Abstract
The achievement of CR after high-dose therapy/ASCT (HDT/ASCT) is a surrogate for prolonged survival in MM. While this is well accepted, the long-term clinical outcome of MM patients achieving CR is still heterogeneous and a small fraction of patients unexpectedly lose their CR status early on after HDT/ASCT and experience a dismal survival. In fact, survival of patients with unsustained CR is even poorer than that of those not achieving CR.
Herein we sought to identify prognostic markers predictive of unsustained CR after HDT/ASCT. The study included a total of 241 patients achieving CR at day+100 after HDT/ASCT treated in two consecutive GEM/PETHEMA trials: GEM2000 (VBMCP/VBAD, n=140) and GEM2005<65y (randomized induction with the same chemotherapy plus bortezomib in the last two cycles or thalidomide/dexamethasone or bortezomib/thalidomide/dexamethasone followed by HDT/ASCT; N=101). All cases were referred for MRD assessment by MFC at day+100 after HDT/ASCT; baseline FISH analysis were available in 110 of the 241 patients.
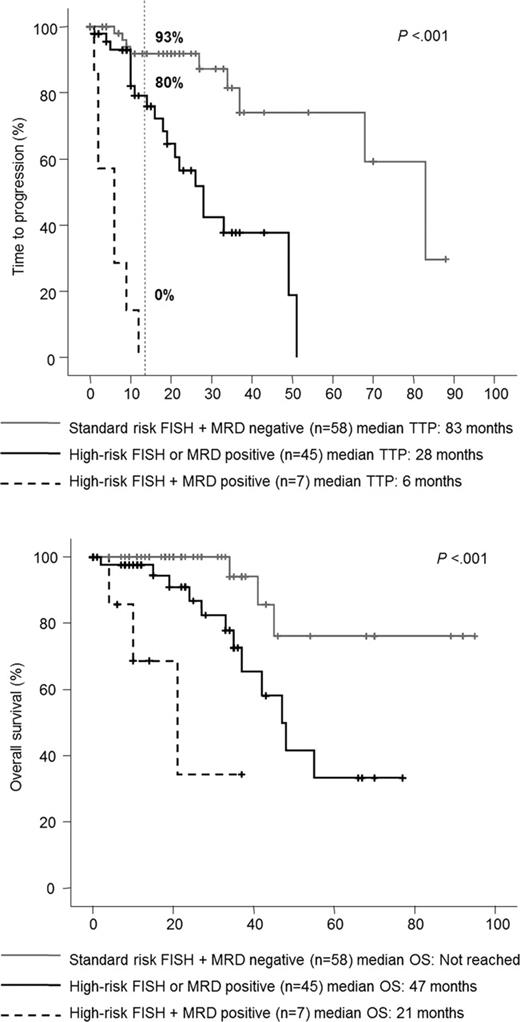
We first investigated which of the most relevant disease characteristics had prognostic influence in patients in CR at day+100 after HDT/ASCT. In this cohort some markers with consistent influence in unselected MM populations such as patient age, ISS stage, serum β2-microglobulin, BMPC burden and % of PC in S-phase were not significantly predictive. In contrast, the presence of baseline anemia was a significant prognostic marker for OS (P=.01). Regarding cytogenetics, 16% of CR patients were considered with high-risk disease at presentation and showed a significantly inferior TTP (3-years, 40% vs 79%; P=.001) and borderline OS (3-years, 73% vs 96%; P=.07) compared to cases with standard-risk. In addition, MFC immunophenotyping showed persistent MRD in 87 of the 241 (36%) CR patients, and the failure to achieve an immunophenotypic CR at day+100 after HDT/ASCT resulted in significantly inferior TTP (3-years, 58% vs 85%; P<.001) and OS (3-years, 80% vs 90%; P=.001). In the multivariate analysis, the best combination of independent predictive parameters for TTP were immunophenotypic CR status (P<.001;HR=7.1) and FISH cytogenetics (P<.001;HR=5.6); in turn, for OS immunophenotypic CR status (P=.001;HR=7.7), FISH cytogenetics (P=.01;HR=5.1) and age ≥60 years (P=.03;HR=3.4) were selected.
We further explored the clinical impact of the immunophenotypic CR in the context of standard-risk and high-risk disease. The best prognosis was for patients with both standard-risk cytogenetics and in immunophenotypic CR (3y: TTP, 92%; OS, 100%), while the worst outcome occurred in cases with both high-risk disease and persistent MRD (3y: TTP, 0%; OS, 32%). The two other categories had an intermediate prognosis, although it was better for patients with high-risk cytogenetics achieving an immunophenotypic CR (3y: TTP, 69%; OS, 100%) than for patients with standard-risk disease but failing to achieve an immunophenotypic CR after HDT/ASCT (3y: TTP, 57%; OS, 90%).
We then investigated the parameters that could help to identify patients in CR at risk of early relapse after HDT/ASCT. Of the 241 patients, 30 (12%) progressed within 1 year after HDT/ASCT (unsustained CR) and this subgroup showed a dismal outcome, with a median OS of only 39 months (P<.001). Patients with unsustained CR showed significantly higher incidence of anemia at baseline (48% vs 24%, P=.01), advanced ISS stage 2 or 3 (86% vs 55%, P=.003), high-risk cytogenetics (40% vs 10%, P=.005) and persistent MRD by MFC (63% vs 32%, P=.001) compared with the remaining cases. At the multivariate analysis, only cytogenetic abnormalities (P=.002;HR=12.5) and the immunophenotypic CR status at day+100 after HDT/ASCT (P=.001;HR=8.8) emerged as independent predictive markers for unsustained CR. Based on these two variables, we established a predictive index by assigning one point for each adverse factor. Accordingly, 3 risk groups of patients in CR after HDT/ASCT were defined, with significantly (P<.001) different rates of disease progression within the first year after HDT/ASCT for patients with none, one, or both risk factors (7%, 20% and 100%, respectively; Figure).
Paiva:Celgene: Honoraria; Janssen: Honoraria. Rosiñol:Janssen: Honoraria; Celgene: Honoraria. Mateos:Janssen: Honoraria; Celgene: Honoraria. Cibeira:Janssen: Honoraria; Celgene: Honoraria. Alegre:Janssen: Honoraria; Celgene: Honoraria. Lahuerta:Janssen: Honoraria; Celgene: Honoraria. Blade:Janssen: Honoraria; Celgene: Honoraria. San Miguel:Janssen-Cilag: Honoraria; Celgene: Honoraria.

This icon denotes a clinically relevant abstract
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal