Abstract
We report the outcome of 229 patients who received an allograft for myeloma with reduced-intensity conditioning (RIC) regimens from 33 centers within the European Group for Blood and Marrow Transplantation (EBMT). The median age was 52 years and 64% were male. Conditioning regimens were heterogeneous, but most were fludarabine based and T cell depleted with antithymocyte globulin or alemtuzumab. Transplantation-related mortality (TRM) at 1 year was 22%. The 3-year overall survival (OS) and progression-free survival (PFS) were 41% and 21%, respectively. Adverse OS was associated with chemoresistant disease (relative risk [RR], 2.9), more than 1 prior transplantation (RR, 2.0), and male patients with female donors (RR, 1.45). Adverse PFS was associated with chemoresistance (RR, 2.4) and alemtuzumab (RR, 1.8). TRM was increased with female-to-male donation (RR, 2.5) and transplantation more than 1 year from diagnosis (RR, 2.3). Grades II to IV acute graft-versus-host disease (aGvHD) occurred in 31%. Chronic GvHD was associated with better OS and PFS and were 84% and 46% for limited, 58% and 30% for extensive, and 29% and 12% in its absence suggesting that a graft-versus-myeloma effect is important. While RIC is feasible, heavily pretreated patients and patients with progressive disease do not benefit.
Introduction
Allogeneic hematopoietic stem cell transplantation is a potentially curative procedure for myeloma. It is associated with lower relapse rates compared with autologous transplantation, and some patients become long-term disease-free survivors.1-4 The use of allogeneic transplantation has, however, remained restricted to a subpopulation of patients, as most are older than the conventionally accepted age limits for transplantation, lack a suitable donor, or both. In addition the high transplantation-related mortality (TRM) of up to 44% has deterred both patients and their physicians.4,5 More recently a reduction in TRM from 38% to 21% has been noted for patients who received a transplant between 1983 to 1993 and 1994 to 1998,6 respectively. This improvement can be attributed at least in part to better patient selection and in part to improvements in supportive care. However, with the upper age of these patients being older than 60 years it is clear that conventional allogeneic transplantation, even with the best results, remains applicable only to a minority of patients with myeloma.
The introduction of reduced-intensity conditioning for transplantation (RICT) regimens, with a reduction in immediate transplant-related mortality7-9 has renewed interest in allogeneic transplantation for myeloma. In the context of RICT regimens, the use of in vivo T-cell depletion with antithymocyte globulin (ATG) or alemtuzumab (Campath-1H) has been associated with a very low incidence of graft-versus-host disease10 and consequently RICT regimens have become the procedure of choice for older patients with hematologic malignancies.11 Studies reporting myeloma-specific outcomes from RICT regimens are currently limited, and their efficacy in this disease remains uncertain. The outcome of a poor-risk group of patients consisting of 25 siblings and 6 unrelated donor transplants conditioned with melphalan 100 mg/m2 with or without fludarabine and total body irradiation (TBI) had a median overall survival of only 15 months, although the day 100 TRM was low at 10%.12 More recent studies have reported better outcomes by using either fludarabine and melphalan as the conditioning chemotherapy or tandem autograft and allograft approaches with event-free survivals at 2 years of 33% and 56%, respectively.13-15 Similar results have been observed by the Seattle group16 by using fludarabine and low-dose TBI. Overall, the published experience in myeloma remains small. The purpose of the present study was to establish evidence of efficacy and to identify clinically important prognostic factors.
Patients, materials, and methods
This study was conducted on behalf of the Chronic Leukaemia Working Party (CLWP) of the European Group for Blood and Marrow Transplantation (EBMT) and was performed in conjunction with a study of reduced-intensity conditioning transplantation in chronic myeloid leukemia. All EBMT centers report a minimal essential data set into a central database. All centers that had reported more than 5 RICT regimens for myeloma, chronic myelogenous leukemia, or both were asked for additional data. This analysis is restricted to those patients with myeloma. Informed consent was obtained locally according to the regulations applicable at the time of transplantation. Since January 1, 2003, the EBMT has required centers to confirm that written informed consent has been obtained prior to data acceptance. The definition of RICT was determined by contributing centers and was based on the current EBMT guidance. Patients who received regimens with equivalent to or lower intensity than melphalan 140 mg/m2 were included in the study. Patients who had received TBI were included, provided the delivered dose was not greater than 6 Gy. The minimum data set required for inclusion of a patient in the study was defined at the outset and included diagnosis, date of diagnosis, date of transplantation, age at transplantation, number of prior transplantations, disease status at transplantation, donor type, conditioning regimen, grade of acute graft-versus-host disease (aGvHD) remission status after transplantation, disease status at follow-up, date of progression or death, date of last follow-up, and cause of death. These data were available for 100% of the patients. The following parameters were available for the majority of patients and were entered into the analysis: disease stage at diagnosis (200 of 229 patients, 87%), isotype of myeloma (225 of 229, 98%), donor sex (226 of 229, 99%), nature of GvHD prophylaxis (207 of 229, 90%), and time to engraftment (217 of 229, 95%). Chimerism studies were performed in the most part in peripheral blood by molecular methodologies (85%) and by cytogenetics (14%). One patient was reported on the basis of ABO group.
Definitions
The diagnosis of myeloma was based on local review. Chemosensitive disease was defined as all patients who were in complete remission (CR) or partial response (PR) following the last course of chemotherapy prior to transplantation. Chemoresistance was defined as patients who had had no or minimal response or disease progression. Response, relapse, and disease progression were defined according to published criteria.17 Response was documented on or after day 100 after transplantation. Survival was measured in months and defined as the time from the date of transplantation until date of death or last follow-up. Progression-free survival was defined as the time from transplantation until date of progression or death from any cause or last follow-up. Transplantation-related mortality was defined as death due to any cause other than disease progression or relapse occurring at any time after transplantation. Landmark analyses were performed for chronic graft-versus-host disease (cGvHD) whereby only patients surviving more than 100 days were included.
Statistical analysis
Probabilities of overall survival (OS) and progression-free survival (PFS) were calculated using the method of Kaplan-Meier, while probabilities of transplantation-related mortality and relapse were estimated using the cumulative incidence procedure. Univariate comparisons were made using the log-rank test, and variables found to be significant at the level of P greater than .2 were entered into a proportional hazards regression analysis using a backward-stepping procedure. Comparisons between groups were made using the chi-squared test for categorical data and the Mann-Whitney test for continuous data. All quoted P values are from 2-sided tests, and values of less than .05 were considered significant. Quoted confidence intervals (CIs) refer to 95% boundaries.
Results
Data from 268 patients who received a transplant between June 1998 and February 2003 were collected from 36 centers within the EBMT group. The minimum essential data set was available for 229 patients from 33 centers, and these patients form the basis for this study.
Patient characteristics
The median age of the cohort was 52 years (range, 32-66 years) and 147 were male. The median time from diagnosis to transplantation was 1.6 years (2.0 months to 11 years). Fifty-nine percent, 21%, and 0.5% were immunoglobulin G (IgG), IgA, and IgM, respectively. Light chain myeloma accounted for 15%, nonsecretory for 3%, and isotype was unknown in 1.5% of patients. At the time of transplantation 63% were in remission (10% complete). The majority had received at least 1 prior autograft, and 10% had received 2 or more. The patient characteristics are shown in Table 1.
Patient characteristics
. | n . |
|---|---|
| Median age at transplantation, y (range) | 52 (32-66) |
| M/F ratio | 1.8:1 |
| Immunoglobulin class (%) | |
| IgG | 134 (59) |
| IgA | 48 (21) |
| IgM | 1 (0.5) |
| Light chain | 35 (15) |
| Nonsecretory | 7 (3) |
| Unknown | 4 (1.5) |
| κ:λ | 2:1 |
| Disease duration, median, y (range) | 1.6 (0.2-11) |
| Disease duration more than 1 y to transplantation (%) | 122 (53) |
| Recipient CMV IgG+ (%) | 165 (74) |
| Status at transplantation (%) | |
| CR | 23 (10) |
| PR | 122 (53) |
| First CR or PR* | 61 (27) |
| Stable | 22 (10) |
| Progression | 62 (27) |
| Number of prior transplantations (%) | |
| 0 | 603 (26) |
| 1 | 146 (64) |
| At least 2 | 23 (10) |
| Donor match (%) | |
| Matched sibling | 178 (78) |
| Syngeneic | 2 (1) |
| Matched related | 5 (2) |
| Mismatched related | 7 (3) |
| Matched unrelated | 32 (14) |
| Mismatched unrelated | 5 (2) |
| Donor sex match (%) | |
| Female to male | 70 (31) |
| Male to female | 42 (19) |
| Other | 114 (50) |
| Cell source (%) | |
| PBSC | 183 (80) |
| BM | 46 (20) |
. | n . |
|---|---|
| Median age at transplantation, y (range) | 52 (32-66) |
| M/F ratio | 1.8:1 |
| Immunoglobulin class (%) | |
| IgG | 134 (59) |
| IgA | 48 (21) |
| IgM | 1 (0.5) |
| Light chain | 35 (15) |
| Nonsecretory | 7 (3) |
| Unknown | 4 (1.5) |
| κ:λ | 2:1 |
| Disease duration, median, y (range) | 1.6 (0.2-11) |
| Disease duration more than 1 y to transplantation (%) | 122 (53) |
| Recipient CMV IgG+ (%) | 165 (74) |
| Status at transplantation (%) | |
| CR | 23 (10) |
| PR | 122 (53) |
| First CR or PR* | 61 (27) |
| Stable | 22 (10) |
| Progression | 62 (27) |
| Number of prior transplantations (%) | |
| 0 | 603 (26) |
| 1 | 146 (64) |
| At least 2 | 23 (10) |
| Donor match (%) | |
| Matched sibling | 178 (78) |
| Syngeneic | 2 (1) |
| Matched related | 5 (2) |
| Mismatched related | 7 (3) |
| Matched unrelated | 32 (14) |
| Mismatched unrelated | 5 (2) |
| Donor sex match (%) | |
| Female to male | 70 (31) |
| Male to female | 42 (19) |
| Other | 114 (50) |
| Cell source (%) | |
| PBSC | 183 (80) |
| BM | 46 (20) |
CMV indicates cytomegalovirus; PBSC, peripheral blood stem cell; BM, bone marrow.
Subset of CR and PR
Conditioning and transplantation details
A number of different reduced-intensity conditioning regimens were used and are listed in Table 2. The majority were fludarabine based (96%) with the addition of either busulfan or melphalan. Seventy-nine patients received ATG as in vivo T-cell depletion, and 55 patients received alemtuzumab, most frequently in combination with fludarabine and melphalan. Peripheral blood was used as the source of cells in 183 patients and bone marrow in 46. Median cell doses for peripheral blood were 5.27 × 106 CD34/kg and for marrow 2.92 × 108 mononuclear cells/kg. GvHD prophylaxis consisted of cyclosporin alone (56%), cyclosporin and methotrexate (30%), cyclosporin and steroid (2%), and methotrexate alone (3%) and was unknown in 9%.
Conditioning regimens with or without antibody (Ab:ATG or alemtuzumab)
Drug Combination . | No Ab . | ATG . | Alemtuzumab . | Total . |
|---|---|---|---|---|
| FD + TBI | 34 | 0 | 1 | 35 |
| FD + busulfan | 5 | 30 | 2 | 37 |
| FD + busulfan + other | 0 | 6 | 1 | 7 |
| FD + melphalan | 34 | 7 | 44 | 85 |
| FD + melphalan + TBI | 4 | 0 | 1 | 5 |
| FD + melphalan + cyclo | 2 | 13 | 4 | 19 |
| FD + cyclo | 12 | 3 | 1 | 16 |
| FD + cyclo + TBI | 0 | 11 | 0 | 11 |
| Other | 4 | 9 | 1 | 13 |
Drug Combination . | No Ab . | ATG . | Alemtuzumab . | Total . |
|---|---|---|---|---|
| FD + TBI | 34 | 0 | 1 | 35 |
| FD + busulfan | 5 | 30 | 2 | 37 |
| FD + busulfan + other | 0 | 6 | 1 | 7 |
| FD + melphalan | 34 | 7 | 44 | 85 |
| FD + melphalan + TBI | 4 | 0 | 1 | 5 |
| FD + melphalan + cyclo | 2 | 13 | 4 | 19 |
| FD + cyclo | 12 | 3 | 1 | 16 |
| FD + cyclo + TBI | 0 | 11 | 0 | 11 |
| Other | 4 | 9 | 1 | 13 |
FD indicates fludarabine; Cyclo, cyclophosphamide; TBI, total body irradiation.
Engraftment and chimerism
Engraftment occurred in 94% regardless of the source of stem cells with an additional 1% subsequently losing the graft. The median times to neutrophil (> 0.5 × 109/L) and platelet (> 50 × 109/L) recovery were 14 days (range, 0-393 days) and 14 days (range, 0-374 days), respectively. There was a significant difference between patients receiving peripheral blood stem cells (PBSCs) and marrow in the time to neutrophil recovery (13 versus 16 days, P = .001) and to platelet recovery (13 versus 22 days, P < .001). All patients receiving marrow became neutropenic and only one did not become thrombocytopenic, whereas 10% of those receiving PBSCs had a neutrophil count more than 0.5 × 109/L at all times, and 13% did not become thrombocytopenic (> 50 × 109/L). Chimerism data were available on 190 patients. At best, 81% achieved full donor chimerism and 86% were greater than 95% donor. On subsequent analysis this had fallen to 74% for full donor and to 80% for greater than 95% donor.
Graft versus host disease
Acute GvHD occurred in 100 patients (44%) with 29 (13%) experiencing grade I and 72 (31%) grades II to IV disease. The incidence of aGvHD was not significantly altered by the use of ATG or alemtuzumab in the conditioning, the use of peripheral blood versus marrow, or GvHD prophylaxis. Grades III to IV aGvHD was associated with inferior survival (43% versus 21% at 3 years, P = .001) but did not alter the incidence of relapse. Of the 174 patients who were evaluable for cGvHD, 44 (25%) developed limited cGvHD and 43 (25%) developed extensive cGvHD. The use of T-cell–depleting antibodies significantly reduced the incidence of cGvHD (71% versus 39%, P < .001), and this reduction was greater in patients receiving alemtuzumab than ATG (32% versus 41%). The use of peripheral blood was associated with an increased incidence of cGvHD (55% versus 31%, P = .01) unlike the donor-recipient sex combination which was not significantly linked with GvHD.
Transplantation-related mortality
Sixty patients died of procedural-related complications, of whom 26 died before day 100. The causes of death were GvHD (n = 11), infection (n = 39), pneumonitis (n = 4), hemorrhage (n = 4), cardiac toxicity (n = 2), graft failure (n = 2), lymphoproliferative disorder (n = 1), and other malignancies (n = 2). In several patients the cause of death was multifactorial. In patients receiving alemtuzumab 16 of 31 deaths were secondary to infection. The treatment-related mortalities at day 100, 1 year, and 2 years were 10%, 22%, and 26%, respectively. Factors on univariate analysis associated with a higher TRM were female donors to male recipients (37% versus 16% 1-year TRM, P = .001; Table 3), transplantation more than 1 year from diagnosis (30% versus 15%, P = .004), and the use of an unrelated donor (40% versus 20%, P = .03). Of these, a female-to-male donation and disease duration more than 1 year retained significance on multivariate analysis (Table 4). Overall the TRM is less than 20% in patients with none or one of these risk factors.
Univariate analysis for overall survival, progression-free survival, and transplantation-related mortality
. | . | Overall survival . | . | PFS . | . | TRM . | . | |||
|---|---|---|---|---|---|---|---|---|---|---|
. | n . | 3-y survival . | P . | 3-y survival . | P . | 1-y TRM . | P . | |||
| Age, y | .44 | .23 | .69 | |||||||
| Younger than 50 | 88 | 43.5 | 17.9 | 22.5 | ||||||
| 50-60 | 116 | 34.2 | 19.7 | 22.7 | ||||||
| Older than 60 | 25 | 51.8 | 43.4 | 25.0 | ||||||
| Patient sex | .12 | .23 | .10 | |||||||
| Male | 147 | 37.1 | 19.2 | 26.9 | ||||||
| Female | 82 | 46.1 | 26.2 | 15.6 | ||||||
| Donor-recipient sex match | .03 | .235 | .001 | |||||||
| Other | 156 | 45.1 | 23.2 | 16.1 | ||||||
| F-M | 70 | 31.7 | 33.0 | 37.3 | ||||||
| Stage | .85 | .87 | ||||||||
| I | 16 | 45.1 | 31.3 | 26.3 | .65 | |||||
| II | 48 | 40.7 | 31.3 | 18.8 | ||||||
| III | 136 | 41.1 | 19.4 | 23.1 | ||||||
| Renal impairment | .38 | .71 | .33 | |||||||
| A | 174 | 41.6 | 19.6 | 21.0 | ||||||
| B | 24 | 34.3 | 22.2 | 35.5 | ||||||
| First vs other remission | .001 | .001 | ||||||||
| 1st remission | 61 | 67.2 | 34.0 | 15.3 | .08 | |||||
| More than 1st | 84 | 35.0 | 22.4 | 22.0 | ||||||
| SD/PD | 84 | 27.6 | 10.3 | 31.0 | ||||||
| Remission status | .006 | .001 | ||||||||
| CR | 23 | 49.0 | 35.8 | 19.0 | .21 | |||||
| PR | 122 | 49.0 | 26.8 | 19.2 | ||||||
| SD | 22 | 32.8 | 22.2 | 14.8 | ||||||
| PD | 62 | 27.9 | 10.5 | 35.5 | ||||||
| Disease duration | .002 | .04 | .001 | |||||||
| Less than 1 y | 107 | 49.9 | 26.5 | 15.1 | ||||||
| Longer than 1 y | 122 | 32.8 | 16.9 | 30.1 | ||||||
| Recipient CMV IgG | .16 | .095 | .64 | |||||||
| Negative | 58 | 29.3 | 26.5 | 18.1 | ||||||
| Positive | 165 | 46.5 | 16.9 | 24.6 | ||||||
| T-cell depletion | .06 | .03 | .34 | |||||||
| No | 83 | 53.5 | 27.4 | 19.1 | ||||||
| Yes | 145 | 34.2 | 18.2 | 35.3 | ||||||
| Use of specific antibody | .17 | .008 | .50 | |||||||
| No Ab | 94 | 52.8 | 27.3 | 19.1 | ||||||
| ATG | 79 | 37.1 | 23.3 | 24.2 | ||||||
| Alemtuzumab | 55 | 26.0 | 8.1 | 29.0 | ||||||
| Cell source | .28 | .48 | .84 | |||||||
| PBSC | 183 | 42.1 | 22.9 | 24.2 | ||||||
| BM | 46 | 35.8 | 16.0 | 22.8 | ||||||
| Prior transplantation | .003 | .08 | .13 | |||||||
| 0 | 60 | 45.8 | 18.4 | 25.2 | ||||||
| 1 | 146 | 40.8 | 24.0 | 20.1 | ||||||
| More than 1 | 23 | 22.8 | 23.9 | 35.1 | ||||||
| Donor match | .002 | .04 | .034 | |||||||
| Related | 192 | 45.3 | 21.8 | 19.5 | ||||||
| VUD | 37 | 17.3 | 21.2 | 40.0 | ||||||
| Conditioning | ||||||||||
| Fludarabine | .3 | .23 | .19 | |||||||
| No | 11 | 58.2 | 36.4 | 9.1 | ||||||
| Yes | 218 | 39.8 | 20.3 | 23.9 | ||||||
| Busulfan | .01 | .37 | .23 | |||||||
| No | 184 | 47.0 | 23.7 | 22.1 | ||||||
| Yes | 45 | 18.0 | 11.2 | 27.2 | ||||||
| Melphalan | .14 | .74 | .98 | |||||||
| No | 117 | 35.3 | 19.3 | 23.0 | ||||||
| Yes | 112 | 46.0 | 22.8 | 23.1 | ||||||
| Cyclophosphamide | .8 | .35 | .74 | |||||||
| No | 176 | 38.1 | 20.7 | 11.6 | ||||||
| Yes | 53 | 45.8 | 22.5 | 28.0 | ||||||
| FD + TBI | .2 | .56 | .15 | |||||||
| No | 194 | 38.7 | 21.1 | 24.4 | ||||||
| Yes | 35 | 63.6 | 28.9 | 13.7 | ||||||
| FD + Bu + ATG | .02 | .267 | .41 | |||||||
| No | 199 | 46.2 | 23.0 | 22.7 | ||||||
| Yes | 30 | 14.8 | 13.2 | 24.9 | ||||||
| GvHD prophylaxis | .8 | .05 | .7 | |||||||
| CSA | 127 | 41.4 | 14.3 | 23.5 | ||||||
| CSA + MTX | 69 | 42.1 | 31.9 | 17.9 | ||||||
. | . | Overall survival . | . | PFS . | . | TRM . | . | |||
|---|---|---|---|---|---|---|---|---|---|---|
. | n . | 3-y survival . | P . | 3-y survival . | P . | 1-y TRM . | P . | |||
| Age, y | .44 | .23 | .69 | |||||||
| Younger than 50 | 88 | 43.5 | 17.9 | 22.5 | ||||||
| 50-60 | 116 | 34.2 | 19.7 | 22.7 | ||||||
| Older than 60 | 25 | 51.8 | 43.4 | 25.0 | ||||||
| Patient sex | .12 | .23 | .10 | |||||||
| Male | 147 | 37.1 | 19.2 | 26.9 | ||||||
| Female | 82 | 46.1 | 26.2 | 15.6 | ||||||
| Donor-recipient sex match | .03 | .235 | .001 | |||||||
| Other | 156 | 45.1 | 23.2 | 16.1 | ||||||
| F-M | 70 | 31.7 | 33.0 | 37.3 | ||||||
| Stage | .85 | .87 | ||||||||
| I | 16 | 45.1 | 31.3 | 26.3 | .65 | |||||
| II | 48 | 40.7 | 31.3 | 18.8 | ||||||
| III | 136 | 41.1 | 19.4 | 23.1 | ||||||
| Renal impairment | .38 | .71 | .33 | |||||||
| A | 174 | 41.6 | 19.6 | 21.0 | ||||||
| B | 24 | 34.3 | 22.2 | 35.5 | ||||||
| First vs other remission | .001 | .001 | ||||||||
| 1st remission | 61 | 67.2 | 34.0 | 15.3 | .08 | |||||
| More than 1st | 84 | 35.0 | 22.4 | 22.0 | ||||||
| SD/PD | 84 | 27.6 | 10.3 | 31.0 | ||||||
| Remission status | .006 | .001 | ||||||||
| CR | 23 | 49.0 | 35.8 | 19.0 | .21 | |||||
| PR | 122 | 49.0 | 26.8 | 19.2 | ||||||
| SD | 22 | 32.8 | 22.2 | 14.8 | ||||||
| PD | 62 | 27.9 | 10.5 | 35.5 | ||||||
| Disease duration | .002 | .04 | .001 | |||||||
| Less than 1 y | 107 | 49.9 | 26.5 | 15.1 | ||||||
| Longer than 1 y | 122 | 32.8 | 16.9 | 30.1 | ||||||
| Recipient CMV IgG | .16 | .095 | .64 | |||||||
| Negative | 58 | 29.3 | 26.5 | 18.1 | ||||||
| Positive | 165 | 46.5 | 16.9 | 24.6 | ||||||
| T-cell depletion | .06 | .03 | .34 | |||||||
| No | 83 | 53.5 | 27.4 | 19.1 | ||||||
| Yes | 145 | 34.2 | 18.2 | 35.3 | ||||||
| Use of specific antibody | .17 | .008 | .50 | |||||||
| No Ab | 94 | 52.8 | 27.3 | 19.1 | ||||||
| ATG | 79 | 37.1 | 23.3 | 24.2 | ||||||
| Alemtuzumab | 55 | 26.0 | 8.1 | 29.0 | ||||||
| Cell source | .28 | .48 | .84 | |||||||
| PBSC | 183 | 42.1 | 22.9 | 24.2 | ||||||
| BM | 46 | 35.8 | 16.0 | 22.8 | ||||||
| Prior transplantation | .003 | .08 | .13 | |||||||
| 0 | 60 | 45.8 | 18.4 | 25.2 | ||||||
| 1 | 146 | 40.8 | 24.0 | 20.1 | ||||||
| More than 1 | 23 | 22.8 | 23.9 | 35.1 | ||||||
| Donor match | .002 | .04 | .034 | |||||||
| Related | 192 | 45.3 | 21.8 | 19.5 | ||||||
| VUD | 37 | 17.3 | 21.2 | 40.0 | ||||||
| Conditioning | ||||||||||
| Fludarabine | .3 | .23 | .19 | |||||||
| No | 11 | 58.2 | 36.4 | 9.1 | ||||||
| Yes | 218 | 39.8 | 20.3 | 23.9 | ||||||
| Busulfan | .01 | .37 | .23 | |||||||
| No | 184 | 47.0 | 23.7 | 22.1 | ||||||
| Yes | 45 | 18.0 | 11.2 | 27.2 | ||||||
| Melphalan | .14 | .74 | .98 | |||||||
| No | 117 | 35.3 | 19.3 | 23.0 | ||||||
| Yes | 112 | 46.0 | 22.8 | 23.1 | ||||||
| Cyclophosphamide | .8 | .35 | .74 | |||||||
| No | 176 | 38.1 | 20.7 | 11.6 | ||||||
| Yes | 53 | 45.8 | 22.5 | 28.0 | ||||||
| FD + TBI | .2 | .56 | .15 | |||||||
| No | 194 | 38.7 | 21.1 | 24.4 | ||||||
| Yes | 35 | 63.6 | 28.9 | 13.7 | ||||||
| FD + Bu + ATG | .02 | .267 | .41 | |||||||
| No | 199 | 46.2 | 23.0 | 22.7 | ||||||
| Yes | 30 | 14.8 | 13.2 | 24.9 | ||||||
| GvHD prophylaxis | .8 | .05 | .7 | |||||||
| CSA | 127 | 41.4 | 14.3 | 23.5 | ||||||
| CSA + MTX | 69 | 42.1 | 31.9 | 17.9 | ||||||
SD indicates stable disease; PD, progressive disease; VUD, volunteer unrelated donor; Bu busulfan; CSA, cyclosporin; MTX, methotrexate
Multivariate analysis for overall survival, progression-free survival, and transplantation-related mortality
Outcome and variables . | Relative risk . | CI . | P . |
|---|---|---|---|
| Overall survival | |||
| Pretransplantation status | |||
| 1st remission | 1.00 | — | — |
| More than 1st remission | 1.93 | 1.10-3.37 | .02 |
| SD/PD | 2.95 | 1.72-5.06 | .001 |
| Previous transplantation | |||
| 0 or 1 | 1.00 | — | — |
| 2 or more | 2.05 | 1.18-3.59 | .01 |
| Donor-recipient sex combination | |||
| All others | 1.00 | — | — |
| Female donor-male recipient | 1.45 | 0.99-2.13 | .06 |
| Progression-free survival | |||
| Pretransplantation status | |||
| 1st remission | 1.00 | — | — |
| More than 1st remission | 1.45 | 0.93-2.25 | .1 |
| SD/PD | 2.42 | 1.58-3.70 | .001 |
| Alemtuzumab conditioning | |||
| No | 1.00 | — | — |
| Yes | 1.83 | 1.29-2.61 | .001 |
| TRM | |||
| Donor-recipient sex combination | |||
| Others | 1.00 | — | — |
| Female donor-male recipient | 2.45 | 1.45-4.15 | .001 |
| Disease duration | |||
| Less than 1 y | 1.00 | — | — |
| At least 1 y | 2.25 | 1.27-3.98 | .005 |
Outcome and variables . | Relative risk . | CI . | P . |
|---|---|---|---|
| Overall survival | |||
| Pretransplantation status | |||
| 1st remission | 1.00 | — | — |
| More than 1st remission | 1.93 | 1.10-3.37 | .02 |
| SD/PD | 2.95 | 1.72-5.06 | .001 |
| Previous transplantation | |||
| 0 or 1 | 1.00 | — | — |
| 2 or more | 2.05 | 1.18-3.59 | .01 |
| Donor-recipient sex combination | |||
| All others | 1.00 | — | — |
| Female donor-male recipient | 1.45 | 0.99-2.13 | .06 |
| Progression-free survival | |||
| Pretransplantation status | |||
| 1st remission | 1.00 | — | — |
| More than 1st remission | 1.45 | 0.93-2.25 | .1 |
| SD/PD | 2.42 | 1.58-3.70 | .001 |
| Alemtuzumab conditioning | |||
| No | 1.00 | — | — |
| Yes | 1.83 | 1.29-2.61 | .001 |
| TRM | |||
| Donor-recipient sex combination | |||
| Others | 1.00 | — | — |
| Female donor-male recipient | 2.45 | 1.45-4.15 | .001 |
| Disease duration | |||
| Less than 1 y | 1.00 | — | — |
| At least 1 y | 2.25 | 1.27-3.98 | .005 |
— indicates not applicable.
Response after transplantation
Overall, 25% (58 of 229) and 48% (110 of 229) of patients attained CR and PR, respectively. The disease was stable in 12% (28 of 229), and 7% (15 of 229) had progressive disease. Eight percent (18 of 229) were not evaluable due to treatment-related deaths. Of the patients (10%) in CR prior to the transplantation, 87% remained in CR. Of the patients who received a transplant in PR, 20% (24 patients) attained a CR after transplantation, 66% (81 patients) remained in PR, and 7% (8 patients) had progressive disease. In patients with progressive disease at transplantation, 18% (11 patients) and 27% (17 patients) achieved CR and PR, respectively, and in 34% (21 patients) their disease stabilized. Only 5 patients in this group had early disease progression (Table 5). Response assessment was at or after 100 days after transplantation.
Remission status before and after RIC allograft
Status at transplantation . | Status after transplantation . | . | . | . | . | . | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| . | CR (%) . | PR (%) . | SD (%) . | Progress/relapse (%) . | Not evaluable (%) . | Total . | |||||
| CR | 20 (87) | 2 (9) | 0 (0) | 1 (4) | 0 (0) | 23 | |||||
| PR | 24 (20) | 81 (66) | 0 (0) | 8 (7) | 7 (7) | 122 | |||||
| SD | 3 (14) | 10 (46) | 7 (32) | 1 (4) | 1 (4) | 22 | |||||
| PD | 11 (18) | 17 (27) | 21 (34) | 5 (8) | 8 (13) | 62 | |||||
| Total | 58 (23) | 110 (48) | 28 (12) | 15 (7) | 18 (8) | 229 | |||||
Status at transplantation . | Status after transplantation . | . | . | . | . | . | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| . | CR (%) . | PR (%) . | SD (%) . | Progress/relapse (%) . | Not evaluable (%) . | Total . | |||||
| CR | 20 (87) | 2 (9) | 0 (0) | 1 (4) | 0 (0) | 23 | |||||
| PR | 24 (20) | 81 (66) | 0 (0) | 8 (7) | 7 (7) | 122 | |||||
| SD | 3 (14) | 10 (46) | 7 (32) | 1 (4) | 1 (4) | 22 | |||||
| PD | 11 (18) | 17 (27) | 21 (34) | 5 (8) | 8 (13) | 62 | |||||
| Total | 58 (23) | 110 (48) | 28 (12) | 15 (7) | 18 (8) | 229 | |||||
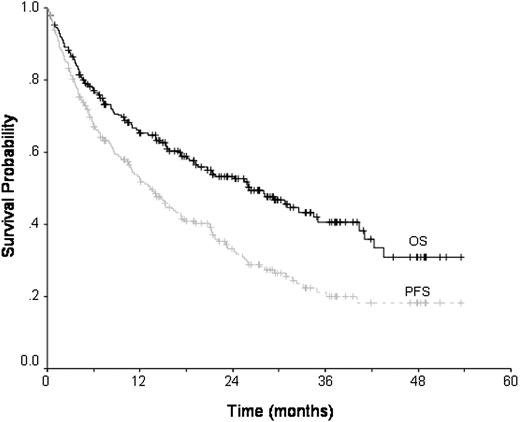
Survival
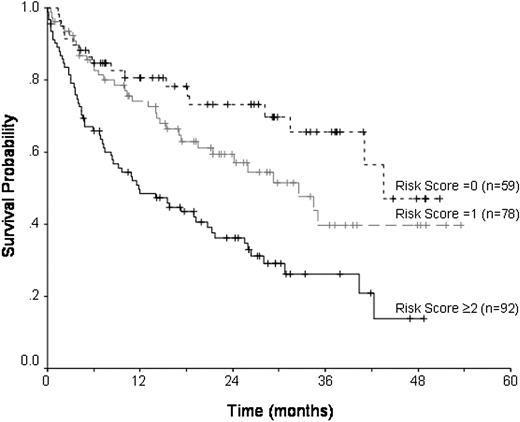
With a median follow-up of 28 months, 115 patients are alive (range, 1-53 months). The estimated overall survival at 3 years is 40.6% (CI, 33%-49%) (Figure 1). On univariate analysis (Table 3) a CR or PR prior to transplantation was associated with an improved overall survival (P = .001), with patients in first remission having the best outcomes (P = .001). One prior autograft did not confer an adverse prognosis compared with no prior autograft, 3-year survivals of 46% and 41%, respectively. Patients receiving more than one prior autograft had an inferior survival of 23% at 3 years (P = .003). Disease duration of less than 1 year (P = .002), and the use of a related versus unrelated donor (P = .002) and sex combinations other than male patients with female donors (P = .03) were also associated with better survival. The use of busulfan in conditioning was associated with inferior survival (P = .01). Factors that retained significance on multivariate analysis were transplantation in first remission, and less than 2 prior autografts (Table 4); donor-recipient sex combinations were of borderline significance (P = .06). In patients with neither of these risk factors, survival at 3 years was 66% (Figure 2).
Overall and progression-free survival. OS indicates overall survival; PFS, progression-free survival.
Overall and progression-free survival. OS indicates overall survival; PFS, progression-free survival.
Disease relapse and progression
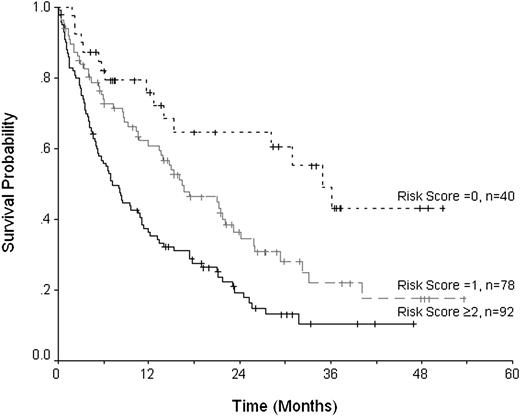
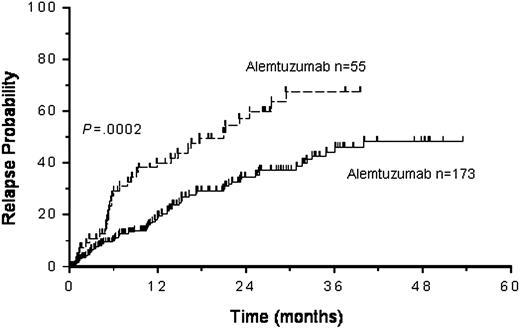
Progression or relapse of disease occurred in 93 patients (41%) following transplantation, and 53 patients have died from disease. The cumulative probability of progression was 50%, and the progression-free survival was 21.3% (CI, 15%-29%) at 3 years. The following factors were associated with an adverse progression-free survival on univariate analysis (Table 3): chemoresistance (P = .001), patients in more than first remission (P = .001), 2 or more prior autografts (P = .02), the use of an unrelated donor (P = .02), and the use of alemtuzumab in the conditioning (P = .004). Chemoresistant disease and the use of alemtuzumab retained significance on multivariate analysis (Table 4 and Figure 3).
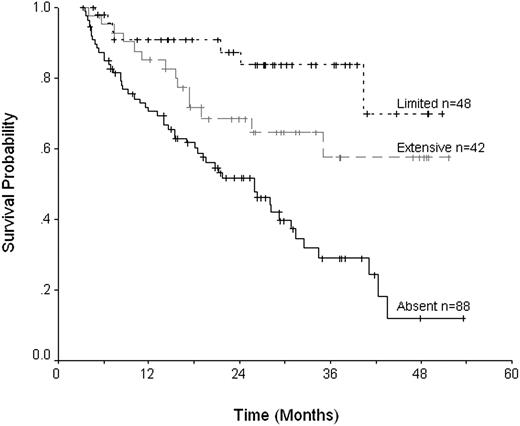
Of the posttransplantation variables, the development of any cGvHD was associated with better overall and progression-free survivals (P < .001). Overall survival was best in those patients with limited disease, 84% at 3 years versus 58% for extensive disease, and 29% in the absence of cGvHD (Figure 4). PFS was likewise better in patients with limited disease, 46% compared with 30% for extensive cGvHD and 12% without cGvHD. An increase in TRM associated with extensive cGvHD did not reach significance. For aGvHD no effect on overall survival or progression-free survival could be demonstrated for either the presence or severity of disease, although grades III to IV aGvHD were associated with an increased TRM.
Donor lymphocyte infusions (DLIs)
Eighty patients received a median number of 2-donor lymphocyte infusions (range, 1-6) with the final cell dose infused being a median of 1 × 107 CD3/kg (range, 1 × 106-3 × 108 CD3/kg). The median time from transplantation to DLI was 176 days. The main indication for DLI was disease relapse or progression in 49 of 80 patients. DLI was given preemptively in 13 patients and to induce full donor chimerism in 12 others. In 6 patients the indication was unknown. The response after DLI is known in 70 patients with CR and PR rates being 23% and 40%, respectively. Thirty-seven percent had stable or progressive disease. Data were available for 46 of 49 of the patients receiving DLI for relapse or disease progression of whom 15% attained CR and 37% PR.
Overall survival with respect to number of risk factors (chemosensitive disease, > 1 prior to transplantation; and female donor-male recipient; 0, 1, and ≥ 2).
Overall survival with respect to number of risk factors (chemosensitive disease, > 1 prior to transplantation; and female donor-male recipient; 0, 1, and ≥ 2).
Progression-free survival with respect to the presence of the risk factors: chemosensitive disease and alemtuzumab conditioning.
Progression-free survival with respect to the presence of the risk factors: chemosensitive disease and alemtuzumab conditioning.
Discussion
Reduced-intensity conditioned transplants are clearly feasible in many patients with myeloma. The sustained engraftment rate at 93% was similar to that seen with conventional allogeneic transplantation and is comparable to data from single and multicenter studies.12-14,16,18 The majority of patients achieved full-donor chimerism at approximately 3 months after transplantation, and secondary graft failure was infrequent.
The primary motivation for the development of reduced-intensity conditioning has been to decrease the TRM. As the TRM in conventional allografts increases with the ages of both the donor and recipient,19,20 a secondary aim of RICT is to extend the age range of patients that might benefit from more aggressive therapy. This is particularly important in a malignancy such as myeloma whereby the median age at onset is in the seventh decade. In our study early TRM at day 100 was low at 10%, but it is important to recognize the continuing attrition rate with the TRM rising to 26% at 2 years. Consistent with this is the observation that many complications of allogeneic transplantation such as GvHD are being delayed by RICT rather than being prevented.21 The low TRM was also not universal, and male patients with a female donor who received a transplant more than 1 year from diagnosis had a TRM of 52% at 1 year. A similarly high risk was seen in other studies of heavily pretreated patients.12,18 In contrast to conventional allografts, age was not found to be an important factor in predicting outcome.
Overall survival with respect to the presence of chronic graft-versus-host disease.
Overall survival with respect to the presence of chronic graft-versus-host disease.
At present there is no clear definition of a RICT. In this study the classification of RICT was based on the current EBMT recommendations. Procedures involving TBI doses greater than 6 Gy were not included. A total of 18 different conditioning regimens were used. This figure rose to 28 when the various combinations of chemotherapy and T-cell depletion methods were included. The commonest regimens were those that included melphalan, reflecting the widespread use of this drug as an antimyeloma agent both in low and high doses. Despite this no clear advantage or disadvantage could be demonstrated for the use of any one drug or the common drug combinations. The use of antibody therapy and most notably the use of alemtuzumab did affect disease outcomes. The negative effect of alemtuzumab is likely to be due to the increased relapse rate (Figure 5), although there was also an increase in infection as a cause of death and a nonsignificant increase in late TRM beyond day 200. The profound T-cell depletion induced by alemtuzumab is likely to be contributing to this late mortality. The incidence of grades III to IV aGvHD was the same in the alemtuzumab and ATG groups, but the incidence of cGvHD was lower in the alemtuzumab group. This difference was not marked (40% versus 32%). CD52 expression is not limited, however, to T cells and depletion of other cellular targets of alemtuzumab such as natural killer cells or host dendritic cells may be important in contributing to the adverse effect on progression-free survival seen.22,23
The rationale for allogeneic transplantation is first the ability to deliver high-dose chemotherapy supported by hematopoietic progenitor cells that are free of tumor contamination and second the harnessing of an immune antitumor effect. The efficacy of RICT is dependent primarily on the latter effect. The improvement in progression-free and overall survival associated with the development of cGvHD has been reported24,25 and supports the existence of a graft-versus-myeloma (GvM) effect, as do the responses seen with donor lymphocyte infusions. There is nothing in these data, however, to support the existence of a specific GvM immune effect as opposed to a more nonspecific allogeneic response. At present there is no evidence of long-term disease control with a continued fall off in the PFS being seen even in those patients with cGvHD. While it may be premature to exclude a plateau to the survival curves in the majority of patients, the disease progresses despite transplantation. Furthermore, while there may be an important GvM effect in a minority of patients, given the importance of disease remission status at the time of transplantation it would appear that it is insufficiently potent to overcome bulk or progressive disease.
Data on the extent of prior treatment are limited, although surrogate markers such as prior high-dose therapy and time from diagnosis to transplantation are associated with adverse outcomes. The latter did not retain significance in multivariate analysis. Consistent with recent data26 there did not appear to be an adverse outcome associated with one prior autograft when this was done as part of a planned 2-stage procedure or as a separate therapy. Several ongoing studies both in Europe and North America are using autologous transplantation as cytoreductive therapy prior to a RICT. These data would suggest that this approach is unlikely to be detrimental.
Male sex and mismatch between donor and recipient sex are well recognized but not universal risk factors for a higher treatment-related mortality in allogeneic transplantation.6,27 Male patients in this series had an inferior survival, which was largely explained by those male patients with female donors who had significantly adverse outcomes with respect to survival and treatment-related mortality.
This series represents the largest cohort of myeloma patients who received a transplant with reduced-intensity conditioning protocols reported to date. It is based on data collected from 33 centers and as such documents outcomes that are being achieved throughout and beyond Europe. The outcome from RICT for more advanced disease and in extensively pretreated patients is disappointing. Clearly, appropriate patient selection is essential to optimize the outcome of this strategy. Incorporating cytogenetics or other biologic markers may be helpful in identifying those patients in whom this approach should be tested early in their disease course; however prognosis of patients with high-risk disease such as those with 13q deletions continues to be poor even after RICT.26
This study begins to identify clinical factors that will help with patient selection. Reserving this treatment for patients in whom other therapies have failed is unlikely to result in beneficial outcomes. The best outcomes were seen in patients who received a transplant in remission and earlier in the course of the disease. This study suggests that sex is an important factor in donor selection and that the use of alemtuzumab may be detrimental. The effect of cGvHD on OS and PFS is suggestive of a GvM effect, although whether this can be separated from a nonspecific alloimmune effect is uncertain.
Appendix
The following centers and investigators contributed patients to this study. Austria: H. Greinix, AKH und Universitaetskliniken Wien, Vienna; Belgium: M. Boogaerts, University Hospital Gasthuisberg, Leuven; Czech Republic: J. Mayer, University Hospital Brno, Brno; Finland: L. Volin and T. Ruutu, Helsinki University Central Hospital, Helsinki; France: F. Garban, Hopital A. Michallon, Grenoble; M. Michallet, Hopital E. Herriot, Lyon; D. Blaise, Institut Paoli Calmettes, Marseille; G. Socie and L. Ades, Hopital St Louis, Paris; J.M. Boiron, CHU Bordeaux Hôpital, Pessac; M. Attal, Hopital de Purpan, Toulouse; Germany: A. Zander, University Hospital Eppendorf, Hamburg; H. Einsele, Medizinische Klinik, Tubingen; Israel: A. Nagler and A. Shimoni, Chaim Sheba Medical Center, Tel-Hashomer; Italy: A. Rambaldi, Ospedale Bergamo, Bergamo; A. Bosi, Ospedale di Careggi, Firenze; A. Bacigalupo, Ospedale San Martino, Genova; F. Ciceri, Istituto Scientifico H.S. Raffaelle, Milano; P. Iacopino, Azienda Ospedaliera, Reggio Calabria; A. Carella, Istituto di Ricovero e Cura a Carattere Scientifico (IRCCS), Casa Sollievo della Sofferenza, San Giovanni Rotondo; F. Locatelli, Azienda Ospedaliera S. Giovanni, Torino; Poland: M. Komarnicki, K. Marcinkowski University, Poznan; Spain: A. Urbano-Ispizua, Institute of Hematology and Oncology, Barcelona; J. Sierra, Hospital Santa Creu I Sant Pau, Barcelona; D. Caballero, Hospital Clínico, Salamanca; Sweden: M. Brune, Sahlgrenska University Hospital, Goeteborg; O. Ringden, Karolinska University Hospital-Huddinge, Stockholm; Switzerland: A. Gratwohl, Kantonsspital, Basel; United Kingdom: D. Milligan, Birmingham Heartlands Hospital, Birmingham; G. Mufti, Guy's, Kings and St Thomas (GKT) School of Medicine, London; J. Apperley and C. Crawley, Hammersmith Hospital, London; J. Cavenagh, St Bartholomew's and The Royal London Hospital, London; S. MacKinnon and K. Peggs, University College London Hospital, London; R. Chopra, Christie Hospital NHS Trust, Manchester.
Prepublished online as Blood First Edition Paper, February 24, 2005; DOI 10.1182/blood-2004-06-2387.
A complete list of the members of the Chronic Leukaemia Working Party of the European Group for Blood and Marrow Transplantation (EBMT) appears in the “Appendix.”
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal