW5q8WlaIU2vxulp6g__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)
This feature is available to Subscribers Only
Sign In or Create an Account Close Modal