Abstract
Jagged1-mediated Notch signaling has been suggested to be critically involved in hematopoietic stem cell (HSC) self-renewal. Unexpectedly, we report here that inducible Cre-loxP–mediated inactivation of the Jagged1 gene in bone marrow progenitors and/or bone marrow (BM) stromal cells does not impair HSC self-renewal or differentiation in all blood lineages. Mice with simultaneous inactivation of Jagged1 and Notch1 in the BM compartment survived normally following a 5FU-based in vivo challenge. In addition, Notch1-deficient HSCs were able to reconstitute mice with inactivated Jagged1 in the BM stroma even under competitive conditions. In contrast to earlier reports, these data exclude an essential role for Jagged1-mediated Notch signaling during hematopoiesis.
Introduction
Hematopoietic stem cells (HSCs) exhibit self-renewing capacity as well as the ability to give rise to more committed progenitors that differentiate into all hematopoietic lineages.1 The molecular mechanisms regulating stem cell self-renewal and/or differentiation are only poorly understood. Among the proteins that have been postulated to be involved in hematopoietic stem cell maintenance are the Notch receptors and their ligands.2 Mammals have 4 Notch receptors (Notch1-4) that bind 5 different ligands (Jagged1-2, Delta-like 1-3-4). Expression of a constitutively active form of Notch1 (N1) in murine bone marrow progenitors can lead to increased HSC-self-renewal3 or to the immortalization of stem cell like progenitors capable of undergoing lymphoid and myeloid differentiation both in vitro and in vivo.4 In addition, coculture of murine or human HSCs with immobilized Notch ligands, or feeder cells expressing such ligands, can maintain or even enhance HSC self-renewal.5-9
Recently, osteoblasts expressing the Notch ligand Jagged1 (J1) were identified as being part of the hematopoietic stem cell niche. Osteoblast-specific expression of the activated parathyroid hormone-related protein receptor results in increased numbers of osteoblasts expressing high levels of J1. The increase in osteoblasts correlates with an increase in the number of HSCs, with evidence of N1 activation in vivo.10 These results were interpreted to mean that J1-expressing osteoblasts regulate HSC homeostasis through N1 activation.
To definitively assess the role of J1 in the hematopoietic system, we have generated inducible gene-targeted mice for J1. Surprisingly, inactivation of J1 in either bone marrow (BM) progenitors or BM stromal cells had no effect on HSC maintenance. In addition, N1-deficient HSCs transplanted into mice with inactivated J1 in the BM stroma reconstituted BM chimeras normally. Our data exclude an essential contribution of J1-mediated N1 signaling for HSC self-renewal or differentiation.
Study design
Generation and conditional inactivation of mice with a loxP-flanked J1
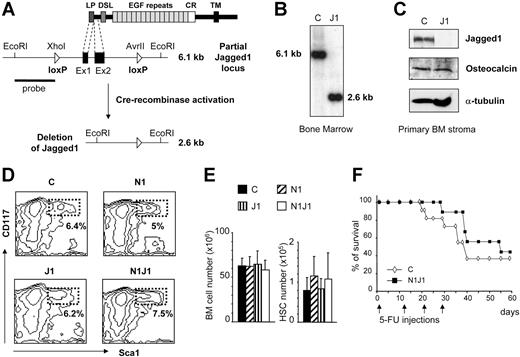
A J1 genomic clone was isolated from a mouse genomic library using an oligonucleotide complementary to part of the first coding exon. LoxP sites were introduced into an XhoI site and into an AvrII site 1075 base pair (bp) upstream and 2364 bp downstream of the ATG, flanking the first 2 coding exons. Targeting vector, embryonic stem (ES) cells, removal of the phosphoglycerate kinase (PGK)–neomycin cassette, and the generation of the chimeric mice were as previously published.11 J1-/- mice generated from the J1lox/lox by Cre recombinase expression in the germ cells showed embryonic lethality as described in the conventional knock-out.12 J1lox/lox mice were interbred with Mx-Cre mice13 to generate J1lox/loxMx-Cre mice. Inducible activation of the Cre recombinase was done as previously described.11 The deletion efficiency was assessed by Southern blot analysis of EcoRI-digested genomic DNA hybridized with the indicated probe (Figure 1A) and quantified using a Phosphorimager (FUJI FILM BAS-1000; Fuji, Tokyo, Japan).
Characterization of mice with inducible inactivation of Jagged1 in the bone marrow. (A) Schematic representation of the mouse J1 protein containing the leader peptide (LP), the Delta, Serrate, Lag-2 (DSL) domain, and epidermal growth factor (EGF)–like repeats, followed by a cystein-rich domain (CR) and a transmembrane domain (TM). Part of the genomic organization of floxed Jagged1 is shown, indicating the first 2 coding exons (black boxes) flanked by loxP sequences (triangles) before and after induction of the Cre recombinase. (B) Southern blot analysis of genomic DNA derived from BM cells of littermate controls (J1lox/lox, LM control) and induced J1lox/loxMx-Cre mice showing a close-to-100% deletion efficiency. (C) Western blot analysis of J1 and osteocalcin expression using protein extracts derived from primary BM stromal cultures of LM controls and induced J1lox/loxMx-Cre mice. (D) Representative FACS analysis of HSCs defined by CD117 and Sca1 gated on Lin-, for BM cells derived from control, induced N1-/-, induced J1-/-, and induced N1-/- J1-/- double-deficient mice. (E) Absolute numbers ± SD of either total BM cells or HSC (lin-CD117+Sca1+) from control and indicated conditional knockout mice (n = 10 mice per group). (F) Survival curve of control (⋄) and N1/J1 double-deficient mice (▪) after 5-FU treatment. Arrows indicate 5-FU injections. Results were analyzed with a log-rank test and expressed as Kaplan-Meier survival curves (n = 11 for control and 9 for N1/J1-induced double knockouts, P = .468).
Characterization of mice with inducible inactivation of Jagged1 in the bone marrow. (A) Schematic representation of the mouse J1 protein containing the leader peptide (LP), the Delta, Serrate, Lag-2 (DSL) domain, and epidermal growth factor (EGF)–like repeats, followed by a cystein-rich domain (CR) and a transmembrane domain (TM). Part of the genomic organization of floxed Jagged1 is shown, indicating the first 2 coding exons (black boxes) flanked by loxP sequences (triangles) before and after induction of the Cre recombinase. (B) Southern blot analysis of genomic DNA derived from BM cells of littermate controls (J1lox/lox, LM control) and induced J1lox/loxMx-Cre mice showing a close-to-100% deletion efficiency. (C) Western blot analysis of J1 and osteocalcin expression using protein extracts derived from primary BM stromal cultures of LM controls and induced J1lox/loxMx-Cre mice. (D) Representative FACS analysis of HSCs defined by CD117 and Sca1 gated on Lin-, for BM cells derived from control, induced N1-/-, induced J1-/-, and induced N1-/- J1-/- double-deficient mice. (E) Absolute numbers ± SD of either total BM cells or HSC (lin-CD117+Sca1+) from control and indicated conditional knockout mice (n = 10 mice per group). (F) Survival curve of control (⋄) and N1/J1 double-deficient mice (▪) after 5-FU treatment. Arrows indicate 5-FU injections. Results were analyzed with a log-rank test and expressed as Kaplan-Meier survival curves (n = 11 for control and 9 for N1/J1-induced double knockouts, P = .468).
BM chimeras
CD45.1+ B6-SJL mice were purchased from The Jackson Laboratory (Bar Harbor, ME) and CD45.2+ C57BL/6 mice from Harlan (Horst, The Netherlands). N1 conditional gene-targeted mice were as previously described.11 Mixed BM chimeras were prepared as previously described.11 3 × 106 BM cells of each genotype were used to set up the mixed BM chimeras. Deletion efficiency of the N1 gene in donor N1lox/loxMx-Cre HSCs was assessed by polymerase chain reaction (PCR) as published,14 using genomic DNA from HSC sorted 8 weeks after transplantation.
5-FU treatment
N1lox/loxJ1lox/loxMx-Cre mice were injected with 5-fluorouracil (Sigma-Aldrich, Buchs, Switzerland) as previously described15 4 weeks after induced deletion of the floxed genes. The survival rates of the groups were determined.
Immunoblot analysis
Immunoblot analysis was performed as described16 using primary BM stromal cells from control and J1lox/loxMx-Cre mice. Primary BM stroma was prepared by cultivating total BM cells in minimum essential media α (MEMα) medium. After 3 days, cultures were washed and adherent cells were left in culture and grown to confluency. The following primary antibodies were used: anti-Jagged1 (H114; Santa Cruz Biotechnology, Santa Cruz, CA), anti-osteocalcin (Anawa, Wangen/Duebendorf, Switzerland), and anti–α-tubulin (B-5-1-2; Sigma-Aldrich).
Flow cytometry
Single-cell suspensions of BM were prepared and stained following standard protocols for fluorescence-activated cell-sorter scanner (FACS) analysis using monoclonal antibodies conjugates as previously published.16 The anti-CD135 antibody (clone A2F10) was from eBioscience (San Diego, CA) and was conjugated to Alexa647 as described.16
Results and discussion
Mx-Cre–induced inactivation of J1 in BM and BM stroma
To circumvent the early embryonic lethality of conventional gene-targeted mice for J1,12 we generated mice in which the first 2 coding exons are flanked by loxP sites (Figure 1A). The Mx-Cre transgene13 was intercrossed into J1lox/lox mice in order to inducibly inactivate the J1 gene in BM progenitors. The deletion efficiency for the floxed J1 gene segment in BM is close to 100% as shown by Southern blot analysis11 (Figure 1B). Western blot analysis of primary BM stromal cultures from induced J1-deficient mice demonstrate a complete absence of J1 protein (Figure 1C), implying that the Mx-Cre system also is suitable for gene inactivation in BM stromal cells. Since J1 is expressed by osteoblasts, which are believed to be part of the HSC stem cell niche,10,17 we investigated whether loss of J1 may influence osteoblast outgrowth or number in primary stromal cultures. No difference between J1-deficient and control BM stromal cultures was observed for expression of osteocalcin (an osteoblast-specific marker18 ) examined by Western blot analysis (Figure 1C) or immunohistochemistry (data not shown), indicating that loss of J1 does not influence the outgrowth of osteoblasts in primary stromal cultures.
Normal HSC self-renewal and differentiation in the absence of J1 and/or N1
To investigate whether J1 and/or N1 may play important roles for HSC homeostasis under steady-state conditions, we phenotypically investigated the HSC compartment of mice 4 to 8 weeks after inactivation of either J1 or N1, or simultaneous inactivation of both genes. As shown in representative FACS contour plots in Figure 1D, no significant differences between control and any of the indicated gene-targeted mice were observed for the HSC compartment defined as lin-CD117+Sca1+. In addition, absolute numbers of the total BM compartment or HSCs showed no significant differences (Figure 1E). These results indicate that J1 and/or N1 are dispensable for HSC steady-state homeostasis. However, this approach does not address whether either of these proteins is important for HSC self-renewal under challenge conditions or in a competitive repopulation system. In order to answer these questions, we first used an in vivo challenge system by exposing control and induced N1J1 double-knockout mice to repeated doses of the antimetabolite 5-fluorouracil (5-FU) (Figure 1F). The percentage of surviving N1J1 mice was at least as high as for control animals, indicating that HSCs of the N1J1-deficient animals could re-enter the cell cycle in order to repopulate all hematopoietic lineages.
The readout of the 5-FU experiment is survival of the challenged animals. However, it does not address the reconstitution efficiency of J1 and/or N1-deficient HSCs. Furthermore it is unclear whether wild-type or N1-deficient HSCs can reconstitute mice in which J1 is inactivated in the BM stroma. Thus, competitive mixed bone chimeras were set up in various combinations to address these questions (Figure 2). All chimeras were analyzed 8 to 20 weeks after transplantation. First, lethally irradiated wild-type hosts (CD45.1+) were reconstituted with either CD45.2+ J1-deficient BM (Figure 2A) or control BM, each mixed at a 2:1 ratio with CD45.1+ wild-type BM. The relative number of CD45.2+ BM cells, lin- cells, and HSCs (lin-CD117+Sca1+) within donor-derived BM was similar in both control and J1-deficient chimeras, indicating that J1-deficient HSCs can self-renew as efficiently as wild-type cells. To investigate whether J1 expression by osteoblasts and BM stroma is important for efficient reconstitution in a competitive situation, mixed BM chimeras were set up using lethally irradiated CD45.2+ J1-deficient mice as hosts. These were reconstituted with either CD45.1+ control or N1-deficient BM mixed with CD45.2+ wild-type BM. Surprisingly, the relative numbers of cells in phenotypically defined progenitor subsets, including HSC (lin-CD117+Sca1+), long-term (LT)–HSC (lin-CD117+Sca1+CD135-), short-term (ST)–HSC (lin-CD117+Sca1+CD135+), common myeloid progenitors (CMP, lin-CD117+Sca1-), and common lymphoid progenitors (CLP, lin-CD117intSca1int) (Figure 2B top panel), as well as more mature lymphoid, myeloid, and erythroid lineages (Figure 2B bottom panel, as indicated), was similar in both control and N1-deficient chimeras, irrespective of whether the host animals were wild-type or J1-deficient. As expected,11 T cells were absent, and only B cells were detected in the thymus of mice reconstituted with N1-deficient HSCs (Figure 2B and data not shown). Identical results were obtained using noncompetitive chimeras (data not shown), excluding the possibility that a defect may have been missed due to a non–cell-autonomous rescue dependent upon wild-type competitor cells. Furthermore, substitution of host Jagged1-deficient stromal cells by contaminating donor wild-type stromal cells from the BM inoculum appears to be very unlikely since BM stromal cultures 15 weeks after transplantation were still deficient for Jagged1 as assessed by Western blot analysis (data not shown). In order to exclude the possibility that reconstitution of the BM chimeras is due to a few BM cells that have escaped deletion for N1, HSCs of the indicated chimeras were sorted and assessed for their deletion status by PCR. The PCR shows the expected pattern of the inactivated N1 allele (Figure 2C), confirming that efficient repopulation can be achieved by N1-deficient HSCs even in absence of J1 expression by the BM stroma.
Simultaneous lack of Jagged1 in the BM stroma and Notch1 in HSCs of mixed BM chimeras does not affect their self-renewal potential. (A) Mixed BM chimeric mice analyzed 14 to 15 weeks after reconstitution with a 1:2 mixture of B6-SJL (CD45.1+) and either J1lox/lox or J1-/- (CD45.2+) BM-derived populations. Percentage of CD45.2+ BM cells, CD45.2+Lin- cells, and HSCs (CD117+Sca1+) gated on lin-CD45.2+ cells from either J1lox/lox (▪) or J1-/- mice (▦). The bars represent mean ± SD (n = 8). (B) Mixed BM chimeric mice using either J1lox/lox or induced J1-/- recipients (both CD45.2+) analyzed 8 to 20 weeks after reconstitution with a 1:1 mixture of C57BL/6 (CD45.2+) and either N1lox/lox or N1-/- (CD45.1+) BM-derived populations. (Top row) Percentage of CD45.1+ BM cells, CD45.1+Lin- cells, and HSCs (CD117+Sca1+), long-term-HSCs (CD117+Sca1+CD135-), short-term-HSCs (CD117+Sca1+CD135+), CMPs (CD117+Sca1-), and CLPs (CD117intSca1int) gated on lin-CD45.1+ BM cells. (Bottom row) B cells (B220+), immature myeloid cells (CD11b+/Gr1lo/int), eosinophils (CD11b+/Gr1+), early erythroblasts (Ter119+) gated on CD45.1+ cells, and percentage of CD45.1+ cells in the thymus. Most of the thymic CD45.1+ cells derived from N1-/- BM cells are B cells. Control into control (▪; n = 8), control into J1-/- (▨; n = 3), N1-/- into control (▥; n = 7), or N1-/- into J1-/- (□; n = 14) mixed BM chimeras. The bars represent mean ± SD. (C) PCR analysis of the deletion of the N1 gene using genomic DNA from CD45.1+ HSCs sorted 8 weeks after transplantation of the indicated chimeric mice.
Simultaneous lack of Jagged1 in the BM stroma and Notch1 in HSCs of mixed BM chimeras does not affect their self-renewal potential. (A) Mixed BM chimeric mice analyzed 14 to 15 weeks after reconstitution with a 1:2 mixture of B6-SJL (CD45.1+) and either J1lox/lox or J1-/- (CD45.2+) BM-derived populations. Percentage of CD45.2+ BM cells, CD45.2+Lin- cells, and HSCs (CD117+Sca1+) gated on lin-CD45.2+ cells from either J1lox/lox (▪) or J1-/- mice (▦). The bars represent mean ± SD (n = 8). (B) Mixed BM chimeric mice using either J1lox/lox or induced J1-/- recipients (both CD45.2+) analyzed 8 to 20 weeks after reconstitution with a 1:1 mixture of C57BL/6 (CD45.2+) and either N1lox/lox or N1-/- (CD45.1+) BM-derived populations. (Top row) Percentage of CD45.1+ BM cells, CD45.1+Lin- cells, and HSCs (CD117+Sca1+), long-term-HSCs (CD117+Sca1+CD135-), short-term-HSCs (CD117+Sca1+CD135+), CMPs (CD117+Sca1-), and CLPs (CD117intSca1int) gated on lin-CD45.1+ BM cells. (Bottom row) B cells (B220+), immature myeloid cells (CD11b+/Gr1lo/int), eosinophils (CD11b+/Gr1+), early erythroblasts (Ter119+) gated on CD45.1+ cells, and percentage of CD45.1+ cells in the thymus. Most of the thymic CD45.1+ cells derived from N1-/- BM cells are B cells. Control into control (▪; n = 8), control into J1-/- (▨; n = 3), N1-/- into control (▥; n = 7), or N1-/- into J1-/- (□; n = 14) mixed BM chimeras. The bars represent mean ± SD. (C) PCR analysis of the deletion of the N1 gene using genomic DNA from CD45.1+ HSCs sorted 8 weeks after transplantation of the indicated chimeric mice.
In conclusion, our results unequivocally demonstrate that J1-mediated N1 signaling is dispensable for HSC self-renewal and differentiation. Nevertheless, we cannot exclude the possibility that other Notch receptors and/or ligands may substitute for N1 and/or J1 in the conditional knockout mice we have used. Moreover, even if Notch signaling is dispensable for hematopoiesis under physiological conditions, its potential to expand HSCs in vitro could still be exploited to achieve specific therapeutic goals.
Prepublished online as Blood First Edition Paper, November 18, 2004; DOI 10.1182/blood-2004-08-3207.
S.J.C.M. was partly supported by the Imperial Cancer Research Fund (Cancer Research UK) and the Roche Foundation. This work was in part funded by the Swiss National Science Foundation and the EMBO Young Investigator Program (YIP).
An Inside Blood analysis of this article appears in the front of this issue.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.
We are grateful to A. Wilson and V. Besseyrias for excellent technical expertise, and to F. Grosjean and P. Zaech for cell sorting.


This feature is available to Subscribers Only
Sign In or Create an Account Close Modal