Comment on Coriu et al, page 829
The molecular basis of nonsecretory myeloma in one patient is shown to be due to a crippling frameshift mutation in the kappa constant region.
Most patients (∼ 80%) with multiple myeloma have a monoclonal immunoglobulin (Ig) detectable in the serum, usually IgG or IgA. About 18% have only monoclonal light chains detectable in the urine (light-chain multiple myelomas [LCMMs]). Finally, about 2% do not secrete any immunoglobulin (nonsecretory multiple myelomas [NSMMs]). Recent analyses of LCMMs have shown that the tumors lack a functional IgH rearrangement, providing a molecular basis for this condition.1 There has not been a comprehensive analysis of NSMMs, and previous reports have emphasized acquired mutations in the immunoglobulin light chain variable genes that lead to a block in secretion.2-4 Thus from the information available there appears to be a fundamental difference in these 2 conditions, which otherwise share many features (eg, very high incidence of t(11;14)). On the one hand, LCMM lacks a functional IgH DNA rearrangement, and hence there is no RNA or protein. On the other hand, NSMM has an IgL DNA rearrangement, RNA, and protein, but suffers from crippling mutations.
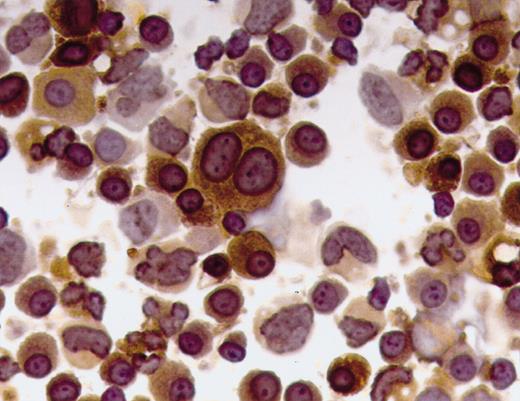
In this issue, the molecular basis of nonsecretory myeloma in one patient is shown to be due to a crippling frameshift mutation in the κ constant region. Coriu and colleagues show that a 2-base deletion in codon 187 resulted in the loss of the normal stop codon. There was a loss of 2 cysteine molecules that are necessary for intrachain and interchain disulfide bonds. The authors postulate that the absence of C194 disrupted the 3-dimensional features of the molecule and prevented binding with the cysteine in the first heavy-chain C-domain. The misfolded κ chains were retained within the plasma cell. They show that this results in an abnormal-sized protein that reacts with some but not all anti-κ antibodies. In contrast to previously described mutations of the variable region, this represents a novel mechanism for nonsecretory myeloma.FIG1
Immunocytochemical analyses of plasma cells from the patient with NSM and chemical characterization of the nonsecreted κ light chains. See the complete figure in the article beginning on page 829.
Immunocytochemical analyses of plasma cells from the patient with NSM and chemical characterization of the nonsecreted κ light chains. See the complete figure in the article beginning on page 829.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal