Abstract
FMS-like tyrosine kinase 3 (FLT3) is a receptor tyrosine kinase that is constitutively activated in approximately 30% of acute myelogenous leukemia (AML) patients and appears to confer an adverse prognosis. Thus, development of inhibitors and/or antibodies that specifically target FLT3 has been of substantial interest. In this regard, phase 1 and 2 trials involving FLT3 inhibitors have recently reported FLT3 inhibition and leukemic blast reduction in some patients. Despite this, issues such as specificity and resistance need to be addressed. Consequently, the development of alternative approaches for targeting FLT3 would be of great consequence. In the present report, we demonstrate that FLT3 siRNA effectively down-regulates FLT3 expression in Ba/F3 cells transfected with FLT3 containing an activating internal tandem duplication (ITD) in the juxtamembrane domain and FLT3-ITD–positive Molm-14 human leukemia cells. Treatment with the FLT3 siRNA results in growth inhibition and apoptosis of these cells. Furthermore, siRNA-induced down-regulation of FLT3 increased the sensitivity of both cell lines to treatment with the FLT3 inhibitor MLN518. This illustrates the potential benefit of combined therapeutic approaches.
Introduction
Acute myelogenous leukemia (AML) is an aggressive hematologic malignancy that accounts for approximately one third of all new leukemia cases. Presently, induction therapies are capable of achieving complete remission in 50% to 70% of patients. However, long-term survival rates remain low (< 20%) due to a high frequency of relapse.1 Consequently, the development of more effective therapeutic strategies is essential. The receptor FMS-like tyrosine kinase 3 (FLT3) has received considerable attention as a potential target for rational therapy due to the fact that it is the single most frequently mutated gene in AML. Nearly 30% of patients possess an internal tandem duplication (ITD) within the juxtamembrane domain, and an additional 7% have point mutations within the catalytic domain of the kinase.2,3 These mutations lead to constitutive activation of the kinase and appear to have an unfavorable prognosis.4-6
To date, several different FLT3 kinase inhibitors have been developed and tested in vitro7-9 and in early clinical trials with varied degrees of success.10-12 More specifically, 12.5% to 81.3% of patients in these studies achieved a more than 50% blast reduction with the duration of response ranging from 2 weeks to 5 months. However, given the fact that AML is an aggressive and heterogeneous disease, these results are not surprising and merely underscore the need for additional therapeutic approaches. It is conceivable that inhibition of kinase activity combined with simultaneous down-regulation of protein expression may be synergistic. One methodology for targeting FLT3 involves RNA interference (RNAi)–induced down-regulation of FLT3 expression. Thus, the goal of the present study was to examine the ability of FLT3 siRNA to effectively down-regulate FLT3 expression in AML cell lines and to determine whether the combination of siRNA and a FLT3 inhibitor is more effective than either agent alone.
Study design
Cell lines and reagents
Ba/F3 FLT3-ITD transfectants (expressing a reduplication of YEYDLK at amino acid positions 597-602)13 were generated and selected as previously described.14 Molm-14 cells (FLT3-ITD+) were generously provided by Dr Yoshinobu Matsuo (Fujisaki Cell Center, Hayashibara Biochemical Labs, Okayama, Japan).15 Cell lines were maintained in RPMI 1640 supplemented with 10% fetal calf serum (FCS), 1 unit/mL penicillin G, and 1 μg/mL streptomycin at 37°C and 5% CO2. MLN518 was kindly provided by Dr Jin-Chen Yu (Millennium Pharmaceuticals, San Francisco, CA).
Source of siRNA
Nonspecific and FLT3 SMARTpool siRNA duplexes (proprietary target sequences) were purchased from Dharmacon Research (Lafayette, CO).
Transfection via electroporation
Prior to transfection, cells were split and cultured in regular media. On the day of transfection, cells were washed once in Opti-MEM (Life Technologies, Rockville, MD) and resuspended at 107 cells/200 μL. Cells (200 μL) were then incubated with FLT3 siRNA or nonspecific siRNA (1 μg/107 cells) for 10 minutes in a 0.4-cm electroporation cuvette. The cell/siRNA mixture was pulsed once (Ba/F3 FLT3-ITD: 20 ms, 300 V; Molm-14: 20 ms, 250 V) using a square-wave electroporator (BTX Genetronics, San Diego, CA), incubated at room temperature for 30 minutes, and transferred to 96- and/or 6-well plates (Becton Dickinson, Franklin Lakes, NJ).
Flow cytometry
Detection of apoptosis was performed using an Annexin-V–FLUOS staining kit (Roche, Mannheim, Germany) on a FACSAria flow cytometer (Becton Dickinson, Mountain View, CA).
Immunoblotting
Detection of FLT3, phospho–signal transducer and activator of transcription 5 (STAT5), STAT5b, phospho–mitogen-activated protein kinase (MAPK), MAPK, phospho-Akt, and Akt was performed using standard immunoblotting procedures. Antibodies for immunoblotting were anti–phospho-STAT5, anti–phospho-MAPkinase, anti-MAPK, anti–phospho-Akt, anti-Akt (Cell Signaling Technology, Beverly, MA), anti-FLT3, and anti-STAT5b (Santa Cruz Biotechnology, Santa Cruz, CA).
Results and discussion
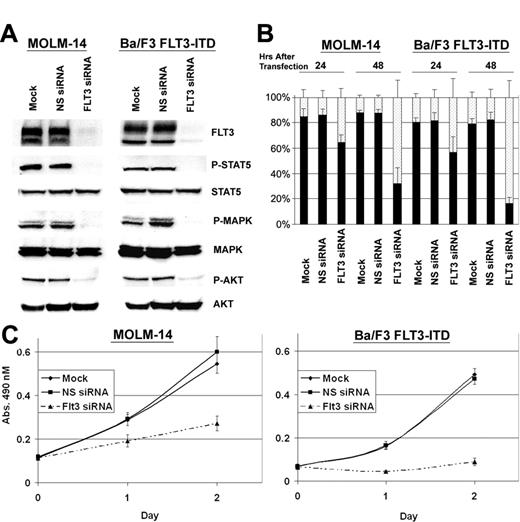
In initial experiments, we determined whether transfection of pooled FLT3 siRNA, targeting 4 separate sites of FLT3, would result in significant down-regulation of FLT3 in Ba/F3 FLT3-ITD cells and Molm-14 cells. Immunoblot analysis revealed that 24 hours following transfection, FLT3 expression was reduced by 91% in the Ba/F3 FLT3-ITD cells and by 85% in the Molm-14 cells, compared with controls transfected with nonspecific siRNA (Figure 1A). Additionally, down-regulation of FLT3 was maintained in both the Ba/F3 FLT3-ITD cells and the Molm-14 cells over a 72-hour period (data not shown). Analysis of the effectiveness of each individual siRNA in the pool was determined to be comparable with the pooled siRNA (data not shown). Immunoblot analysis of downstream signaling molecules demonstrated that siRNA-induced down-regulation of FLT3 resulted in a significant decrease in tyrosine phosphorylation of STAT5 (Ba/F3 FLT3-ITD: 93%; Molm-14: 98%), MAP kinase (Ba/F3 FLT3-ITD: 65%; Molm-14: 72%), and Akt (Ba/F3 FLT3-ITD: 80%; Molm-14: 83%) (Figure 1A). Of note, the decrease in phosphorylation was observed in the absence of an effect on the total protein levels of these proteins. These data suggest that FLT3 is required for the activation and phosphorylation of STAT5, MAP kinase, and Akt, which concurs with previous reports demonstrating heightened activation of these pathways in cell lines and patient samples possessing FLT3 activating mutations.16-19
Electroporation of Molm-14 cells and Ba/F3 FLT3-ITD transfectants with FLT3 siRNA results in down-regulation of FLT3, decreased activation of downstream signaling molecules, and induction of apoptosis. (A) siRNA-induced silencing of FLT3 was assessed by immunoblot of whole cell lysates 24 hours after siRNA transfection. RNAi-induced down-regulation of FLT3 also resulted in decreased phosphorylation of STAT5, MAPkinase, and Akt in both Ba/F3 FLT3-ITD transfectants and Molm-14 cells. NS indicates nonspecific. (B) Ba/F3 FLT3-ITD transfectants and Molm-14 cells demonstrate an increase in apoptosis (annexin/PI-positive cells indicated by ▦; viable cells indicated by ▪) over time when treated with FLT3 siRNA (n = 3 ± SE). (C) Treatment of Ba/F3 FLT3-ITD transfectants and Molm-14 cells (n = 5 ± SE) with FLT3-specific siRNA (▴) results in an overall decrease of proliferation (n = 8 ± SE).  indicates mock; ▪, nonspecific siRNA.
indicates mock; ▪, nonspecific siRNA.
Electroporation of Molm-14 cells and Ba/F3 FLT3-ITD transfectants with FLT3 siRNA results in down-regulation of FLT3, decreased activation of downstream signaling molecules, and induction of apoptosis. (A) siRNA-induced silencing of FLT3 was assessed by immunoblot of whole cell lysates 24 hours after siRNA transfection. RNAi-induced down-regulation of FLT3 also resulted in decreased phosphorylation of STAT5, MAPkinase, and Akt in both Ba/F3 FLT3-ITD transfectants and Molm-14 cells. NS indicates nonspecific. (B) Ba/F3 FLT3-ITD transfectants and Molm-14 cells demonstrate an increase in apoptosis (annexin/PI-positive cells indicated by ▦; viable cells indicated by ▪) over time when treated with FLT3 siRNA (n = 3 ± SE). (C) Treatment of Ba/F3 FLT3-ITD transfectants and Molm-14 cells (n = 5 ± SE) with FLT3-specific siRNA (▴) results in an overall decrease of proliferation (n = 8 ± SE).  indicates mock; ▪, nonspecific siRNA.
indicates mock; ▪, nonspecific siRNA.
Given that RNAi-induced down-regulation of FLT3 results in diminished phosphorylation of several downstream molecules, including Akt, which is known to play a role in cell survival,20 we next assessed whether down-regulation of FLT3 had an effect on cell viability and/or cell proliferation. Indeed, as shown in Figure 1B, the viability of both cell lines decreased following treatment with FLT3 siRNA. Likewise, growth of the siRNA-treated cells was strongly inhibited, compared with controls (Figure 1C). From these data, we conclude that constitutive activation of FLT3 results in activation of downstream signaling pathways, which promote cell survival and/or prevent apoptosis, and that RNAi-induced down-regulation of FLT3 prevents their activation, and thus results in the initiation of apoptosis.
Finally, we determined whether RNAi-induced down-regulation of FLT3 would affect the sensitivity of the Ba/F3 FLT3-ITD transfectants and the Molm-14 cells to treatment with the FLT3 inhibitor MLN518. As previously shown, via annexin/propidium iodide (PI) staining, both cell lines are dependent on the presence of FLT3 protein to prevent apoptosis (Figure 1B). Furthermore, immunoblots demonstrate that FLT3 siRNA is efficient at down-regulating the level of FLT3 in these cells (Figure 1A). To detect synergistic or additive effects of combination treatment with FLT3 kinase inhibitors, we treated the cells with lower concentrations of FLT3 siRNA (range of 1/16 times to 1/256 times the normal dose). FLT3 siRNA treatment alone reduced cell proliferation in a dose-dependent manner, and even the lowest concentrations of FLT3 siRNA tested were effective (Figure 2A). Furthermore, all concentrations of siRNA reduced expression levels of FLT3 and induced apoptosis over a 48-hour period (data not shown).
RNAi-induced down-regulation of FLT3 sensitizes Ba/F3 FLT3-ITD transfectants and Molm-14 cells to treatment with MLN518. (A) Dose-dependent decrease in cell proliferation following treatment with decreasing concentrations of FLT3 siRNA (Ba/F3 FLT3-ITD cells [n = 8] and Molm-14 cells [n = 5]). (B) Treating Ba/F3 FLT3-ITD cells (n = 8) and Molm-14 cells (n = 5) with FLT3 siRNA increases their sensitivity to treatment with the FLT3 inhibitor MLN518. Error bars indicate standard deviation.
RNAi-induced down-regulation of FLT3 sensitizes Ba/F3 FLT3-ITD transfectants and Molm-14 cells to treatment with MLN518. (A) Dose-dependent decrease in cell proliferation following treatment with decreasing concentrations of FLT3 siRNA (Ba/F3 FLT3-ITD cells [n = 8] and Molm-14 cells [n = 5]). (B) Treating Ba/F3 FLT3-ITD cells (n = 8) and Molm-14 cells (n = 5) with FLT3 siRNA increases their sensitivity to treatment with the FLT3 inhibitor MLN518. Error bars indicate standard deviation.
The combination of FLT3 siRNA treatment and MLN518 treatment lowered the MLN518 median inhibitory concentration (IC50) for growth inhibition by MLN518 in the Ba/F3 FLT3-ITD cells 3.4-fold (using 1/256 times siRNA) and up to 3.7-fold in the Molm-14 cells (using 1/32 times siRNA) (Figure 2B; Table 1). These data suggest that growth of both cell lines depends on FLT3 kinase activity and protein expression levels. Furthermore, these data suggest that using a combination approach to inhibit FLT3 activation may be more effective than a single method alone. Given that in chronic myelogenous leukemia (CML) resistance to imatinib involves kinase domain mutations and/or overexpression of Bcr-Abl protein,21 combination therapy involving siRNA might be even more relevant in the context of resistance to FLT3 inhibitors.
IC50 values for Ba/F3 FLT3 ITD and Molm-14 cells following treatment with FLT3 siRNA and MLN518
. | Mock . | NS-siRNA . | 1/32 FLT3 . | 1/64 FLT3 . | 1/128 . | 1/256 . |
|---|---|---|---|---|---|---|
| Ba/F3 FLT3 ITD | 53 | 50 | NA | NA | NA | 15 |
| Molm-14 | 260 | 255 | 70 | 75 | 125 | NA |
. | Mock . | NS-siRNA . | 1/32 FLT3 . | 1/64 FLT3 . | 1/128 . | 1/256 . |
|---|---|---|---|---|---|---|
| Ba/F3 FLT3 ITD | 53 | 50 | NA | NA | NA | 15 |
| Molm-14 | 260 | 255 | 70 | 75 | 125 | NA |
NA indicates not available.
Despite the powerful ability of siRNA to induce robust down-regulation, there are obvious obstacles, such as drug delivery, to the use of siRNA to treat leukemia in vivo. However, recent studies have demonstrated that injection of influenza-specific siRNA and Fas-specific siRNA into mice can prevent diseases such as influenza and fulminant hepatitis, respectively.22,23 Additionally, there has been some concern regarding the ability of siRNA to remain functional in the context of mutations. However, the use of pooled siRNA, as used in the present study, consisting of several functional siRNAs targeting distinct sites of the gene of interest, may be able to overcome this potential limitation. Thus, if issues of delivery can be circumvented, siRNA to FLT3 has promise in combination with FLT3 inhibitors for patients with FLT3-ITD–positive AML.
Prepublished online as Blood First Edition Paper, December 7, 2004; DOI 10.1182/blood-2004-07-2758.
Supported by The Howard Hughes Medical Institute and a grant from the Doris Duke Charitable Foundation and VA Merit Review Grant Program. M.W.D. is also a Junior Faculty Scholar of the American Society of Hematology.
An Inside Blood analysis of this article appears in the front of this issue.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.

![Figure 2. RNAi-induced down-regulation of FLT3 sensitizes Ba/F3 FLT3-ITD transfectants and Molm-14 cells to treatment with MLN518. (A) Dose-dependent decrease in cell proliferation following treatment with decreasing concentrations of FLT3 siRNA (Ba/F3 FLT3-ITD cells [n = 8] and Molm-14 cells [n = 5]). (B) Treating Ba/F3 FLT3-ITD cells (n = 8) and Molm-14 cells (n = 5) with FLT3 siRNA increases their sensitivity to treatment with the FLT3 inhibitor MLN518. Error bars indicate standard deviation.](https://ash.silverchair-cdn.com/ash/content_public/journal/blood/105/7/10.1182_blood-2004-07-2758/6/m_zh80070576530002.jpeg?Expires=1769088838&Signature=HHsqdNaa7RImce65QEZdn32vKV3gmyHpG0C50SI5MmAX5zNh6zBMqIUH5davantYwc8XdPs1rPRbNHzL2ff4myhkG-eJT7D6RP1ufQ8m8h3WXPuBwGtxSX4fsGGKa4CeX8L8FX4VSy0ZnmSH6jbg~9hRK6d~NE0EOKD20TOenkifmr3HDGpllyd9X-o1u1R0Mn2QgNlTN4gReHEl2adPM9ZtxVYAwATITWD9wJ2YoQBU279JO-Im1is9t4AJ2SD9cGihxYiCNeIs~TmARIRfRiA1GKSlhv4KA72bk3-8Xo~W~6NYcJgpbpJD6qYoDlHm4lQ5XQM9jhKmaLfLsSdQKQ__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)
This feature is available to Subscribers Only
Sign In or Create an Account Close Modal