Abstract
Data on incidence, characteristics, and prognosis in stage I childhood anaplastic large cell lymphoma are scarce. Of 463 patients enrolled in the international ALCL99 trial, 36 (8%) had stage I disease and were treated with a prephase chemotherapy, followed by either 3 chemotherapy courses in case of initial complete resection (6 patients) or otherwise by 6 courses of chemotherapy (30 patients). Disease localization was to the peripheral lymph nodes in 26, soft tissue mass in 8, and solitary bone and bronchial disease in 1 patient each. Of the 6 patients with complete resection, none experienced relapse, whereas of the 30 remaining stage I patients, 9 (30%) relapsed, including in all cases a new site of disease involvement and including 3 of 5 anaplastic lymphoma kinase–negative patients. In summary, the failure rate for incompletely resected stage I disease was similar to that for patients with stage II and stage III/IV disease. Whether anaplastic lymphoma kinase negativity contributed to this moderate outcome has to be proven prospectively. This study was registered at www.clinicaltrials.gov as NCT00006455.
Introduction
Anaplastic large cell lymphoma (ALCL) is a distinct clinicopathologic entity of non-Hodgkin lymphoma (NHL) and accounts for 10%-15% of childhood NHL.1,2 Clinical presentation is heterogeneous and includes the frequent involvement of peripheral lymph nodes and extranodal sites, as well as the presence of B-symptoms.3 Optimal treatment of pediatric ALCL has not yet been defined, and therapeutic strategies differ considerably, with some groups adapting chemotherapy according to the extent of disease at diagnosis and others treating all patients with the same regimen.4-9 Regardless of the type of chemotherapy, failure-free survival rates are 70%-75%.4-9
More than 70% of children with ALCL present with advanced-stage disease, so that systematic data are scarce regarding stage I disease, defined as a single tumor (extranodal) or anatomic area (nodal) involved with the exclusion of the mediastinum and abdomen.10 In 1999, the European Intergroup for Childhood NHL (EICNHL) designed a prospective multinational trial for childhood ALCL (ALCL99). Herein, we report on the incidence, clinical characteristics, and outcome of stage I patients included in that trial.
Methods
Between 1999 and 2006, 463 children and adolescents 22 years old or younger with systemic ALCL were enrolled into the ALCL99 trial, including patients from 10 study groups and 12 countries. The diagnosis of ALCL was based on morphologic and immunophenotypic criteria according to the World Health Organization classification system and centrally reviewed in 96% of the patients included in the trial.11 All cases were tested for expression of anaplastic lymphoma kinase (ALK). Staging procedures were performed as described elsewhere.3,10,12-14 All patients were treated with informed consent from the patients, patient's parents, or legal guardians. Studies were conducted in accordance with the Declaration of Helsinki, and approval was given by the ethic committees of all participating centers.
Details concerning eligibility criteria, stratification, and therapy of the ALCL99 trial, as well as overall outcome, have been published previously.12,14 In brief, patients were stratified into 3 subgroups (low-, standard-, and high-risk group) and received prephase chemotherapy followed by pulselike chemotherapy with 5-day courses. The number of courses was based on stage of disease and risk factors as established by the EICNHL.3 There were no strict criteria outlined in the protocol to determine in which patients a complete resection should be performed; however, our guidelines suggested that patients were candidates for complete surgical resection (1) if they had localized disease, (2) if the operation could be performed easily and safely without any significant delay in beginning chemotherapy, and (3) most importantly, if the operation could be performed without resulting in any functional impairment. In patients without completely resectable disease, initial surgery included the least invasive procedure to establish the diagnosis.
Patients included in the low-risk group had complete resection of a stage I tumor (except for completely resected isolated skin disease, which underwent a wait-and-see strategy) and received 3 courses of chemotherapy for a total treatment duration of up to 10 weeks. Patients enrolled in the high-risk group had at least 1 risk factor (skin and/or mediastinal and/or visceral involvement), whereas all others were included in the standard-risk group. Treatment of the standard- and high-risk groups consisted of 6 chemotherapy courses that involved 2 randomized questions with respect to the dosage and type of administration of high-dose methotrexate (standard- and high-risk groups) and the addition of vinblastine to standard chemotherapy and for maintenance therapy (high-risk group only).12,14 Total treatment duration was 4-5 months for patients who did not receive vinblastine and 12 months for the others.
Event-free and overall survival were estimated with Kaplan-Meier curves, and comparisons were performed with log-rank tests. All but 2 patients were followed up for > 2 years (median, 5.7 years).
Results and discussion
Among the 463 patients, we identified 36 children (8%) with stage I disease (6 with and 30 without initial complete resection). Twenty-six patients (72%) had peripheral lymph node involvement only, 8 (22%) presented with a soft tissue mass, and 1 patient each (2.5%) had solitary bone and bronchial disease, respectively. Of the 8 cases with a soft tissue tumor, 3 had underlying bone disease, and 3 had contiguous skin involvement.
All 36 cases were CD30+, 31 (86%) were ALK+, and 31 (86%) expressed at least 1 T-cell marker. Histologic subtyping demonstrated classic ALCL in 26 (72%); mixed ALCL in 6 (17%), including a small cell or lymphohistiocytic component in each of them; small cell ALCL in 2 (6%); and Hodgkin-like and not further specified ALCL in 1 patient each (2.5%). Of note, all 5 ALK− patients showed classic histopathology, including 4 with a T-cell phenotype. Sites of involvement were soft tissue in 3 patients and peripheral lymph nodes in 2. All 5 ALK− patients were assigned to the group without initial complete resection.
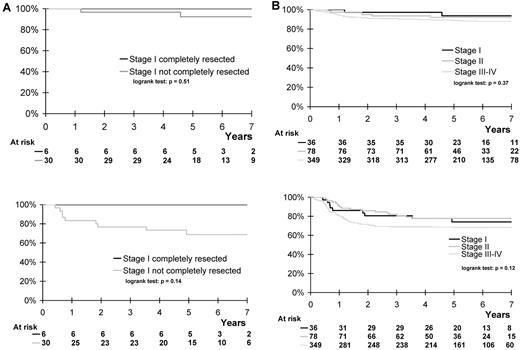
The incidence of peripheral lymph node involvement, B-symptoms, ALK positivity, and high levels of lactate dehydrogenase was significantly lower in stage I than in stage II or stage III/IV ALCL99 patients (Table 1). All 6 patients with complete resection were included in the low-risk group and neither experienced relapse nor died, whereas the other 30 patients were treated in the standard-risk group, with 9 of them (30%; 95% confidence interval 14%-46%) having recurrent disease (new localization, n = 7; new localization associated with local relapse, n = 2; Figure 1A). Seven of these cases occurred among the 26 patients with initial peripheral lymph node involvement, whereas the other 2 cases had soft tissue and bone/soft tissue disease, respectively. The histologic subtype of the relapsed patients was classic ALCL in 6 patients and mixed, small cell, and unspecified ALCL in 1 patient each. Three relapses occurred among the 5 ALK− patients. Seven of the relapsed patients are still alive in second remission. Three-year event-free and overall survival of the 36 patients with stage I disease was not superior to that of the 78 stage II and 349 stage III/IV patients (Figure 1B). Among the entire cohort of 36 stage I patients, histologic subtype (P = .583), ALK expression (P = .066), and peripheral lymph node involvement (P = .689) were not significantly associated with outcome.
Comparison of clinical and laboratory features at initial diagnosis of 463 ALCL99 patients according to stage of disease
| . | Stage I (n = 36) . | Stage II (n = 78) . | Stage III/IV (n = 349) . | P, I vs II vs III/IV . |
|---|---|---|---|---|
| Male sex, n (%) | 20 (56) | 49 (63) | 213 (61) | .76 |
| Mean age at biopsy,y (±SD) | 11.2 ± 4.1 | 11.0 ± 3.9 | 10.2 ± 4.5 | .21 |
| Histologic subtype: LH/SC/mixed ALCL, n (%) | 8 (23) | 15 (21) | 106 (33) | .08 |
| Immunophenotype, n (%) | ||||
| ALK+ | 31 (86) | 75 (96) | 335 (96) | .05 |
| Cell lineage, n (%) | ||||
| Null | 5 (14) | 12 (15) | 39 (11) | .49 |
| T | 31 (86) | 66 (85) | 310 (89) | |
| B-symptoms, n (%) | 4 (11) | 21 (27) | 234 (68) | < .0001 |
| Localization, n (%) | ||||
| Lymph node | 26 (72) | 72 (92) | 301 (86) | .02 |
| Soft tissue | 8 (22) | 15 (19) | 51 (15) | .34 |
| Bone lesion | 4* (11) | 6 (8) | 67 (23) | .008 |
| LDH (before treatment), U/L | 314 (±232) | 366 (±216) | 563 (±651) | .004 |
| . | Stage I (n = 36) . | Stage II (n = 78) . | Stage III/IV (n = 349) . | P, I vs II vs III/IV . |
|---|---|---|---|---|
| Male sex, n (%) | 20 (56) | 49 (63) | 213 (61) | .76 |
| Mean age at biopsy,y (±SD) | 11.2 ± 4.1 | 11.0 ± 3.9 | 10.2 ± 4.5 | .21 |
| Histologic subtype: LH/SC/mixed ALCL, n (%) | 8 (23) | 15 (21) | 106 (33) | .08 |
| Immunophenotype, n (%) | ||||
| ALK+ | 31 (86) | 75 (96) | 335 (96) | .05 |
| Cell lineage, n (%) | ||||
| Null | 5 (14) | 12 (15) | 39 (11) | .49 |
| T | 31 (86) | 66 (85) | 310 (89) | |
| B-symptoms, n (%) | 4 (11) | 21 (27) | 234 (68) | < .0001 |
| Localization, n (%) | ||||
| Lymph node | 26 (72) | 72 (92) | 301 (86) | .02 |
| Soft tissue | 8 (22) | 15 (19) | 51 (15) | .34 |
| Bone lesion | 4* (11) | 6 (8) | 67 (23) | .008 |
| LDH (before treatment), U/L | 314 (±232) | 366 (±216) | 563 (±651) | .004 |
LH indicates lymphohistiocytic variant; SC, small cell variant; mixed ALCL, LH and/or SC component; and LDH, lactate dehydrogenase.
Of the 4 patients,1 had solitary bone disease and 3 had a concomitant soft tissue mass.
Overall and event-free survival according to initial resection and stage of disease. (A) Overall and event-free survival of 36 ALCL99 patients with stage I disease, according to initial resection. Three-year overall survival: patients with complete resection, 100%; patients without complete resection, 97% (83%-99%). Three-year event-free survival: patients with complete resection, 100%; patients without complete resection, 77% (59%-88%). (B) Overall and event-free survival of 463 ALCL99 patients* according to stage of disease. Three-year overall survival: stage I patients, 97% (86%-100%); stage II patients, 94% (86%-97%); stage III/IV patients, 91% (87%-93%). Three-year event-free survival: stage I patients, 81% (65%-90%); stage II patients, 82% (72%-89%); stage III/IV patients, 69% (64%-74%). *All but 2 patients were followed up for > 2 years (median, 5.7 years)
Overall and event-free survival according to initial resection and stage of disease. (A) Overall and event-free survival of 36 ALCL99 patients with stage I disease, according to initial resection. Three-year overall survival: patients with complete resection, 100%; patients without complete resection, 97% (83%-99%). Three-year event-free survival: patients with complete resection, 100%; patients without complete resection, 77% (59%-88%). (B) Overall and event-free survival of 463 ALCL99 patients* according to stage of disease. Three-year overall survival: stage I patients, 97% (86%-100%); stage II patients, 94% (86%-97%); stage III/IV patients, 91% (87%-93%). Three-year event-free survival: stage I patients, 81% (65%-90%); stage II patients, 82% (72%-89%); stage III/IV patients, 69% (64%-74%). *All but 2 patients were followed up for > 2 years (median, 5.7 years)
Childhood ALCL inherently is a rare disease, with disease-free survival rates (70%-75%) lagging behind the good outcome achieved in mature B-cell and lymphoblastic lymphoma (80%-85%).2,15,16 While two-thirds of patients present with disseminated disease at initial diagnosis, patients with limited-stage disease are rarely observed and reported.4,5,7-9,17,18 Before designing the ALCL99 trial, the EICNHL undertook an analysis on 225 ALCL patients to identify risk factors at diagnosis that predicted treatment failure.3 That study demonstrated that the St Jude staging system did not adequately address the high proportion of extranodal involvement in childhood ALCL. Moreover, the St Jude classification also lost prognostic value when the identified risk factors (skin, mediastinal, and visceral involvement) were considered for analysis of outcome. Nevertheless, completely resected stage I disease remained as an excellent prognostic subgroup. Thus, in the ALCL99 trial, treatment stratification did not rely primarily on the St Jude staging criteria (except for patients with completely resected stage I) but on the newly established risk factors.
To the best of our knowledge, the ALCL99 trial, which covers a 6-year time period, represents the largest series of uniformly treated children with ALCL. Thus, incidence rates and distribution of disease stages can be considered reproducible. The present data show that limited stage I ALCL disease has an incidence of 8%, which is in the same range as in other available studies.4,5,7-9,19
Although the number of patients identified was rather low, the present study showed that initially completely resected stage I ALCL has a good prognosis with 3 courses of chemotherapy; however, patients with stage I disease and no complete resection had a relapse rate of 30% at 5 years, which was no better than the rate observed in patients with stage II or stage III/IV disease. Because a new site of disease involvement was found in all cases at the time of relapse, there appeared to be a lack of systemic disease control in an apparently limited-stage lymphoma. Whether this fairly good outcome for pediatric stage I ALCL could be improved by intensification of conventional contemporary chemotherapy, refinement of risk stratification with new prognostic markers, or new treatment approaches such as additional immunotherapy (ie, anti-CD30 antibody) or long-term vinblastine monotherapy, as used in relapsed patients, is an issue of ongoing debate for future clinical trials.
Moreover, because several recent studies showed that the extent of submicroscopic disease in bone marrow or peripheral blood at diagnosis (as assessed by polymerase chain reaction for NPM1-ALK) correlated with the stage of disease and allowed the identification of patients who had an increased risk of relapse, we can only speculate that the relapses in stage I disease may have occurred because of high minimal disseminated disease at diagnosis.20,21 Although not statistically significant, the moderate outcome of stage I ALCL may also be explained by the adverse biologic factor of ALK negativity, with 3 of 5 ALK− patients and 6 of 31 ALK+ patients having a relapse (P = .066). In addition, except for the higher proportion of ALK− patients among the group with no complete resection, reasons explaining the difference in outcome between completely resected and nonresected stage I disease remain elusive. However, this observation should in no way suggest that physicians should aim for complete resection in a disease that responds readily to polychemotherapy, because this leaves patients at risk for mutilation and functional impairment.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Acknowledgments
We thank all participating institutions and physicians and nurses for their support of the study, as well as Nathalie Bouvet-Forteau for data management of the international database.
This EICNHL paper was written on behalf of the Société Française de Lutte contre les Cancers et Leucémies de l'enfants, Berlin-Frankfurt-Münster (BFM) Study Group, Associazione Italiana Ematologia Oncologia Pediatrica (AIEOP), United Kingdom Children's Cancer and Leukemia Study Group (CCLG), Nordic Society of Pediatric Hematology and Oncology (NOPHO), Polish Pediatric Leukemia and Lymphoma Study Group (PPLLSG), Belgian Society of Pediatric Hematology and Oncology, Dutch Childhood Oncology Group (DCOG), and Japanese Pediatric Leukemia/Lymphoma Study Group (JPLSG).
This work was supported by the Association Cent Pour Sang la Vie (Institute Gustave-Roussy), Cancer Research United Kingdom, the Forschungshilfe Station Peiper (BFM Germany), the St Anna Kinderkrebsforschung (BFM Austria), the Associazione Italiana Contro le Leucemie and Fondazione Citta della Speranza (AIEOP), and the Ministry of Health, Labor, and Welfare of Japan.
Authorship
Contribution: A.A., M.L.D., and L.B. wrote the manuscript; A. Rosolen, D.W., A. Reiter, M.L.D., and L.B. designed and planned the study; M.L.D. was in charge of data pooling, data checking, and statistical analysis; A.M. revised the data and performed statistical analyses; and A.A., G.M., A. Rosolen, D.W., A.U., I.M., L.L., K.H., G.W., A.B., W.W., A. Reiter, and L.B. served as principal or coinvestigators in their study groups and institutions, coordinated the study in their countries, provided study materials, and recruited patients. All authors read and approved the final version of the manuscript.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Andishe Attarbaschi, Associate Professor, MD, St Anna Children's Hospital, Kinderspitalgasse 6, 1090 Vienna, Austria; e-mail: andishe.attarbaschi@stanna.at.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal