Abstract
Patients with myeloma, treated on the thalidomide arm of total therapy 2 (TT2), had a higher complete response (CR) rate and improved event-free survival (EFS) but not overall survival (OS). To evaluate the benefit of TT2's posttandem autotransplant consolidation chemotherapy and dexamethasone maintenance, outcomes were compared on TT2 without thalidomide (n = 345; median follow-up, 3.5 years) and on predecessor trial TT1 (n = 231; median follow-up, 11.5 years). CR rates were similar (43% vs 41%); however, 5-year estimates of continuous CR (45% vs 32%, P < .001) and 5-year EFS (43% vs 28%, P < .001) were superior with TT2, with a trend for improved OS (62% vs 57%; P = .11). OS was also superior among patients achieving CR and receiving the second transplantation early after the first transplantation. Superior EFS and OS with TT2 versus TT1 was noted in the two thirds presenting without cytogenetic abnormalities (CAs); 4-year posttandem transplantation OS for patients with CAs was 47% with TT1 and 76% with TT2 when combination chemotherapy rather than DEX was applied for consolidation (P = .040). Thus, TT2 (without thalidomide) improved OS of patients without CAs; those with CAs benefited from posttransplantation consolidation chemotherapy. The favorable effects of CR and rapidly sequenced second transplantation attest to the validity of a melphalan dose-response effect in myeloma.
Introduction
High-dose melphalan (MEL) has become the mainstay of therapy for patients with multiple myeloma (MM), especially for those under the age of 65 years.1,2 Complete response (CR) rates up to 40% have been reported with tandem transplantations, doubling 7-year overall survival (OS) and event-free survival (EFS) rates in comparison to single transplantations.3 Total therapy 2 (TT2)4 evaluated, in a randomized trial design, whether EFS and OS could be extended by the addition of thalidomide,5 the first new active agent for MM since the introduction of melphalan and prednisone in the early 1960s.6 Although raising CR and prolonging EFS, thalidomide failed to improve OS due to a higher failure rate to salvage therapies among those initially randomized to thalidomide.7 In comparison to total therapy 1 (TT1),8,9 TT2 applied more intensive chemotherapy for induction prior to and for consolidation after tandem transplantations as well as high-dose dexamethasone (DEX) during the first year of maintenance with interferon. We now report on the clinical outcome of 345 patients treated with TT2 without thalidomide in comparison with 231 patients receiving TT1.
Patients, materials, and methods
Eligibility criteria
For both trials, patients had to have an established diagnosis of symptomatic or progressive MM that required treatment. Up to one cycle of prior systemic therapy was permitted; local radiation for painful bone fractures was also allowed. Cardiopulmonary and hepatic functions had to be preserved. Renal failure, even if hemodialysis-dependent, was not an exclusion criterion if its duration did not exceed 6 months and was likely due to cast nephropathy, as suggested by Bence Jones proteinuria. The upper age limit was 70 years with TT1 and 75 years with TT2. In keeping with institutional and federal policies, all patients had to provide written informed consent, indicating the investigational nature of the trials proposed. Approval for these studies was obtained from the institutional review board of the University of Arkansas for Medical Sciences (UAMS). Response and toxicity data were monitored, on both trials, by independent auditors.
Response and relapse criteria
CR required the absence of M-protein in serum and urine on immunofixation analysis as well as normal bone marrow aspirate and biopsy by microscopy and flow cytometry (absence of aneuploidy and monoclonal cytoplasmic light chain); in addition, metaphase cytogenetic abnormalities (CAs) had to be absent.9 Partial response (PR) required at least 75% reduction in serum M-protein and at least 90% reduction in urinary M-protein excretion, along with normal bone marrow findings (as in CR). Response criteria had to be documented on at least 2 subsequent occasions at least 2 months apart. Relapse from CR implied the reappearance of M-protein in serum or urine, reappearance of monoclonal bone marrow plasmacytosis or of CAs, the development extramedullary disease or of new bone lesions recognized on periodically (at least semiannually) performed magnetic resonance imaging or skeletal survey. Relapse from PR was diagnosed when M-protein levels/excretion doubled from the lowest level or increased by 50%, whichever occurred earlier; similar consideration as with relapse from CR applied to development of new medullary or extramedullary lesions and bone marrow findings.
Treatment regimens
The details of both TT1 and TT2 regimens have been reported previously and are briefly summarized in Table 1.4,8 Instead of induction therapy in TT1 with 3 cycles of VAD (4-day continuous daily infusions of vincristine and doxorubicin plus oral high-dose dexamethasone [DEX] pulsing), high-dose cyclophosphamide with filgrastim for peripheral-blood stem-cell (PBSC) mobilization and EDAP (etoposide, DEX, arabinosyl cytosine, cis-platin), TT2 employed only one cycle of VAD followed by DCEP (DEX, cyclophosphamide, etoposide, cisplatin), CAD (cyclophosphamide, doxorubicin, DEX) with PBSC collection, and further DCEP. Both protocols employed tandem autotransplantations in support of MEL 200 mg/m2 (MEL 200), which were spaced 2 to 6 months apart. With TT2, patients aged 70 years or more and those with creatinine levels exceeding 265.2 mM/L (3 mg/dL) received a reduced MEL dose of 140 mg/m2. In case patients had achieved less than PR status prior to the second transplantation, TT1 called for MEL 140 with added total body irradiation; patients on TT2 received the carmustine, etoposide, cytarabine, and melphalan (BEAM) regimen (Table 1). TT2 also introduced consolidation chemotherapy, initially with DCEP every 3 months for 4 cycles versus DCEP alternating with CAD every 6 weeks for a total of 8 cycles; because of similar results from these 2 consolidation arms, the last 90 patients were offered D-PACE (DEX, cis-platin, doxorubicin, cyclophosphamide, etoposide). In case platelet recovery to at least 100 000/μL failed or DCEP was ineffective during induction (< 25% further decrease in M-protein after VAD), DEX pulsing (40 mg/d for 4 days) was applied monthly as an alternative consolidation strategy. DEX pulsing (40 mg on days 1-4, 9-12, 17-20) was added quarterly during the first year of interferon maintenance therapy in TT2.
Treatment details of total therapy 1 and total therapy 2
Regimen . | Regimen details . |
|---|---|
| Total Therapy 1 | |
| Induction phase | |
| Cycles 1-3, VAD | Vincristine (0.5 mg/d × 4 d) CI; Adriamycin (10 mg/m2/d × 4 d) CI; Dexamethasone 40 mg PO d 1-4, 9-12, 17-20 |
| Cycle 4, HD-CTX | Cyclophosphamide (6 g/m2 in 5 divided doses, 1.2 g/m2 every 3 h) + Mesna (3.6 g/m2); GM-CSF 250 μg/m2/d → PBSC collection |
| Cycle 5, EDAP | Etoposide (100 mg/m2/d × 4 d) CI; cis-Platin (25 mg/m2/d × 4 d)Arabinosyl-cytosine (1 g/m2 d 5); Dexamethasone 40 mg d 1-5; GM-CSF 250 μg/m2/d |
| Transplantation phase | |
| First transplantation | Melphalan (100 mg/m2 on d -3 and -2); PBSC d 0; GM-CSF 250 μg/m2/d |
| Second transplantation | Melphalan (100 mg/m2 on d -3 and -2); PBSC d 0; GM-CSF 250 μg/m2/d; (In case of < PR: melphalan 140 mg/m2 plus total body irradiation 8.5 Gy in 5 equal fractions of 1.7 Gy every 12 h) |
| Maintenance phase | |
| Interferon | 3 million U/m2 SQ 3 × wk until relapse |
| Total therapy 2 | |
| Induction | |
| Cycle 1, VAD | Vincristine (0.5 mg/d × 4 d) CI; Adriamycin (10 mg/m2/d × 4 d) CI; Dexamethasone 40 mg PO d 1-4, 9-12, 17-20 |
| Cycle 2, DCEP | Cyclophosphamide (400 mg/m2/day × 4 d) CI; Etoposide (40 mg/m2/day × 4 d) CI; cis-Platin (10 mg/m2/day × 4 d) CI; Dexamethasone (40 mg/d) PO days 1-4 |
| Cycle 3, CAD | Cyclophosphamide (750 mg/m2/day × 4 d) CI; Adriamycin (15 mg/m2/d × 4 d) CI; G-CSF 10 μg/kg SQ twice daily → PBSC collection |
| Cycle 4, DCEP | Cyclophosphamide (400 mg/m2/day × 4 d) CI; Etoposide (40 mg/m2/day × 4 d) CI; cis-Platin (10 mg/m2/day × 4 d) CI; Dexamethasone (40 mg/d) PO days 1-4 |
| Transplantation | |
| First transplantation | Melphalan 200 mg/m2 d -1 (reduce to 140 mg/m2 for age >70 years or creatinine >3 mg/dL) |
| Second transplantation | Melphalan 200 mg/m2 d -1 (reduced to 140 mg/m2 for age >70 years or creatinine >3 mg/dL) < PR: BCNU 300 mg/m2 d -5; Etoposide 200 mg/m2; Arabinosyl cytosine 400 mg/m2 d -5 through -2; Melphalan 140 mg/m2 d -2 |
| Consolidation | |
| Randomized arm A: DCEP | Dexamethasone 40 mg/d PO d 1-4; cyclophosphamide (300 mg/m2/d × 4 d) CI; etoposide (30 mg/m2/d × 4 d) CI; cis-platin (7.5 mg/m2/d × 4 d) CI every 3 months for 4 cycles |
| Randomized arm B | |
| DCEP* | Dexamethasone 40 mg/d PO d 1-4; cyclophosphamide (300 mg/m2/d × 4 d) CI; etoposide (30 mg/m2/d × 4 d) CI; cis-platin (75 mg/m2/d × 4 d) |
| CAD* | Cyclophosphamide (400 mg/m2/d × 4 d) CI; adriamycin (10 mg/m2/d × 4 d) CI; dexamethasone 40 mg/d PO d 1-4 |
| DPACE | Dexamethasone 40 mg/d PO d 1-4; cis-platin (75 mg/m2/d × 4 d) CI; adriamycin (10 mg/m2/d × 4 d) CI; cyclophosphamide (300 mg/m2/d × 4 d) CI; etoposide (30 mg/m2/d × 4 d) CI; every 3 months for 4 cycles. |
| DEX | In case of failure to recover platelets to at least 100 000/μL or failure to achieve >25% M-protein reduction with first cycle of DCEP in induction: DEX 40 mg d 1-4 every 28 d for 1 year. |
| Maintenance | |
| Interferon | 3 million U/m2 SQ 3 × wk plus first year of maintenance: Dexamethasone 40 mg every 3 months on d 1-4, 9-12, 17-20 |
| Second year onward; interferon alone |
Regimen . | Regimen details . |
|---|---|
| Total Therapy 1 | |
| Induction phase | |
| Cycles 1-3, VAD | Vincristine (0.5 mg/d × 4 d) CI; Adriamycin (10 mg/m2/d × 4 d) CI; Dexamethasone 40 mg PO d 1-4, 9-12, 17-20 |
| Cycle 4, HD-CTX | Cyclophosphamide (6 g/m2 in 5 divided doses, 1.2 g/m2 every 3 h) + Mesna (3.6 g/m2); GM-CSF 250 μg/m2/d → PBSC collection |
| Cycle 5, EDAP | Etoposide (100 mg/m2/d × 4 d) CI; cis-Platin (25 mg/m2/d × 4 d)Arabinosyl-cytosine (1 g/m2 d 5); Dexamethasone 40 mg d 1-5; GM-CSF 250 μg/m2/d |
| Transplantation phase | |
| First transplantation | Melphalan (100 mg/m2 on d -3 and -2); PBSC d 0; GM-CSF 250 μg/m2/d |
| Second transplantation | Melphalan (100 mg/m2 on d -3 and -2); PBSC d 0; GM-CSF 250 μg/m2/d; (In case of < PR: melphalan 140 mg/m2 plus total body irradiation 8.5 Gy in 5 equal fractions of 1.7 Gy every 12 h) |
| Maintenance phase | |
| Interferon | 3 million U/m2 SQ 3 × wk until relapse |
| Total therapy 2 | |
| Induction | |
| Cycle 1, VAD | Vincristine (0.5 mg/d × 4 d) CI; Adriamycin (10 mg/m2/d × 4 d) CI; Dexamethasone 40 mg PO d 1-4, 9-12, 17-20 |
| Cycle 2, DCEP | Cyclophosphamide (400 mg/m2/day × 4 d) CI; Etoposide (40 mg/m2/day × 4 d) CI; cis-Platin (10 mg/m2/day × 4 d) CI; Dexamethasone (40 mg/d) PO days 1-4 |
| Cycle 3, CAD | Cyclophosphamide (750 mg/m2/day × 4 d) CI; Adriamycin (15 mg/m2/d × 4 d) CI; G-CSF 10 μg/kg SQ twice daily → PBSC collection |
| Cycle 4, DCEP | Cyclophosphamide (400 mg/m2/day × 4 d) CI; Etoposide (40 mg/m2/day × 4 d) CI; cis-Platin (10 mg/m2/day × 4 d) CI; Dexamethasone (40 mg/d) PO days 1-4 |
| Transplantation | |
| First transplantation | Melphalan 200 mg/m2 d -1 (reduce to 140 mg/m2 for age >70 years or creatinine >3 mg/dL) |
| Second transplantation | Melphalan 200 mg/m2 d -1 (reduced to 140 mg/m2 for age >70 years or creatinine >3 mg/dL) < PR: BCNU 300 mg/m2 d -5; Etoposide 200 mg/m2; Arabinosyl cytosine 400 mg/m2 d -5 through -2; Melphalan 140 mg/m2 d -2 |
| Consolidation | |
| Randomized arm A: DCEP | Dexamethasone 40 mg/d PO d 1-4; cyclophosphamide (300 mg/m2/d × 4 d) CI; etoposide (30 mg/m2/d × 4 d) CI; cis-platin (7.5 mg/m2/d × 4 d) CI every 3 months for 4 cycles |
| Randomized arm B | |
| DCEP* | Dexamethasone 40 mg/d PO d 1-4; cyclophosphamide (300 mg/m2/d × 4 d) CI; etoposide (30 mg/m2/d × 4 d) CI; cis-platin (75 mg/m2/d × 4 d) |
| CAD* | Cyclophosphamide (400 mg/m2/d × 4 d) CI; adriamycin (10 mg/m2/d × 4 d) CI; dexamethasone 40 mg/d PO d 1-4 |
| DPACE | Dexamethasone 40 mg/d PO d 1-4; cis-platin (75 mg/m2/d × 4 d) CI; adriamycin (10 mg/m2/d × 4 d) CI; cyclophosphamide (300 mg/m2/d × 4 d) CI; etoposide (30 mg/m2/d × 4 d) CI; every 3 months for 4 cycles. |
| DEX | In case of failure to recover platelets to at least 100 000/μL or failure to achieve >25% M-protein reduction with first cycle of DCEP in induction: DEX 40 mg d 1-4 every 28 d for 1 year. |
| Maintenance | |
| Interferon | 3 million U/m2 SQ 3 × wk plus first year of maintenance: Dexamethasone 40 mg every 3 months on d 1-4, 9-12, 17-20 |
| Second year onward; interferon alone |
CI indicates continuous infusion; SQ, subcutaneously.
†There was a protocol modification after entry of 121 patients (66 DCEP, 55 DCEP/CAD); see DPACE and DEX under total therapy 2.
In randomized arm B, DCEP was alternated with CAD every 6 weeks for 8 cycles.
Statistical analyses
EFS and OS comparisons between TT1 and TT2 were performed using an intent-to-treat approach. Comparisons of clinical response required the administration of at least one cycle of protocol induction therapy. EFS and OS were estimated by the Kaplan-Meier method; survival differences were examined by the log-rank test.10,11 The timing of treatment-related mortality (TRM) and CR were visualized using cumulative incidence;12 frequency differences in TRM and CR were compared using the chi-square test. Chi-square or Fisher exact tests were used to compare cross tabulations of categorical variables; the Wilcoxon 2-sample test compared the distribution of continuous factors between TT1 and TT2.13 Cox regression was used to evaluate multivariate prognostic models of EFS and OS.14 Where appropriate, continuous prognostic factors were used for multivariate models of EFS and OS with the following notable exceptions: nonparametric plots of log relative risk indicated that the relationship between EFS/OS and lactate dehydrogenase (LDH) was best modeled using a threshold at the upper limit of normal; serum levels of beta-2-microglobulin (B2M), C-reactive protein (CRP), and creatinine were log-linear with respect to the log relative risk.15,16 Logistic regression evaluated multivariate prognostic models of TRM and CR. Each prognostic factor was modeled as either a continuous covariate or dichotomized at an appropriate threshold based on a visual examination of the relationship between the log odds of CR or TRM and the prognostic factor of interest using a cubic spline.17 Variables were selected for model inclusion in Tables 4 and 5 using a best subset algorithm (models rated using a score chi-square statistic both for Cox and logistic regression) subject to the restriction that the Wald test statistic for each variable was significant at the conventional .05 level.
Factors independently associated with complete remission and treatment-related mortality
Model . | Dependent variables . | Independent variables . | OR (95% CI)* . | P . |
|---|---|---|---|---|
| IgA + LDH + CAs | Complete remission | IgA isotype | 1.69 (1.11-2.57) | .015 |
| LDH above upper limit of normal | 1.89 (1.26-2.82) | .002 | ||
| No cytogenetic abnormalities | 0.56 (0.38-0.81) | .002 | ||
| Albumin + Age | Treatment-related mortality | Albumin < 30 g/L (3.0 g/dL) | 2.80 (1.14-6.86) | .024 |
| Age ≥ 60 y | 2.60 (1.35-5.03) | .005 |
Model . | Dependent variables . | Independent variables . | OR (95% CI)* . | P . |
|---|---|---|---|---|
| IgA + LDH + CAs | Complete remission | IgA isotype | 1.69 (1.11-2.57) | .015 |
| LDH above upper limit of normal | 1.89 (1.26-2.82) | .002 | ||
| No cytogenetic abnormalities | 0.56 (0.38-0.81) | .002 | ||
| Albumin + Age | Treatment-related mortality | Albumin < 30 g/L (3.0 g/dL) | 2.80 (1.14-6.86) | .024 |
| Age ≥ 60 y | 2.60 (1.35-5.03) | .005 |
Odds ratio (OR) and 95% confidence interval (CI) from multivariate logistic regression.
Multivariate model of prognostic factors of EFS and OS in TT1, TT2, and TT1/TT2 combined
. | EFS, HR (95% CI)/P . | . | . | OS, HR (95% CI)/P . | . | . | ||||
|---|---|---|---|---|---|---|---|---|---|---|
| Variable . | TT1 . | TT2 . | TT1 + TT2 . | TT1 . | TT2 . | TT1 + TT2 . | ||||
| Enrolled to TT2 | NA | NA | 0.68 (0.54-0.84)/<.001 | NA | NA | 0.79 (0.60-1.04)/.093 | ||||
| IgA | NS | 1.72 (1.19-2.47)/.004 | 1.31 (1.02-1.69)/.036 | NS | 1.84 (1.16-2.92)/.010 | 1.53 (1.14-2.06)/.004 | ||||
| B2M, natural log of mg/L | 13.0 (10.4-16.3)/.022 | NS | 12.6 (10.7-14.8)/.005 | NS | NS | NS | ||||
| Albumin, g/L | NS | 6.5 (5.3-7.9)/<.001 | 7.9 (6.8-9.3)/.003 | NS | 6.0 (4.7-7.5)/<.001 | 7.3 (6.2-8.7)/<.001 | ||||
| Cytogenetic abnormalities | 1.38 (1.02-1.86)/.034 | 1.84 (1.34-2.54)/<.001 | 1.52 (1.22-1.90)/<.001 | 1.59 (1.15-2.21)/.005 | 2.32 (1.56-3.46)/<.001 | 1.84 (1.44-2.37)/<.001 | ||||
| LDH above upper limit of normal | 1.43 (1.02-2.02)/.039 | 1.44 (1.02-2.03)/.040 | 1.37 (1.07-1.75)/.011 | 1.46 (1.02-2.10)/.041 | 1.51 (0.98-2.32)/.063 | 1.57 (1.19-2.06)/.001 | ||||
| Age, decades | NS | NS | NS | NS | 1.36 (1.10-1.69) /.005 | 1.16 (1.02-1.32)/.026 | ||||
| Hemoglobin, g/L | NS | NS | NS | 9.3 (8.6-9.9)/.023 | NS | NS | ||||
| Creatinine, | ||||||||||
| natural log of | ||||||||||
| μM/L | NS | NS | NS | 141.4 (99.0-201.6)/.009 | NS | NS | ||||
. | EFS, HR (95% CI)/P . | . | . | OS, HR (95% CI)/P . | . | . | ||||
|---|---|---|---|---|---|---|---|---|---|---|
| Variable . | TT1 . | TT2 . | TT1 + TT2 . | TT1 . | TT2 . | TT1 + TT2 . | ||||
| Enrolled to TT2 | NA | NA | 0.68 (0.54-0.84)/<.001 | NA | NA | 0.79 (0.60-1.04)/.093 | ||||
| IgA | NS | 1.72 (1.19-2.47)/.004 | 1.31 (1.02-1.69)/.036 | NS | 1.84 (1.16-2.92)/.010 | 1.53 (1.14-2.06)/.004 | ||||
| B2M, natural log of mg/L | 13.0 (10.4-16.3)/.022 | NS | 12.6 (10.7-14.8)/.005 | NS | NS | NS | ||||
| Albumin, g/L | NS | 6.5 (5.3-7.9)/<.001 | 7.9 (6.8-9.3)/.003 | NS | 6.0 (4.7-7.5)/<.001 | 7.3 (6.2-8.7)/<.001 | ||||
| Cytogenetic abnormalities | 1.38 (1.02-1.86)/.034 | 1.84 (1.34-2.54)/<.001 | 1.52 (1.22-1.90)/<.001 | 1.59 (1.15-2.21)/.005 | 2.32 (1.56-3.46)/<.001 | 1.84 (1.44-2.37)/<.001 | ||||
| LDH above upper limit of normal | 1.43 (1.02-2.02)/.039 | 1.44 (1.02-2.03)/.040 | 1.37 (1.07-1.75)/.011 | 1.46 (1.02-2.10)/.041 | 1.51 (0.98-2.32)/.063 | 1.57 (1.19-2.06)/.001 | ||||
| Age, decades | NS | NS | NS | NS | 1.36 (1.10-1.69) /.005 | 1.16 (1.02-1.32)/.026 | ||||
| Hemoglobin, g/L | NS | NS | NS | 9.3 (8.6-9.9)/.023 | NS | NS | ||||
| Creatinine, | ||||||||||
| natural log of | ||||||||||
| μM/L | NS | NS | NS | 141.4 (99.0-201.6)/.009 | NS | NS | ||||
Continuous prognostic factors used when appropriate.
NA indicates not applicable; NS, not significant.
Results
Patient characteristics
TT1 and TT2 baseline characteristics were comparable with regard to most prognostic features, especially the presence of CAs (Table 2). TT2 comprised a higher proportion of patients aged 65 years and older; fewer TT2 patients were anemic and hypoalbuminemic or had been exposed to one cycle of prior therapy.
Patient characteristics and prestudy prognostic factors by study
Factor . | TT1, no./N (%) . | TT2 No THAL, no./N (%) . | P . |
|---|---|---|---|
| Median age, y | 51.0 | 56.6 | <.001* |
| Age at least 65 y | 21/231 (9) | 72/345 (21) | <.001 |
| Age at least 70 y | 2/231 (1) | 26/345 (8) | <.001 |
| White | 206/231 (89) | 308/345 (89) | .97 |
| IgA isotype | 41/231 (18) | 76/345 (22) | .21 |
| Female | 88/231 (38) | 135/345 (39) | .80 |
| CRP ≥ 8 mg/L | 70/223 (31) | 127/339 (37) | .14 |
| B2M ≥ 3.5 mg/L | 95/229 (41) | 126/345 (37) | .23 |
| Bone marrow plasma cells > 30% | 135/231 (58) | 170/291 (58) | .99 |
| Albumin < 35 g/L (3.5 g/dL) | 62/231 (27) | 59/343 (17) | .005 |
| Hemoglobin < 100 g/L (10 g/dL) | 78/231 (34) | 80/345 (23) | .005 |
| Creatinine ≥ 176.8 μM/L (2 mg/dL) | 22/231 (10) | 37/339 (11) | .59 |
| LDH above upper limit of normal | 49/230 (21) | 86/344 (25) | .31 |
| Cytogenetic abnormalities (CAs) | 74/231 (32) | 104/339 (31) | .73 |
| ISS Stage 1 | 110/229 (48) | 195/343 (57) | .070 |
| Stage 2 | 74/229 (32) | 83/343 (24) | — |
| Stage 3 | 45/229 (20) | 65/343 (19) | — |
| One cycle of prior therapy | 76/231 (33) | 88/345 (26) | .054 |
| Completed Tx1 | 195/231 (84) | 292/345 (85) | .94 |
| Completed Tx2 | 165/231 (71) | 235/345 (68) | .40 |
Factor . | TT1, no./N (%) . | TT2 No THAL, no./N (%) . | P . |
|---|---|---|---|
| Median age, y | 51.0 | 56.6 | <.001* |
| Age at least 65 y | 21/231 (9) | 72/345 (21) | <.001 |
| Age at least 70 y | 2/231 (1) | 26/345 (8) | <.001 |
| White | 206/231 (89) | 308/345 (89) | .97 |
| IgA isotype | 41/231 (18) | 76/345 (22) | .21 |
| Female | 88/231 (38) | 135/345 (39) | .80 |
| CRP ≥ 8 mg/L | 70/223 (31) | 127/339 (37) | .14 |
| B2M ≥ 3.5 mg/L | 95/229 (41) | 126/345 (37) | .23 |
| Bone marrow plasma cells > 30% | 135/231 (58) | 170/291 (58) | .99 |
| Albumin < 35 g/L (3.5 g/dL) | 62/231 (27) | 59/343 (17) | .005 |
| Hemoglobin < 100 g/L (10 g/dL) | 78/231 (34) | 80/345 (23) | .005 |
| Creatinine ≥ 176.8 μM/L (2 mg/dL) | 22/231 (10) | 37/339 (11) | .59 |
| LDH above upper limit of normal | 49/230 (21) | 86/344 (25) | .31 |
| Cytogenetic abnormalities (CAs) | 74/231 (32) | 104/339 (31) | .73 |
| ISS Stage 1 | 110/229 (48) | 195/343 (57) | .070 |
| Stage 2 | 74/229 (32) | 83/343 (24) | — |
| Stage 3 | 45/229 (20) | 65/343 (19) | — |
| One cycle of prior therapy | 76/231 (33) | 88/345 (26) | .054 |
| Completed Tx1 | 195/231 (84) | 292/345 (85) | .94 |
| Completed Tx2 | 165/231 (71) | 235/345 (68) | .40 |
— indicates not applicable.
Wilcoxon 2-sample test, otherwise chi-square test for independence.
Completion of therapy
TT1 enrolled 231 patients (median follow-up, 11 years) and TT2 enrolled 345 patients (median follow-up, 3.5 years). The entire induction phase was completed in 203 (88%) patients receiving TT1 and in 299 (87%) patients enrolled on TT2. Completion rates of first and second transplantations were also similar between the 2 studies: 84% and 71% with TT1 versus 85% and 68% with TT2 (Table 2; Figure 1A). Among the 218 (63%) patients initiating the consolidation phase of TT2, 64% received chemotherapy (DCEP, CAD, or D-PACE) and the remainder DEX (when platelets failed to recover or no benefit was documented from induction DCEP). In the case of TT1 (no consolidation therapy), 55% started interferon (IFN) maintenance compared with 54% with TT2 (P = .72). Table 3 provides an account of all patients proceeding through or coming off serial phases of both treatment regimens.
Patient flow-through protocols
. | . | . | Off-study reason, no. . | . | . | . | . | No. (%) in PR†through treatment phase . | No. (%) in CR‡through treatment phase . | |||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Phase . | No. at start of phase . | Percent enrollees . | Toxicity . | Progression/relapse . | Death (TRM*) . | Other . | Notes . | . | . | |||
| Total therapy 1 | ||||||||||||
| Enrollment | 231 | 100 | 0 | 0 | 0 | 0 | None | NA | NA | |||
| Induction | 231 | 100 | 8 | 6 | 8 (6) | 14 | None | 145 (63) | 27 (12) | |||
| First transplantation | 195 | 84 | 9 | 9 | 2 (2) | 10 | None | 175 (76) | 60 (26) | |||
| Second transplantation | 165 | 71 | 16 | 2 | 6 (3) | 13 | None | 188 (81) | 84 (36) | |||
| Maintenance | 128 | 55 | 0 | 83 | 15 (2) | 21 | None | 189 (82) | 94 (41) | |||
| Total therapy 2, no thalidomide | ||||||||||||
| Enrollment | 345 | 100 | 0 | 0 | 1 | 7 | None | NA | NA | |||
| Induction | 337 | 98 | 3 | 6 | 8 (9§) | 28 | None | 136 (40) | 34 (10) | |||
| First transplantation | 292 | 85 | 2 | 7 | 2 (2) | 24 | 22 skipped second transplantation | 213 (63) | 79 (23) | |||
| Second transplantation | 235 | 68 | 0 | 5 | 8 (9§) | 23 | 3 skipped consolidation | 249 (74) | 109 (32) | |||
| Consolidation | 218 | 63 | 0 | 17 | 0 | 6 | 12 still on consolidation | 262 (78) | 138 (41) | |||
| Maintenance | 186 | 54 | 0 | 39 | 3 (1) | 3 | 141 still on maintenance | 264 (78) | 145 (43) | |||
. | . | . | Off-study reason, no. . | . | . | . | . | No. (%) in PR†through treatment phase . | No. (%) in CR‡through treatment phase . | |||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Phase . | No. at start of phase . | Percent enrollees . | Toxicity . | Progression/relapse . | Death (TRM*) . | Other . | Notes . | . | . | |||
| Total therapy 1 | ||||||||||||
| Enrollment | 231 | 100 | 0 | 0 | 0 | 0 | None | NA | NA | |||
| Induction | 231 | 100 | 8 | 6 | 8 (6) | 14 | None | 145 (63) | 27 (12) | |||
| First transplantation | 195 | 84 | 9 | 9 | 2 (2) | 10 | None | 175 (76) | 60 (26) | |||
| Second transplantation | 165 | 71 | 16 | 2 | 6 (3) | 13 | None | 188 (81) | 84 (36) | |||
| Maintenance | 128 | 55 | 0 | 83 | 15 (2) | 21 | None | 189 (82) | 94 (41) | |||
| Total therapy 2, no thalidomide | ||||||||||||
| Enrollment | 345 | 100 | 0 | 0 | 1 | 7 | None | NA | NA | |||
| Induction | 337 | 98 | 3 | 6 | 8 (9§) | 28 | None | 136 (40) | 34 (10) | |||
| First transplantation | 292 | 85 | 2 | 7 | 2 (2) | 24 | 22 skipped second transplantation | 213 (63) | 79 (23) | |||
| Second transplantation | 235 | 68 | 0 | 5 | 8 (9§) | 23 | 3 skipped consolidation | 249 (74) | 109 (32) | |||
| Consolidation | 218 | 63 | 0 | 17 | 0 | 6 | 12 still on consolidation | 262 (78) | 138 (41) | |||
| Maintenance | 186 | 54 | 0 | 39 | 3 (1) | 3 | 141 still on maintenance | 264 (78) | 145 (43) | |||
Treatment-related mortality (TRM) was defined as death partially attributed to treatment and occurring either while patient was on study or within 2 months of going off study.
Partial response (PR; >75% reduction): denominator for % in remission includes only patients receiving one cycle of protocol therapy.
Complete remission (CR; absence of M-protein by immunofixation): denominator for % in remission includes only patients receiving one cycle of protocol therapy.
Two patients (1 during induction and 1 during second transplantation) went off study due to other reasons (patient/physician choice); these were coded as TRM since death was attributable to treatment and occurred within 2 months of going off TT2.
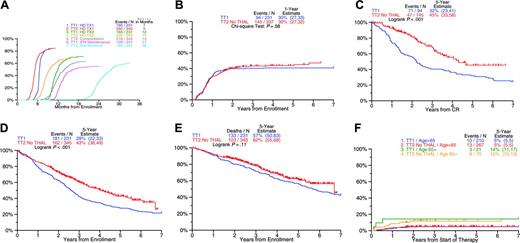
Comparison of clinical outcomes on total therapy 2 without thalidomide (TT2) and total therapy 1 (TT1). (A) Months to treatment step: median time to first and second transplantation 2 months earlier on TT2 compared with TT1. (B) Cumulative incidence of complete remission (CR): similar timing and frequency of CR with TT1 and TT2. (C) Duration of CR: CR duration for TT2 was significantly longer than for TT1. (D) Event-free survival: event-free survival was superior for TT2 compared with TT1. (E) Overall survival: overall survival tended to be superior on TT2 compared with TT1. (F) Cumulative incidence of treatment-related mortality (TRM): similar timing and frequency of TRM with TT1 and TT2.
Comparison of clinical outcomes on total therapy 2 without thalidomide (TT2) and total therapy 1 (TT1). (A) Months to treatment step: median time to first and second transplantation 2 months earlier on TT2 compared with TT1. (B) Cumulative incidence of complete remission (CR): similar timing and frequency of CR with TT1 and TT2. (C) Duration of CR: CR duration for TT2 was significantly longer than for TT1. (D) Event-free survival: event-free survival was superior for TT2 compared with TT1. (E) Overall survival: overall survival tended to be superior on TT2 compared with TT1. (F) Cumulative incidence of treatment-related mortality (TRM): similar timing and frequency of TRM with TT1 and TT2.
Clinical outcome by study
Recognizing that TT2 was the successor trial to TT1, outcome comparisons have to be interpreted with caution. Despite similar CR rates of 43% and 41% with TT2 and TT1 (Figure 1A), a higher proportion of patients receiving TT2 remained in CR at 5 years (45% vs 32%, P < .001; Figure 1C). The 5-year EFS was superior with TT2 (43% vs 28%; P < .001; Figure 1D), and a trend was noted for improved OS (62% vs 57%; P = .11; Figure 1E). TRM was similar and reached 7% for both TT2 and TT1 (Figure 1F).
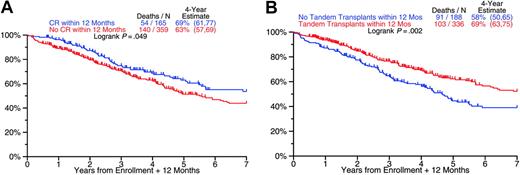
Prognostic impact of achieving CR and of completing tandem transplantations within 12 months (both studies). (A) Overall survival from 12 months after study enrollment by whether or not CR was achieved by month 12: superior outcome among patients with CR. (B) Overall survival from 12 months after study enrollment by whether or not second transplantation was completed by month 12: superior outcome among patients completing second transplantation.
Prognostic impact of achieving CR and of completing tandem transplantations within 12 months (both studies). (A) Overall survival from 12 months after study enrollment by whether or not CR was achieved by month 12: superior outcome among patients with CR. (B) Overall survival from 12 months after study enrollment by whether or not second transplantation was completed by month 12: superior outcome among patients completing second transplantation.
Prognostic factors
CR rates were higher in case of IgA isotype, LDH elevation, and in the absence of CAs; TRM was correlated with low albumin and older age (Table 4). EFS and OS were inferior in the presence of CAs and high LDH with both trials (Table 5). In the case of TT2, EFS and OS both were shorter in patients with IgA MM and low albumin, and OS was inferior in the elderly (higher upper age limit of 75 years with TT2 vs 70 years with TT1). Appreciating the noncontemporaneous nature of the 2 trials, a multivariate analysis was performed that included treatment (TT2, TT1) along with baseline features as variables: TT2 was superior to TT1 for EFS with a trend apparent for OS (Table 5). Patients achieving CR within 12 months and completing tandem transplantations within 12 months had superior OS (Figure 2).
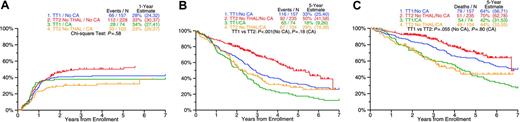
Although the statistical interaction between CAs and therapies was not significant (P > .05), a subgroup analysis was still performed because of the important prognostic implications of CAs. CR rates with TT1 were similar in the absence and in the presence of CAs (42% vs 38%; P = .54); in the case of TT2, 49% of patients without CAs as opposed to 32% with CAs achieved CR (P = .004; Figure 3A). TT2 extended both EFS (Figure 3B) and OS in the no-CAs group (Figure 3C). Among patients with CAs receiving TT2, posttandem transplantation consolidation chemotherapy improved 4-year OS (measured from a 6-month landmark after second transplantation) in comparison with DEX (76% vs 34%, P = .020); in the case of TT1, 4-year OS was 69% in the absence and 47% in the presence of CAs (P = .003; Figure 4). Thus, consolidation chemotherapy in TT2 improved the outcome of the high-risk CA group to the level obtained with TT1 in the low-risk group (76% vs 69%; P = .65).
Clinical outcome by study (TT2 vs TT1) in relationship to presence or absence of cytogenetic abnormalities (CAs). (A) Cumulative incidence of CR: frequency of CR was similar between patients with and without CAs for TT1. TT2 patients with CAs had a lower frequency of CR than patients without CAs. (B) Event-free survival: TT2 improves EFS for both patients with and without CAs. (C) Overall survival: TT2 improves OS only for patients without CAs.
Clinical outcome by study (TT2 vs TT1) in relationship to presence or absence of cytogenetic abnormalities (CAs). (A) Cumulative incidence of CR: frequency of CR was similar between patients with and without CAs for TT1. TT2 patients with CAs had a lower frequency of CR than patients without CAs. (B) Event-free survival: TT2 improves EFS for both patients with and without CAs. (C) Overall survival: TT2 improves OS only for patients without CAs.
Discussion
We have previously reported on the superior CR rate and extended EFS in patients randomized to the thalidomide arm of TT2; due to shorter postrelapse survival among those randomized to thalidomide, OS was similar in the 2 groups.7 A comparison of outcomes of patients randomized to the TT2 arm without thalidomide with those treated with the predecessor TT1 trial was undertaken to examine the potential contributions of dose-intensified induction chemotherapy and the novel concept of posttransplantation consolidation therapy delivered with TT2. Results revealed that, without suffering increased mortality, the twothirds of good-risk patients presenting without CAs entered CR more frequently and enjoyed longer CR, EFS, and OS. High-risk patients presenting with CAs seemed to gain from consolidation chemotherapy, whereas DEX pulsing failed to improve prognosis beyond results obtained with TT1 in this high-risk group. Thirty-nine percent of patients receiving DEX had failed DCEP with induction and thus might constitute per se a less favorable subgroup; their baseline features, however, were similar to those patients receiving DCEP. Those receiving DEX due to failure of recovering an adequate platelet count fared worse than DCEP recipients in the presence of CAs (data not shown), suggesting a beneficial role of DCEP consolidation therapy. Timely application of 2 transplantations and achieving CR both prolonged OS, independent of treatment and other baseline features.
MM with CAs remains a formidable therapeutic problem.18 Even newer agents (thalidomide, lenalidomide, bortezomib), highly effective in the salvage setting, proved more effective among good-risk patients without CAs.19 When applied for the primary management of patients with MM, trials involving thalidomide plus dexamethasone20-22 and especially with added melphalan,23 bortezomib alone,24-26 and especially in combination with dexamethasone and thalidomide19 or with pegylated doxorubicin,27 all have yielded surprisingly high CR rates approaching those reported with single and even tandem autotransplantations. However, follow-up is short, and many patients received subsequently high-dose therapy consolidation, and information on cellular and molecular genetic features is largely lacking. Based on results presented here for TT2, 4-year posttransplantation OS should be in excess of 50%, similar to results recently reported for high-risk MM (high B2M, chromosome 13 deletion by FISH analysis) in trial IFM99-04 (tandem autotransplantation).28
Overall survival from 6 months after second transplantation by study (TT2 with consolidation chemotherapy vs dexamethasone [DEX] vs TT1) and presence or absence of cytogenetic abnormalities (CAs). TT2 patients with baseline CAs, treated with chemotherapy for consolidation, had superior OS compared with those receiving DEX for consolidation. OS was similar for TT2 patients without CAs, regardless of consolidation therapy. OS for TT1 patients with CAs was similar to OS for TT2 patients receiving DEX.
Overall survival from 6 months after second transplantation by study (TT2 with consolidation chemotherapy vs dexamethasone [DEX] vs TT1) and presence or absence of cytogenetic abnormalities (CAs). TT2 patients with baseline CAs, treated with chemotherapy for consolidation, had superior OS compared with those receiving DEX for consolidation. OS was similar for TT2 patients without CAs, regardless of consolidation therapy. OS for TT1 patients with CAs was similar to OS for TT2 patients receiving DEX.
Data reported here suggest that trials are in order that test, prospectively, whether posttransplantation consolidation chemotherapy can improve the prognosis of patients with high-risk CA-type MM. To minimize patient dropout prior to this investigative step, several trials are currently under way in our program to determine whether, by shortening induction therapy to 2 cycles, a higher proportion of patients will complete the intended tandem transplantations in a timely fashion, thereby prolonging OS. Total therapy 3 (TT3), incorporating bortezomib into the frontline therapy of MM, employs only 2 cycles each for induction and consolidation.29 Preliminary results indicate that 79% of 162 patients currently enrolled in TT3 (minimum of 6 months follow-up) indeed completed both scheduled autotransplantations (median completion time of 5 months); in contrast, only 68% of patients on TT2 completed both transplantations (median completion time of 10 months). More than 80% of those on TT3 achieved CR or near CR (immunofixation-positive)30 compared with 77% on the thalidomide arm of TT2 and 66% on the control arm. Other studies, applying CDR3 polymerase chain reaction analysis for quantitation of subclinical disease, are in progress to determine whether the poor outcome of CA-type MM reflects inferior tumor cytoreduction or more rapid disease regrowth.31
Prepublished online as Blood First Edition Paper, December 1, 2005; DOI 10.1182/blood-2005-10-4084.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.

![Figure 4. Overall survival from 6 months after second transplantation by study (TT2 with consolidation chemotherapy vs dexamethasone [DEX] vs TT1) and presence or absence of cytogenetic abnormalities (CAs). TT2 patients with baseline CAs, treated with chemotherapy for consolidation, had superior OS compared with those receiving DEX for consolidation. OS was similar for TT2 patients without CAs, regardless of consolidation therapy. OS for TT1 patients with CAs was similar to OS for TT2 patients receiving DEX.](https://ash.silverchair-cdn.com/ash/content_public/journal/blood/107/7/10.1182_blood-2005-10-4084/5/m_zh80070693400004.jpeg?Expires=1769150528&Signature=RVrgSouuxho3l-ywgmlrWZbXmY51BnJyoFk1C622ufyVqxsMtPUpkqASmGmqbuf-t2wgSQDry2ViabxyL4arFELeog0jGJMffBg77NkVx4-1FS7fcfEW9r9YqOlH1x~Kv0jIPbP-tVL0-ljls5N~EKruwe0O9THXRDYKgrNc2tTbPyw9c23Yc3lyvXDYOHkqbJ7yAqf8sF~Y5uAvS64hfdKtCUfXWbfOn8NANp0Sb-ConvOyrrwRDgt4PmeVAAvwOgrJ3WvU6LaDQVGKcPb06LbrcsAOhzjSG9u8xvvbYJyGvBz~CcPUmfBrYa-LsAbTlFKhd-58S792t45qs9NcCQ__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)
This feature is available to Subscribers Only
Sign In or Create an Account Close Modal