Abstract
Telomere length has been linked to disease stage and degree of (pan-)cytopenia in patients with bone marrow failure syndromes. The aim of the current study was to analyze the impact of replicative stress on telomere length in residual glycosylphosphatidylinositol-positive (GPI+) versus GPI– hematopoiesis in patients with paroxysmal nocturnal hemoglobinuria (PNH). Peripheral blood granulocytes from 16 patients and 22 healthy individuals were analyzed. For this purpose, we developed proaerolysin flow-FISH, a novel methodology that combines proaerolysin staining (for GPI expression) with flow-FISH (for telomere length measurement). We found significantly shortened telomeres in GPI– granulocytes (mean ± SE: 6.26 ± 0.27 telomere fluorescence units [TFU]), both compared with their GPI+ counterparts (6.88 ± 0.38 TFU; P = .03) as well as with age-matched healthy individuals (7.73 ± 0.23 TFU; P < .001). Our findings are in support of a selective growth advantage model of PNH assuming that damage to the GPI+ hematopoietic stem-cell (HSC) compartment leads to compensatory hyperproliferation of residual GPI–HSCs.
Introduction
Paroxysmal nocturnal hemoglobinuria (PNH) is an acquired bone marrow failure syndrome originating in hematopoietic stem cells (HSCs)1 carrying a mutation in the X-linked PIG-A gene.2 This gene is responsible for the first step in the synthesis of the glycosylphosphatidylinositol (GPI) anchor, which is needed to attach a family of extracellular proteins to the cell membrane.3 By analogy to the suspected pathogenesis of aplastic anemia (AA), it is assumed that GPI+ stem cells are selectively eliminated by an autoimmune-mediated process. An alternative hypothesis is based on a suspected intrinsic growth advantage of GPI– HSCs and/or a disadvantage of their GPI+ counterparts, although in vitro studies and data from mouse models are still controversial (reviewed in Young et al4 ).
Telomeres consist of repetitive DNA sequences and associated proteins located at the end of linear chromosomes. Due to the “end-replication problem,” telomeres undergo replication-dependent shortening5 until erosion beyond a minimum critical length results in genetic instability and cell cycle arrest.6 The enzyme telomerase is capable of synthesizing telomere repeats de novo onto the ends of chromosomes.7 In contrast to normal somatic cells, up-regulated telomerase expression is found in germ cells, immortal cell lines, and malignant tumors.8 Several studies investigating telomere dynamics in acquired bone marrow failure syndromes have been published recently.9-11
Based on recent data indicating that cytogenetic abnormalities are predominantly found in GPI+ cells from patients with PNH,12 we have attempted to selectively analyze telomere length in GPI+ and GPI– granulocytes from patients with PNH by using newly developed proaerolysin flow–fluorescence in-situ hybridization (FISH).
Study design
Patient population
For this study, 16 anonymized ethylenediaminetetraacetic acid (EDTA) blood samples from patients diagnosed with PNH or AA/PNH syndrome as well as 22 healthy donors were analyzed with consent from the ethics committee of the University of Ulm and of the University of Tübingen. Informed consent for all healthy donors (n = 22) that were included in the study and for the patients who were enrolled at the University of Ulm and Tübingen was obtained per the Declaration of Helsinki. However, in the majority of the patients, blood was sent to the reference diagnostic lab in Ulm for analysis of GPI deficiency. In cases where blood was leftover after the diagnostic procedure, the remaining samples were anonymized and used for both quality control (ie, analysis of GPI expression) and telomere length measurement as part of the proaerolysin flow-FISH protocol. The diagnosis of AA/PNH syndrome was confirmed by the Institute for Clinical Transfusion Medicine and Immunogenetics (University of Ulm, Germany) using CD24/CD66b and fluorescent proaerolysin variant (FLAER)/CD16 antibodies.
Proaerolysin and multicolor flow-FISH
The newly developed methodology is based on previously published flow-FISH protocols,13,14 with the following modifications. For multicolor flow-FISH, aliquots of 2 × 106 peripheral blood leukocytes (PBLs) and 1 × 106 cow thymocytes were stained with biotinylated immunoglobulin G1 (IgG1), CD45, or CD59 (all from Becton Dickinson, San Jose, CA) at room temperature (RT) for 15 minutes. The cells were washed twice with phosphate-buffered saline (PBS) followed by an incubation with streptavidin–cyanin 5 (Cy5) for 15 minutes at RT. Alternatively, the cells were directly incubated with Alexa-647–conjugated proaerolysin (Protox Biotech, Victoria, BC) under the same conditions. Proaerolysin represents the nontoxic GPI-binding derivative of aerolysin, a toxin that binds to the GPI anchor and forms channels in the cell membrane.15 After washing, antibodies or proaerolysin were cross-linked to the cell surface with the water-soluble cross-linker Bis(sulfosuccinimidyl)subberate (BS3; Pierce, Rockford, IL) at 1 mM for 30 minutes at 4°C according to Batliwalla et al.16
Analysis of proaerolysin flow-FISH
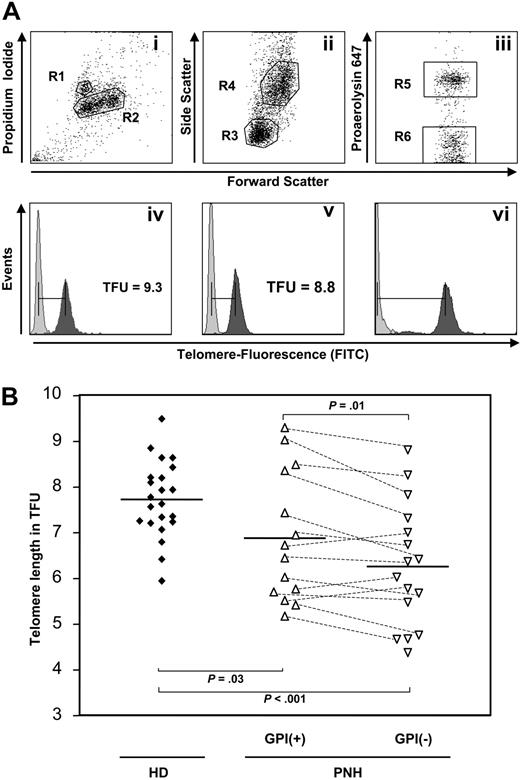
Telomere fluorescence was expressed in telomere fluorescence units (TFU, 1 TFU = 1 kb) based on Southern blot analysis of cow thymocytes (19.52 kb) used as internal controls (Figure 1Avi).17 Cow cells can be distinguished from human diploid cells based on propidium iodide (PI) staining (R1, Figure 1Ai) and telomere length (Figure 1Avi). Proaerolysin-positive (R5 in Figure 1Aiii-iv) and -negative (R6 in Figure 1Aiii,Av) granulocytes (R4, Figure 1Aii) were identified within diploid cells (R2, Figure 1Ai) based on size and granularity. Analysis was performed with (dark gray peaks in Figure 1Aiv-Avi) and without (light gray peaks) fluorescein isothiocyanate (FITC)–labeled peptide nucleic acid (PNA) probes in order to allow subtraction of autofluorescence.
Proaerolysin flow-FISH. (A) Cow cells (R1 in Ai,Avi) can be distinguished from human diploid cells (R2 in Ai) based on PI staining. Lymphocytes (R3 in Aii) and granulocytes (R4 in Aii) were discriminated within diploid cells (R2 in Ai) based on size and granularity (Aii). Granulocytes can be separated into proaerolysin-positive and -negative cells (R5 and R6, respectively, in Aiii). This analysis was performed with (dark-gray peaks in Aiv-vi) and without (light-gray peaks in Aiv-vi) FITC-labeled PNA probes in order to allow subtraction of autofluorescence. (B) Scatter plots of the telomere length measurements of 14 GPI+ and 16 GPI– granulocyte populations determined by proaerolysin flow-FISH in 16 patients with PNH and 22 age-matched healthy donors (HD). The horizontal lines represent the mean values of each group.
Proaerolysin flow-FISH. (A) Cow cells (R1 in Ai,Avi) can be distinguished from human diploid cells (R2 in Ai) based on PI staining. Lymphocytes (R3 in Aii) and granulocytes (R4 in Aii) were discriminated within diploid cells (R2 in Ai) based on size and granularity (Aii). Granulocytes can be separated into proaerolysin-positive and -negative cells (R5 and R6, respectively, in Aiii). This analysis was performed with (dark-gray peaks in Aiv-vi) and without (light-gray peaks in Aiv-vi) FITC-labeled PNA probes in order to allow subtraction of autofluorescence. (B) Scatter plots of the telomere length measurements of 14 GPI+ and 16 GPI– granulocyte populations determined by proaerolysin flow-FISH in 16 patients with PNH and 22 age-matched healthy donors (HD). The horizontal lines represent the mean values of each group.
Statistics
Data were expressed as mean plus or minus SE and given in TFU. Means were compared by the 2-sample t test or by the paired t test, as appropriate. Associations were assessed by Pearson correlation coefficient. Significance was accepted for P less than .05. If not stated otherwise, all reported values represent at least 2 independent measurements. For statistical analysis, JMP Software version 5.1 (SAS Institute, Cary, NJ) was used.
Results and discussion
Telomere length in nucleated peripheral blood (PB) cells indirectly reflects the mitotic history of their precursors, the HSCs.13 Previously, we found significantly shortened telomeres in granulocytes from patients with AA.11 An inverse correlation between age-adjusted telomere length and PB counts was observed, in support of a model according to which the degree of cytopenia and the amount of telomere shortening are positively correlated.11
Dramatically reduced telomere length has recently been described in patients with dyskeratosis congenita (DKC),18 a progressive bone marrow failure syndrome characterized by abnormal skin pigmentation, leukoplakia, and nail dystrophy. Interestingly, the X-linked form of the disease is due to mutations in the gene DKC1. The affected protein, dyskerin, is a nucleolar protein that is associated with human telomerase RNA (hTERC).19 Furthermore, in patients with autosomal dominant DKC, both deletions as well as mutations involving the hTERC have recently been described.20 These mutations can also (although rarely) be found in patients with otherwise clinically typical bone marrow failure syndromes.21,22 Furthermore, just recently Keith et al23 reported a mutation in a functional Sp1 binding site of the hTERC promoter that could be responsible for dysregulated telomerase activity in a PNH patient.
In a previous study, Karadimitris et al,9 using Southern blotting, reported significantly shortened telomere length in granulocytes from 10 patients with PNH and found no tendency toward either GPI+ or GPI– cells. In the current study, we developed a novel methodology called proaerolysin flow-FISH, which allows the selective measurement of telomere length in GPI+ versus GPI– cells from patients with PNH by combining the flow-FISH methodology with GPI staining using proaerolysin24 coupled to the heat-stable fluorochrome Alexa-647 (Figure 1A). PNH clone size determined by proaerolysin flow-FISH was highly positively correlated with the results from clinical diagnostic flow cytometric staining of native cells using antibodies against CD24/CD66b and FLAER/CD16 as well as with multicolor flow-FISH using CD59 staining (r > 0.92; P < .001; Table 1).
Age and clone size of 16 patients with PNH
. | . | GPI- granulocytes, % . | . | . | . | |||
|---|---|---|---|---|---|---|---|---|
| Patient no. . | . | Clinical routine assessment . | . | Multicolor flow-FISH . | . | |||
| . | Age, y . | CD24/CD66b . | FLAER/CD16 . | CD59 . | Proaerolysin . | |||
| 1 | 13 | 31.6 | 31.4 | 4.2 | 42.0 | |||
| 2 | 31 | 98.5 | 93.2 | 98.8 | 97.8 | |||
| 3 | 33 | ND | ND | 90.9 | 95.8 | |||
| 4 | 42 | 26.7 | 54.2 | ND | 55.8 | |||
| 5 | 26 | 61.2 | 59.7 | 53.2 | 52.7 | |||
| 6 | 74 | 13.5 | 6.5 | 19.4 | 18.8 | |||
| 7 | 65 | 74.2 | 78.8 | 76.7 | 79.9 | |||
| 8 | 48 | 76.3 | 75.7 | ND | 68.4 | |||
| 9 | 18 | 65.7 | 83.6 | 69.8 | 76.4 | |||
| 10 | 16 | 12.2 | 14.4 | 13.5 | 14.9 | |||
| 11 | 16 | 74.3 | 76.5 | 46.3 | 37.6 | |||
| 12 | 46 | 98.1 | 99.5 | 90.0 | 92.8 | |||
| 13 | 40 | 99.5 | 99.4 | 99.2 | 97.9 | |||
| 14 | 34 | 99.9 | 98.6 | 99.1 | 99.3 | |||
| 15 | 36 | 61.0 | 65.4 | 71.0 | 76.6 | |||
| 16 | 19 | 98.7 | 97.2 | 96.6 | 97.6 | |||
. | . | GPI- granulocytes, % . | . | . | . | |||
|---|---|---|---|---|---|---|---|---|
| Patient no. . | . | Clinical routine assessment . | . | Multicolor flow-FISH . | . | |||
| . | Age, y . | CD24/CD66b . | FLAER/CD16 . | CD59 . | Proaerolysin . | |||
| 1 | 13 | 31.6 | 31.4 | 4.2 | 42.0 | |||
| 2 | 31 | 98.5 | 93.2 | 98.8 | 97.8 | |||
| 3 | 33 | ND | ND | 90.9 | 95.8 | |||
| 4 | 42 | 26.7 | 54.2 | ND | 55.8 | |||
| 5 | 26 | 61.2 | 59.7 | 53.2 | 52.7 | |||
| 6 | 74 | 13.5 | 6.5 | 19.4 | 18.8 | |||
| 7 | 65 | 74.2 | 78.8 | 76.7 | 79.9 | |||
| 8 | 48 | 76.3 | 75.7 | ND | 68.4 | |||
| 9 | 18 | 65.7 | 83.6 | 69.8 | 76.4 | |||
| 10 | 16 | 12.2 | 14.4 | 13.5 | 14.9 | |||
| 11 | 16 | 74.3 | 76.5 | 46.3 | 37.6 | |||
| 12 | 46 | 98.1 | 99.5 | 90.0 | 92.8 | |||
| 13 | 40 | 99.5 | 99.4 | 99.2 | 97.9 | |||
| 14 | 34 | 99.9 | 98.6 | 99.1 | 99.3 | |||
| 15 | 36 | 61.0 | 65.4 | 71.0 | 76.6 | |||
| 16 | 19 | 98.7 | 97.2 | 96.6 | 97.6 | |||
ND indicates not done.
Using the proaerolysin flow-FISH methodology, in 11 of 14 patients with paired samples telomeres of GPI–, granulocytes (mean ± SE: 6.51 ± 0.33 TFU) were found to be shorter than their GPI+ counterparts (6.88 ± 0.38 TFU, P = .013). Interestingly, both GPI– as well as GPI+ cells from PNH patients showed significant telomere shortening compared with age-matched GPI+ healthy individuals (7.73 ± 0.23 TFU, P = .001 and P = .029, respectively). Within the PNH patient population, the telomere length difference between GPI+ and GPI– cells decreases linearly with age (r =–0.80, P = .001). These data support the growth advantage hypothesis, which argues that a depletion of the GPI+ HSC compartment is (at least partly) compensated by hyperproliferation of residual GPI– cells. Alternatively, accelerated replication-independent telomere shortening could account for this phenomenon, for example, due to increased sensitivity to oxidative stress or altered expression of telomerase/telomere-binding proteins. If secondary clonal disorders were linked to progressive telomere shortening in PNH, based on these data, one would expect karyotypic abnormalities to occur predominantly in GPI– cells. In a recent report, however, Sloand et al12 found that within a subgroup of 5 patients with AA/PNH before the development of cytogenetic abnormalities, all had aberrant karyotypes in the GPI+ population but only 3 exhibited these changes in GPI– cells, although in one case at a higher frequency. At first glance, these data argue against replicative telomere shortening being the major cause of secondary karyotypic abnormalities in PNH patients. However, one could speculate that failure to up-regulate telomerase activity upon telomere shortening (eg, due to hTERC promoter mutations23 ) could lead to replicative exhaustion and loss of genetically (and karyotypically) instable PNH clones with short telomeres rather than outgrowth of these clones. In support of this hypothesis, Araten et al25 demonstrated that on follow-up, the proportion of cells with abnormal karyotypes decreased significantly in 7 of 11 patients with PNH. None of the patients developed excess blasts or leukemia, suggesting that cytogenetically abnormal cells may potentially undergo replicative exhaustion and that these cells neither necessarily are nor become malignant during the course of disease. Use of this novel methodology will allow us to address the role of telomere biology for this process in future prospective studies including sequential measurements in individual patients.
Prepublished online as Blood First Edition Paper, April 5, 2005; DOI 10.1182/blood-2004-10-3996.
Supported by the Sonderforschungsbereich 510 (Teilprojekt A6) of the Deutsche Forschungsgemeinschaft (DFG).
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.
We would like to thank Alexandra Wahl, Iris Schäfer, and Bettina Kirchner for excellent technical assistance.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal