Abstract
HSCT is being increasingly offered as a curative option for children with hematologic malignancies. Although survival has improved, the long-term morbidity ascribed to the HSCT procedure is not known. We compared the risk of chronic health conditions and adverse health among children with cancer treated with HSCT with survivors treated conventionally, as well as with sibling controls. HSCT survivors were drawn from BMTSS (N = 145), whereas conventionally treated survivors (N = 7207) and siblings (N = 4020) were drawn from CCSS. Self-reported chronic conditions were graded with CTCAEv3.0. Fifty-nine percent of HSCT survivors reported ≥ 2 conditions, and 25.5% reported severe/life-threatening conditions. HSCT survivors were more likely than sibling controls to have severe/life-threatening (relative risk [RR] = 8.1, P < .01) and 2 or more (RR = 5.7, P < .01) conditions, as well as functional impairment (RR = 7.7, P < .01) and activity limitation (RR = 6.3, P < .01). More importantly, compared with CCSS survivors, BMTSS survivors demonstrated significantly elevated risks (severe/life-threatening conditions: RR = 3.9, P < .01; multiple conditions: RR = 2.6, P < .01; functional impairment: RR = 3.5, P < .01; activity limitation: RR = 5.8, P < .01). Unrelated donor HSCT recipients were at greatest risk. Childhood HSCT survivors carry a significantly greater burden of morbidity not only compared with noncancer populations but also compared with conventionally treated cancer patients, providing evidence for close monitoring of this high-risk population.
Introduction
HSCT has been increasingly offered as a curative option for children with hematologic malignancies.1,2 For Hodgkin lymphoma (HL), non-Hodgkin lymphoma (NHL), and acute lymphoblastic leukemia (ALL), HSCT is often the treatment of choice after conventional therapy fails to control disease. However, for acute myeloid leukemia (AML) or very high-risk ALL, HSCT may be used as a frontline consolidative measure, given availability of a matched related donor; if such a donor is not available, HSCT is reserved for patients whose disease recurs after conventional chemotherapy.
Long-term survival is an expected outcome for many children and adolescents who undergo HSCT.1,3,4 The growing population of long-term survivors has brought to the medical forefront a host of chronic and debilitating conditions attributed to toxicity from pretransplantation exposure, transplantation conditioning regimens, chronic immunosuppression, and GVHD.3-8 Late-occurring sequelae such as delayed immune reconstitution, recurrent infections, and chronic dermatologic conditions may be the result of chronic GVHD and are unique to allogeneic HSCT survivors.4,7,9 However, well-described complications such as second malignant neoplasms, cardiac dysfunction, growth and gonadal failure, neurocognitive delay, and other end-organ dysfunction may result from the combined effects of pre-HSCT treatment exposures, HSCT-related conditioning, and post-HSCT complications such as GVHD.3,4,9 As a result, any assessment of long-term health-related outcomes for survivors of HSCT performed in childhood has to be considered in the context of the pre-HSCT and HSCT-related exposures unique to that population.
There is paucity of information regarding the burden of morbidity carried by survivors of HSCT performed in childhood. In particular, there exists very few data on the morbidity and mortality associated with HSCT-related conditioning and GVHD that is above and beyond the well-documented risk related to conventional therapy.10-13 In the current study we address this gap in knowledge by using 2 unique resources of childhood cancer survivors, the Childhood Cancer Survivor Study (CCSS: conventionally treated patients) and the Bone Marrow Transplant Survivor Study (BMTSS: children treated with HSCT) to understand the individual and independent contribution of pre-HSCT therapeutic exposures, conditioning-related exposures, and GVHD in the development of adverse health outcomes in long-term HSCT survivors.
Methods
Subjects
CCSS is a multiinstitutional cohort of 5-year survivors of cancer diagnosed between 1970 and 1986 at age ≤ 21 years. The current study included CCSS participants with AML, ALL, HL, and NHL treated conventionally (ie, without HSCT). Fourteen percent were lost to follow-up, and the study participation rate was 70% for the remaining eligible patients. BMTSS examined long-term outcomes in patients undergoing HSCT between 1974 and 1998 at City of Hope or University of Minnesota. Recognizing differences in baseline eligibility criteria between CCSS and BMTSS, we limited BMTSS participants to those < 21 years of age at diagnosis of a hematologic malignancy; who had survived ≥ 5 years from their primary diagnosis; who had undergone HSCT within 3 years of diagnosis; and who were ≥ 2 years from myeloablative HSCT. Sixteen percent were lost to follow-up, and the study participation rate was 60% for the remaining eligible patients. To allow for comparisons with a noncancer population, a random sample of CCSS survivors were asked to identify a nearest-age living sibling to be included in the study. All study participants provided written informed consent for participation in the study in accordance with the Declaration of Helsinki, and the study was approved by the St. Jude Research Hospital institutional review board. Detailed descriptions of the CCSS14,15 and BMTSS16 study designs have been reported previously. Study materials for the CCSS, including questionnaires and treatment abstraction forms, are available at http://ccss.stjude.org.
Sociodemographic and clinical characteristics
Information related to sociodemographic and clinical characteristics, including age at diagnosis and participation, sex, race/ethnicity, and primary diagnosis, were obtained from the CCSS and BMTSS databases. The BMTSS cohort was also characterized in terms of stem cell source (autologous, related or unrelated donor), disease status at HSCT (first complete remission [CR1], all others), and history of GVHD. The presence or absence of active chronic GVHD in the preceding 12 months was self-reported.
Therapeutic exposures
Treatment summaries were generated from medical records for the CCSS and BMTSS cohorts and included detailed information regarding chemotherapy, radiation, and surgery.10 For BMTSS participants, treatment-related exposures were categorized as pre-HSCT exposures (corresponding to the entire treatment experience of the CCSS cohort) and HSCT-related exposures. HSCT-related exposures included conditioning regimens, prophylaxis and management of GVHD, and post-HSCT therapeutic exposures (in the event of disease recurrence).
Chronic health conditions
All study participants (BMTSS and CCSS survivors and siblings) completed a nearly identical questionnaire that covers the following general areas: questions regarding diagnosis by a health care provider of physical health conditions (endocrinopathies, CNS compromise, cardiopulmonary dysfunction, gastrointestinal and hepatic sequelae, musculoskeletal abnormalities, and subsequent malignancies), along with age at diagnosis; access to and use of medical care; and sociodemographic characteristics (race/ethnicity, education, marital status, employment, household income, and insurance). The BMTSS questionnaire also asked questions about the diagnosis and extent of chronic GVHD and presence of active GVHD within the past 12 months of study participation. The reliability and validity of the BMTSS questionnaire have been tested by researchers, 17 who have confirmed that survivors are able to report the occurrence of adverse health conditions with accuracy.
Chronic physical health conditions were graded by the use of the Common Terminology Criteria for Adverse Events version 3.0 (CTCAEv3.0; supplemental Table 1, available on the Blood Web site; see the Supplemental Materials link at the top of the online article). The CTCAEv3.0 has been used to grade acute and chronic conditions in patients with cancer and cancer survivors,10,16 from grade 1 (mild) through 5 (adverse event-related death). This scoring system was developed by Oeffinger and colleagues10 for grading severity of conditions in childhood cancer survivors and their siblings. Adverse psychosocial outcomes were not included in this analysis. Because of the design of the study, there were no siblings with a grade 5 (fatal) condition.
Health status measures
Five domains of health status were measured: general health, functional status, limitations of activity, pain, and cancer-related anxiety or fear. Criteria used to define adverse health status were adapted from those used by Hudson et al11 in the CCSS and subsequently reported in adult BMTSS participants (supplemental Table 2).18
Statistical analyses
Chronic health conditions.
The prevalence of chronic health conditions was determined for the BMTSS and CCSS cohorts and siblings. Chronic health conditions were evaluated as any chronic health condition (grades 1 through 4); severe/life-threatening conditions (grade 3 or 4); and multiple (≥ 2) conditions (grades 1 through 4). For participants with more than one condition, the maximum grade was used in the analysis. The BMTSS cohort was compared with unaffected siblings and with the CCSS cohort. Standard parametric and nonparametric techniques were used for comparison between clinical, demographic, and treatment-related variables.
BMTSS versus sibling controls.
For deceased HSCT survivors, the maximum grade of a condition reported before death was used for comparison with siblings.
BMTSS versus CCSS.
Fatal health conditions (grade 5) were included in the comparison between these 2 populations.
Adverse health status.
Prevalence of adverse health status (poor general health, functional impairment, activity limitation) was compared between BMTSS survivors and siblings as well as between BMTSS and CCSS survivors. Comparison of pain and fear/anxiety was limited to the BMTSS and CCSS survivors.
Predictors of chronic health conditions or adverse health status.
Comparisons between BMTSS survivors and siblings and between BMTSS and CCSS survivors were conducted by the use of generalized linear models with a log link function and were reported as relative risks (RR) with 95% confidence intervals (95% CI). A fixed set of explanatory HSCT-related variables were selected a priori and included stem cell source (autologous, related, and unrelated donor), the presence of active chronic GVHD, and conditioning exposures (chemotherapy alone, chemotherapy plus total body irradiation [TBI]). In addition, the model included sociodemographic variables (age at study participation, race/ethnicity, sex, and health insurance). Recognizing that there could possibly be an inherent difference between BMTSS and CCSS survivors, the following variables were also included in all comparisons between the 2 survivor cohorts: age at diagnosis, time from diagnosis to study participation, year of diagnosis, underlying diagnosis, cranial radiation, chest radiation, cumulative anthracycline dose,19 alkylating agent score,20 and exposure to platinum agents and epipodophyllotoxins. Data were analyzed with SPSS 18.0 (IBM Corporation). All statistical tests were 2-sided, and P < .05 was considered statistically significant.
Results
Sociodemographic characteristics
Sociodemographic characteristics of the BMTSS survivors (N = 145), CCSS survivors (N = 7207), and the sibling comparison group (N = 4020) are presented in Table 1. Compared with the siblings, BMTSS survivors were more likely to be male, younger at study participation, and less likely to be non-Hispanic white. Compared with CCSS survivors, BMTSS survivors were more likely to be insured.
Sociodemographic characteristics of HSCT survivors (BMTSS), conventionally treated cancer survivors (CCSS), and sibling controls (CCSS)
| Characteristic . | BMTSS (N = 145) . | Conventionally treated, CCSS (N = 7207) . | P* . | Sibling controls, CCSS (N = 4020) . | P† . |
|---|---|---|---|---|---|
| Sex, no. (%) | |||||
| Male | 82 (56.6) | 3972 (55.1) | .73 | 1936 (48.2) | .05 |
| Female | 63 (43.4) | 3235 (44.9) | 2084 (51.8) | ||
| Ethnicity, no. (%) | .77 | .01 | |||
| Non-Hispanic white | 122 (84.1) | 5973 (83.2) | 3506 (90.4) | ||
| Other | 23 (15.9) | 1205 (16.8) | 373 (9.6) | ||
| Age at study participation, y | .10 | < .01 | |||
| Mean (± SD) | 24.0 ± 8.4 | 25.1 ± 7.9 | 26.6 ± 9.2 | ||
| Range | 5.37-44.96 | 8.8-49.1 | 1.8-56.2 | ||
| Education, no. (%) | .15 | .14 | |||
| < High school | 42 (31.8) | 2227 (32.6) | 961 (25.0) | ||
| High school graduate | 15 (11.4) | 1181 (17.3) | 591 (15.4) | ||
| > High school education | 75 (56.8) | 3430 (50.2) | 2294 (59.6) | ||
| Household income, no. (%) | .18 | .28 | |||
| < $20 000/yr | 20 (15.4) | 1285 (20.1) | 443 (12.1) | ||
| ≥ $20 000/yr | 111 (84.7) | 5122 (79.9) | 3216 (87.9) | ||
| Health insurance, no. (%) | .01 | .76 | |||
| No | 10 (6.9) | 953 (14.3) | 390 (9.8) | ||
| Yes/Canada | 134 (92.5) | 5700 (85.7) | 3583 (90.2) |
| Characteristic . | BMTSS (N = 145) . | Conventionally treated, CCSS (N = 7207) . | P* . | Sibling controls, CCSS (N = 4020) . | P† . |
|---|---|---|---|---|---|
| Sex, no. (%) | |||||
| Male | 82 (56.6) | 3972 (55.1) | .73 | 1936 (48.2) | .05 |
| Female | 63 (43.4) | 3235 (44.9) | 2084 (51.8) | ||
| Ethnicity, no. (%) | .77 | .01 | |||
| Non-Hispanic white | 122 (84.1) | 5973 (83.2) | 3506 (90.4) | ||
| Other | 23 (15.9) | 1205 (16.8) | 373 (9.6) | ||
| Age at study participation, y | .10 | < .01 | |||
| Mean (± SD) | 24.0 ± 8.4 | 25.1 ± 7.9 | 26.6 ± 9.2 | ||
| Range | 5.37-44.96 | 8.8-49.1 | 1.8-56.2 | ||
| Education, no. (%) | .15 | .14 | |||
| < High school | 42 (31.8) | 2227 (32.6) | 961 (25.0) | ||
| High school graduate | 15 (11.4) | 1181 (17.3) | 591 (15.4) | ||
| > High school education | 75 (56.8) | 3430 (50.2) | 2294 (59.6) | ||
| Household income, no. (%) | .18 | .28 | |||
| < $20 000/yr | 20 (15.4) | 1285 (20.1) | 443 (12.1) | ||
| ≥ $20 000/yr | 111 (84.7) | 5122 (79.9) | 3216 (87.9) | ||
| Health insurance, no. (%) | .01 | .76 | |||
| No | 10 (6.9) | 953 (14.3) | 390 (9.8) | ||
| Yes/Canada | 134 (92.5) | 5700 (85.7) | 3583 (90.2) |
Percentages are determined by the total number of participants who provided data for each variable, rather than on the total number of subjects in each cohort; percentages may not total 100 because of rounding.
BMTSS indicates Bone Marrow Transplant Survivor Study; and CCSS, Childhood Cancer Survivor Study.
BMTSS vs conventionally treated cancer survivors (CCSS).
BMTSS vs siblings controls (CCSS).
Treatment-related characteristics
Clinical characteristics of the BMTSS and CCSS survivors are presented in Table 2. BMTSS survivors were more likely to have been diagnosed during a more recent era; there was an overrepresentation of AML and an underrepresentation of ALL, NHL, and HL among BMTSS survivors. BMTSS survivors were older at diagnosis, had a shorter duration of follow-up, and were more likely to have been treated with anthracyclines, epidophyllotoxins, and platinum agents. However, CCSS survivors were more likely to have received alkylating agents and cranial and chest radiation.
Treatment-related characteristics of HSCT survivors (BMTSS) and conventionally treated cancer survivors (CCSS)
| Treatment-related variables . | BMTSS (N = 145) . | Conventionally treated, CCSS (N = 7207) . | P . |
|---|---|---|---|
| Year of diagnosis | < .01 | ||
| 1970-1975 | 3 (2.1) | 1745 (24.2) | |
| 1976-1981 | 21 (14.5) | 2636 (36.6) | |
| 1982-1986 | 24 (16.6) | 2826 (39.2) | |
| 1987-1991 | 49 (33.8) | N/A | |
| 1992-1997 | 48 (33.1) | N/A | |
| Age at diagnosis, y | < .01 | ||
| Mean (± SD) | 10.9 ± 6.9 | 8.9 ± 5.7 | |
| Range | 0.1-20.9 | 0.1-20.9 | |
| Diagnosis | < .01 | ||
| Acute myeloid leukemia | 73 (50.3) | 267 (3.7) | |
| Acute lymphoblastic leukemia | 51 (35.2) | 4017 (55.7) | |
| Hodgkin lymphoma | 11 (7.6) | 1879 (26.1) | |
| Non-Hodgkin lymphoma | 10 (6.9) | 1044 (14.5) | |
| Time from diagnosis, y | < .01 | ||
| Mean (± SD) | 12.9 ± 5.6 | 16.2 ± 4.9 | |
| Range | 5.1-26.1 | 6.4-31.1 | |
| Chemotherapy | |||
| Anthracycline, mg/m2 | < .01 | ||
| None | 12 (8.3) | 3689 (61.0) | |
| 1-249 | 73 (50.3) | 1167 (19.3) | |
| ≥ 250 | 60 (41.4) | 1194 (19.7) | |
| Alkylating agent | < .01 | ||
| None | 99 (68.3) | 2467 (44.5) | |
| Alkylating agent score 1-2 | 38 (26.2) | 2322 (41.9) | |
| Alkylating agent score ≥ 3 | 8 (5.5) | 752 (13.6) | |
| Epidophyllotoxins | < .01 | ||
| Any | 65 (44.8) | 497 (8.0) | |
| Platinum agents | < .01 | ||
| Any | 8 (5.5) | 19 (0.3) | |
| Radiation | |||
| Brain | 25 (17.2) | 2550 (35.4) | < .01 |
| Chest | 8 (5.5) | 1665 (23.1) | < .01 |
| HSCT-related | |||
| Autologous HSCT | 40 (27.6) | N/A | |
| Allogeneic-related | 85 (58.6) | N/A | |
| Allogeneic-unrelated | 20 (13.8) | N/A | |
| Disease status at HSCT | |||
| First complete remission (CR1) | 85 (58.6) | N/A | |
| ≥ CR1 | 50 (34.5) | N/A | |
| Conditioning | |||
| Chemotherapy | 34 (23.4) | N/A | |
| Chemotherapy + TBI | 111 (76.6) | N/A | |
| Chronic GVHD* | |||
| No | 71 (67.6) | N/A | |
| Resolved | 20 (19.0) | N/A | |
| Active | 14 (13.3) | N/A |
| Treatment-related variables . | BMTSS (N = 145) . | Conventionally treated, CCSS (N = 7207) . | P . |
|---|---|---|---|
| Year of diagnosis | < .01 | ||
| 1970-1975 | 3 (2.1) | 1745 (24.2) | |
| 1976-1981 | 21 (14.5) | 2636 (36.6) | |
| 1982-1986 | 24 (16.6) | 2826 (39.2) | |
| 1987-1991 | 49 (33.8) | N/A | |
| 1992-1997 | 48 (33.1) | N/A | |
| Age at diagnosis, y | < .01 | ||
| Mean (± SD) | 10.9 ± 6.9 | 8.9 ± 5.7 | |
| Range | 0.1-20.9 | 0.1-20.9 | |
| Diagnosis | < .01 | ||
| Acute myeloid leukemia | 73 (50.3) | 267 (3.7) | |
| Acute lymphoblastic leukemia | 51 (35.2) | 4017 (55.7) | |
| Hodgkin lymphoma | 11 (7.6) | 1879 (26.1) | |
| Non-Hodgkin lymphoma | 10 (6.9) | 1044 (14.5) | |
| Time from diagnosis, y | < .01 | ||
| Mean (± SD) | 12.9 ± 5.6 | 16.2 ± 4.9 | |
| Range | 5.1-26.1 | 6.4-31.1 | |
| Chemotherapy | |||
| Anthracycline, mg/m2 | < .01 | ||
| None | 12 (8.3) | 3689 (61.0) | |
| 1-249 | 73 (50.3) | 1167 (19.3) | |
| ≥ 250 | 60 (41.4) | 1194 (19.7) | |
| Alkylating agent | < .01 | ||
| None | 99 (68.3) | 2467 (44.5) | |
| Alkylating agent score 1-2 | 38 (26.2) | 2322 (41.9) | |
| Alkylating agent score ≥ 3 | 8 (5.5) | 752 (13.6) | |
| Epidophyllotoxins | < .01 | ||
| Any | 65 (44.8) | 497 (8.0) | |
| Platinum agents | < .01 | ||
| Any | 8 (5.5) | 19 (0.3) | |
| Radiation | |||
| Brain | 25 (17.2) | 2550 (35.4) | < .01 |
| Chest | 8 (5.5) | 1665 (23.1) | < .01 |
| HSCT-related | |||
| Autologous HSCT | 40 (27.6) | N/A | |
| Allogeneic-related | 85 (58.6) | N/A | |
| Allogeneic-unrelated | 20 (13.8) | N/A | |
| Disease status at HSCT | |||
| First complete remission (CR1) | 85 (58.6) | N/A | |
| ≥ CR1 | 50 (34.5) | N/A | |
| Conditioning | |||
| Chemotherapy | 34 (23.4) | N/A | |
| Chemotherapy + TBI | 111 (76.6) | N/A | |
| Chronic GVHD* | |||
| No | 71 (67.6) | N/A | |
| Resolved | 20 (19.0) | N/A | |
| Active | 14 (13.3) | N/A |
Percentages are determined from the total number of participants who provided data for each variable, rather than on total number in each cohort.
BMTSS indicates Bone Marrow Transplant Survivor Study; CCSS, Childhood Cancer Survivor Study; N/A, not applicable; and TBI, total body irradiation.
Limited to allogeneic HSCT (N = 105).
Among the BMTSS survivors, 72.4% had undergone allogeneic HSCT (58.6% from related donor, 13.8% from unrelated donor); 58.6% were in CR1 at HSCT; TBI was used as conditioning for 76.6%; 32.4% of allogeneic HSCT recipients reported a history of chronic GVHD; of these, 41.1% reported active chronic GVHD at study participation. Median time from HSCT to study participation was 11.0 years (range, 2.3-25.9 years).
Prevalence of chronic health conditions and adverse health status
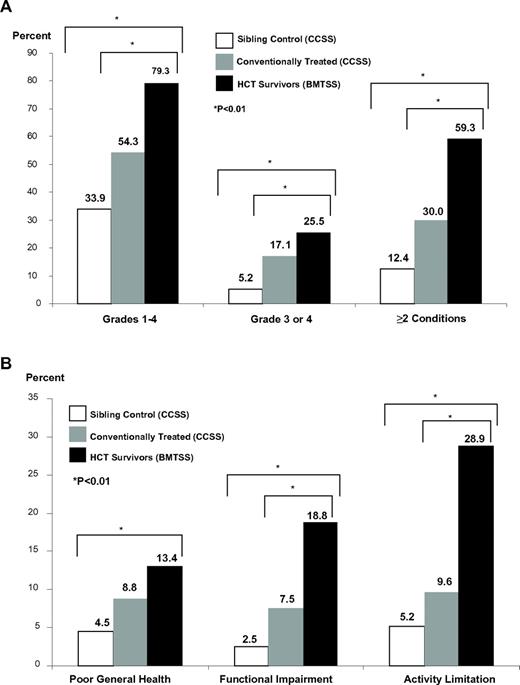
The prevalence and severity of chronic health conditions (Figure 1A) and adverse health status (Figure 1A) were greater for BMTSS patients compared with CCSS patients and the sibling comparison groups across nearly all domains. Seventy-nine percent of BMTSS patients reported at least one chronic health condition (grades 1-4); 59.3% had 2 or more health conditions; and 25.5% reported a severe/life-threatening condition (grade 3 or 4). Thirteen percent of BMTSS patients reported poor general health, 18.8% had functional impairment, and 28.9% had activity limitation. The prevalence of functional impairment (23.3%) and activity limitation (36.7%) was greatest among allogeneic HSCT recipients with active chronic GVHD. There were no differences in the prevalence and severity of chronic health conditions among BMTSS participants who underwent HSCT in CR1 compared with more advanced disease (data not shown).
Health-related outcomes. (A) Prevalence of chronic health conditions among HSCT survivors (BMTSS) compared with conventionally treated cancer survivors (CCSS) and sibling controls (CCSS). (B) Prevalence of adverse health status among HSCT survivors (BMTSS) compared with conventionally treated cancer survivors (CCSS) and sibling controls (CCSS).
Health-related outcomes. (A) Prevalence of chronic health conditions among HSCT survivors (BMTSS) compared with conventionally treated cancer survivors (CCSS) and sibling controls (CCSS). (B) Prevalence of adverse health status among HSCT survivors (BMTSS) compared with conventionally treated cancer survivors (CCSS) and sibling controls (CCSS).
Predictors of chronic health conditions and adverse health status
BMTSS versus siblings.
After adjustment for age at study participation, sex, and ethnicity, BMTSS patients were significantly more likely than the sibling comparison group to report chronic health conditions (Table 3), ie, grades 1-4: RR = 2.6 (95% CI, 2.4-2.9); grades 3-4: RR = 8.1 (95% CI, 6.2-10.5); ≥ 2 conditions: RR = 5.7 (95% CI, 4.9-6.7). Furthermore, BMTSS patients were significantly more likely to report compromised health status, ie, adverse general health: RR = 3.2 (95% CI, 2.0-4.9); functional impairment: RR = 7.7 (95% CI, 5.1-11.5); and activity limitation: RR = 6.3 (95% CI, 4.7-8.5). Unrelated donor HSCT recipients were at the greatest risk, ie, grades 1-4: RR = 3.5 (95% CI, 2.7-4.6); grades 3-4: RR = 13.5 (95% CI, 6.9-26.5); ≥ 2 conditions: RR = 8.9 (95% CI, 5.9-13.4), functional impairment: RR = 8.4 (95% CI, 2.8-24.9); activity limitation: RR = 10.1 (95% CI, 5.4-18.8).
Relative risk of chronic health conditions and adverse health status among HSCT survivors (BMTSS), as compared with sibling controls (CCSS)
| . | Grades 1-4 (95% CI) . | Grade 3 or 4 (95% CI) . | ≥ 2 conditions (95% CI) . | Poor general health (95% CI) . | Functional impairment (95% CI) . | Activity limitation (95% CI) . |
|---|---|---|---|---|---|---|
| Siblings (CCSS) | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 |
| BMTSS | 2.6 (2.4-2.9) | 8.1 (6.2-10.5) | 5.7 (4.9-6.7) | 3.2 (2.0-4.9) | 7.7 (5.1-11.5) | 6.3 (4.7–8.5) |
| P | < .01 | < .01 | < .01 | < .01 | < .01 | < .01 |
| Donor source, BMTSS* | ||||||
| Autologous | 2.5 (2.0-3.1) | 6.5 (3.7-11.6) | 4.7 (3.2-6.8) | 3.5 (1.5-7.9) | 6.9 (3.2-14.8) | 6.2 (3.6–10.9) |
| Allogeneic, related | 2.5 (2.3-2.9) | 8.0 (5.9-10.8) | 5.7 (4.8-8.9) | 3.2 (1.9-5.4) | 7.8 (4.9-12.6) | 5.9 (4.0–8.5) |
| Allogeneic, unrelated | 3.5 (2.7-4.6) | 13.5 (6.9-26.5) | 8.9 (5.9-13.4) | 2.0 (0.3-13.3) | 8.4 (2.8-24.9) | 10.1 (5.4–18.8) |
| P value (trend) | < .01 | < .01 | < .01 | < .01 | < .01 | < .01 |
| . | Grades 1-4 (95% CI) . | Grade 3 or 4 (95% CI) . | ≥ 2 conditions (95% CI) . | Poor general health (95% CI) . | Functional impairment (95% CI) . | Activity limitation (95% CI) . |
|---|---|---|---|---|---|---|
| Siblings (CCSS) | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 |
| BMTSS | 2.6 (2.4-2.9) | 8.1 (6.2-10.5) | 5.7 (4.9-6.7) | 3.2 (2.0-4.9) | 7.7 (5.1-11.5) | 6.3 (4.7–8.5) |
| P | < .01 | < .01 | < .01 | < .01 | < .01 | < .01 |
| Donor source, BMTSS* | ||||||
| Autologous | 2.5 (2.0-3.1) | 6.5 (3.7-11.6) | 4.7 (3.2-6.8) | 3.5 (1.5-7.9) | 6.9 (3.2-14.8) | 6.2 (3.6–10.9) |
| Allogeneic, related | 2.5 (2.3-2.9) | 8.0 (5.9-10.8) | 5.7 (4.8-8.9) | 3.2 (1.9-5.4) | 7.8 (4.9-12.6) | 5.9 (4.0–8.5) |
| Allogeneic, unrelated | 3.5 (2.7-4.6) | 13.5 (6.9-26.5) | 8.9 (5.9-13.4) | 2.0 (0.3-13.3) | 8.4 (2.8-24.9) | 10.1 (5.4–18.8) |
| P value (trend) | < .01 | < .01 | < .01 | < .01 | < .01 | < .01 |
Model adjusted for age at the time of the study, sex, and race or ethnicity.
BMTSS indicates Bone Marrow Transplant Survivor Study; CCSS, Childhood Cancer Survivor Study; CI, confidence interval; and N/A, not applicable.
Referent group: sibling controls (CCSS).
BMTSS versus CCSS.
As shown in Table 4, BMTSS survivors were more likely to report chronic health conditions, ie, grades 1-5: RR = 1.5 (95% CI, 1.1-2.2), grades 3-5: RR = 3.9 (95% CI, 2.4-5.4), and multiple health conditions: RR = 2.6 (95% CI, 1.7-3.8), compared with the CCSS survivors. The risk was especially high for unrelated donor HSCT recipients, ie, grades 1-5: RR = 1.7 (95% CI, 1.2-2.4); grades 3-5: RR = 6.8 (95% CI, 3.1-14.9); and multiple conditions: RR = 3.4 (95% CI, 2.1-5.6). Conditioning with TBI was associated with an increased risk of grade 3-5 (RR = 4.1; 95% CI, 2.8-6.1, P < .01) and multiple (RR = 2.4; 95% CI, 1.2-4.8, P = .01) health conditions. The presence of active chronic GVHD did not add to the magnitude of risk for grade 3-5 conditions (GVHD, active: RR = 3.0; 95% CI, 1.0-7.8, P = .04; GVHD, resolved: RR = 4.1; 95% CI, 2.2-7.9, P < .01) or multiple health conditions (GVHD, active: RR = 3.5; 95% CI, 2.4-5.1, P < .01; GVHD, resolved: RR = 3.5; 95% CI, 2.5-5.0, P < .01).
Relative risk of chronic health conditions and adverse health status among HSCT survivors (BMTSS), as compared with conventionally treated cancer survivors (CCSS)
| . | Grades 1-5 (95% CI) . | Grade 3-5 (95% CI) . | ≥ 2 conditions (95% CI) . | Poor general health (95% CI) . | Functional impairment (95% CI) . | Activity limitation (95% CI) . |
|---|---|---|---|---|---|---|
| Conventionally treated (CCSS) | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 |
| BMTSS | 1.5 (1.1-2.2) | 3.9 (2.4-6.4) | 2.6 (1.7-3.8) | 2.8 (1.8-6.8) | 3.5 (1.8-6.8) | 5.8 (3.2-10.5) |
| P | < .01 | < .01 | < .01 | < .01 | < .01 | < .01 |
| Donor source, BMTSS* | ||||||
| Autologous | 1.3 (1.0-1.8) | 3.0 (1.6-5.6) | 1.8 (1.1-2.8) | 3.3 (1.2-9.6) | 2.7 (0.8-9.1) | 3.1 (1.2-8.2) |
| Allogeneic, related | 1.6 (1.4-1.9) | 4.1 (2.7-6.3) | 2.8 (2.1-3.7) | 2.7 (1.3-5.6) | 3.7 (1.9-7.0) | 6.9 (3.7-12.8) |
| Allogeneic, unrelated | 1.7 (1.2-2.4) | 6.8 (3.1-14.9) | 3.4 (2.1-5.6) | 1.7 (0.2-20.2) | 4.1 (0.7-24.6) | 9.5 (3.5-25.5) |
| P value (trend) | < .01 | < .01 | < .01 | .05 | < .01 | < .01 |
| . | Grades 1-5 (95% CI) . | Grade 3-5 (95% CI) . | ≥ 2 conditions (95% CI) . | Poor general health (95% CI) . | Functional impairment (95% CI) . | Activity limitation (95% CI) . |
|---|---|---|---|---|---|---|
| Conventionally treated (CCSS) | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 |
| BMTSS | 1.5 (1.1-2.2) | 3.9 (2.4-6.4) | 2.6 (1.7-3.8) | 2.8 (1.8-6.8) | 3.5 (1.8-6.8) | 5.8 (3.2-10.5) |
| P | < .01 | < .01 | < .01 | < .01 | < .01 | < .01 |
| Donor source, BMTSS* | ||||||
| Autologous | 1.3 (1.0-1.8) | 3.0 (1.6-5.6) | 1.8 (1.1-2.8) | 3.3 (1.2-9.6) | 2.7 (0.8-9.1) | 3.1 (1.2-8.2) |
| Allogeneic, related | 1.6 (1.4-1.9) | 4.1 (2.7-6.3) | 2.8 (2.1-3.7) | 2.7 (1.3-5.6) | 3.7 (1.9-7.0) | 6.9 (3.7-12.8) |
| Allogeneic, unrelated | 1.7 (1.2-2.4) | 6.8 (3.1-14.9) | 3.4 (2.1-5.6) | 1.7 (0.2-20.2) | 4.1 (0.7-24.6) | 9.5 (3.5-25.5) |
| P value (trend) | < .01 | < .01 | < .01 | .05 | < .01 | < .01 |
Model adjusted for: age at the time of the study, sex, race or ethnicity, health insurance, treatment era, time from diagnosis, underlying diagnosis, radiation (brain, chest), and chemotherapy (anthracycline, alkylating agent, platinum agent, epidophyllotoxin).
BMTSS indicates Bone Marrow Transplant Survivor Study; CCSS, Childhood Cancer Survivor Study; and CI, confidence interval.
Referent group: conventionally treated cancer survivors (CCSS).
Compared with CCSS survivors, BMTSS survivors were significantly more likely to report compromised health status, ie, adverse general health (RR = 2.8; 95% CI, 1.8-6.8); functional impairment (RR = 3.5; 95% CI, 1.8-6.8; and activity limitation (RR = 5.8; 95% CI, 3.2-10.5). Unrelated donor HSCT recipients were at the greatest risk for functional impairment and activity limitation but not so for adverse general health. Allogeneic HSCT recipients with active chronic GVHD at study participation were at greatest risk for poor general health (RR = 7.9; 95% CI, 3.0-21.0), functional impairment (RR = 19.5; 95% CI, 8.0-47.4), activity impairment (RR = 14.8; 95% CI, 6.8-32.1), and pain because of cancer or its treatment (RR = 5.0; 95% CI, 1.2-21.9), compared with CCSS survivors. Health status was comparable between those with resolved chronic GVHD and those with no history of GVHD.
Specific chronic health conditions
Table 5 presents the prevalence and relative risk of grades 3-5 heath conditions among BMTSS patients compared with CCSS patients, according to organ-system involvement. Of note, endocrine complications were found in 29.7% of BMTSS patients, compared with 4.9% of the CCSS patients; nearly half (47.6%) of female BMTSS patients reported gonadal failure, compared with 4% of the female CCSS patients (P < .01).
Prevalence and relative risk of common chronic health conditions among HSCT survivors (BMTSS), as compared with conventionally treated cancer survivors (CCSS) and sibling controls (CCSS)
| . | Relative risk grade 3-5 conditions . | Relative risk grade 3-4 conditions . | |||
|---|---|---|---|---|---|
| BMTSS (N = 145), % . | Conventionally treated, CCSS (N = 7207), % . | RR (95% CI)* . | Sibling control, CCSS (N = 4020), % . | RR (95% CI)† . | |
| Second malignant neoplasm‡ | 6.9 | 3.1 | 8.6 (2.9-25.3) | 0.6 | 14.5 (7.1-29.8) |
| Endocrine | 29.7 | 4.9 | 7.7 (4.2-14.3) | 1.3 | 29.6 (21.0-41.8) |
| Musculoskeletal | 2.1 | 0.5 | 7.4 (2.4-23.1) | 0.05 | 76.5 (11.0-531.3) |
| Gastrointestinal | 2.8 | 2.0 | 4.8 (1.0-21.7) | 0.4 | 10.4 (3.5-31.1) |
| Neurosensory impairment | 9.0 | 3.9 | 3.8 (1.4-10.3) | 1.4 | 6.6 (3.6-12.1) |
| Genitourinary | 1.4 | 0.3 | 2.9 (1.1-7.8) | 0.05 | 26.4 (3.5-196.2) |
| Cardiovascular | 4.8 | 3.2 | 0.5 (0.1-2.5) | 0.5 | 12.7 (5.4-30.0) |
| . | Relative risk grade 3-5 conditions . | Relative risk grade 3-4 conditions . | |||
|---|---|---|---|---|---|
| BMTSS (N = 145), % . | Conventionally treated, CCSS (N = 7207), % . | RR (95% CI)* . | Sibling control, CCSS (N = 4020), % . | RR (95% CI)† . | |
| Second malignant neoplasm‡ | 6.9 | 3.1 | 8.6 (2.9-25.3) | 0.6 | 14.5 (7.1-29.8) |
| Endocrine | 29.7 | 4.9 | 7.7 (4.2-14.3) | 1.3 | 29.6 (21.0-41.8) |
| Musculoskeletal | 2.1 | 0.5 | 7.4 (2.4-23.1) | 0.05 | 76.5 (11.0-531.3) |
| Gastrointestinal | 2.8 | 2.0 | 4.8 (1.0-21.7) | 0.4 | 10.4 (3.5-31.1) |
| Neurosensory impairment | 9.0 | 3.9 | 3.8 (1.4-10.3) | 1.4 | 6.6 (3.6-12.1) |
| Genitourinary | 1.4 | 0.3 | 2.9 (1.1-7.8) | 0.05 | 26.4 (3.5-196.2) |
| Cardiovascular | 4.8 | 3.2 | 0.5 (0.1-2.5) | 0.5 | 12.7 (5.4-30.0) |
BMTSS indicates Bone Marrow Transplant Survivor Study; CCSS, Childhood Cancer Survivor Study; CI, confidence interval; and RR, relative risk.
Model adjusted for age at the time of the study, sex, race or ethnicity, health insurance, treatment era, time from diagnosis, underlying diagnosis, radiation (brain, chest), and chemotherapy (anthracycline, alkylating agent, platinum agent, epidophyllotoxin).
Model adjusted for age at the time of the study, sex, and race or ethnicity.
For all groups, this category excludes basal cell and squamous cell carcinoma (grade 2). For siblings, this category includes a first cancer.
Compared with CCSS patients, BMTSS patients had a markedly increased risk of developing a severe/life-threatening complication across nearly all organ systems, with the greatest risk observed for subsequent malignant neoplasms, followed by endocrinopathy and musculoskeletal impairment. Compared with siblings, BMTSS patients had a significantly elevated risk of health conditions across all organ systems, with the risk ranging from 6.6-fold (neurosensory impairment) to 76.5-fold (musculoskeletal) that of the siblings.
Of note, female BMTSS patients were at a 9.3-fold increased risk (95% CI, 4.0-28.1, P < .01) of ovarian failure compared with CCSS survivors, and at a 39.3-fold increased risk (95% CI, 26.0-59.8, P < .01) compared with sibling controls.
Discussion
The overall goals of this study were to understand the long-term burden of morbidity associated with HSCT in childhood; compare this burden of morbidity with that reported by an unaffected comparison group; and evaluate the burden of morbidity among HSCT survivors that was over and above that associated with children treated with conventional therapy. The burden of morbidity in this study was defined as the cumulative burden because of chronic health conditions related to therapeutic exposures. By taking advantage of the similarities in study design and questionnaires between the CCSS and BMTSS, we were able to demonstrate that unrelated donor recipients were at greatest risk for all adverse health-related outcomes and that conditioning with TBI and active chronic GVHD were critical modifiers of risk of chronic health conditions and health status, respectively.
The burden of chronic health conditions among children treated with HSCT is high, ie, 79.3% reported at least one chronic health condition; 25.5% had severe/life-threatening conditions; and nearly 60% had 2 or more conditions. In contrast, 5.2% of siblings reported severe/life-threatening conditions, and 12.4% had 2 or more conditions. Compared with sibling controls, BMTSS patients were 2.6 times as likely to report a chronic health condition of any severity; the risk for severe/life-threatening condition was 8.1-fold increased, and the risk was 5.7-fold increased for having 2 or more conditions. Although the risk of adverse health status was elevated across all domains, the cohort was at the greatest risk for functional impairment. This highlights the increasingly recognized need for specialized, multidisciplinary, long-term follow-up care that incorporates screening and surveillance for survivors of childhood HSCT.21-23
To evaluate the risk for adverse health-related outcomes that was above and beyond that because of conventional therapy, detailed pre-HSCT therapeutic information was obtained for BMTSS patients and was included in the adjusted comparison with CCSS patients. As with the comparison with siblings, BMTSS patients were at increased risk of chronic health conditions across all domains, and TBI was a modifier of this risk.24 Unrelated donor HSCT recipients had an especially high risk, with a nearly 7-fold risk for severe/life-threatening conditions.
Despite the high burden of chronic disease, unrelated HSCT recipients rated their overall health as equivalent to CCSS survivors. Yet, when asked specifically about functional impairment or activity limitations, unrelated donor HSCT recipients were the most vulnerable. This observation supports previously published findings that general inquiries regarding overall health may fail to identify the extent of compromise in health status and that in-depth assessments may be needed to evaluate limitations in functional and physical health for vulnerable sub-populations of HSCT survivors.25-28
Chronic GVHD contributed significantly to the increased risk of adverse health status across nearly all domains. Specifically, BMTSS patients with active chronic GVHD were at an especially high risk of functional and physical impairment. However, the health status in survivors with resolved chronic GVHD was equivalent to those who had never been diagnosed with GVHD, a finding that has been previously reported among adult long-term HSCT survivors.18 These findings are encouraging and suggest that the residual impact of GVHD on adverse health status is likely to be minimal after resolution of GVHD.29 However, the high burden of grade 3-5 or multiple (≥ 2) health conditions among BMTSS survivors, irrespective of GVHD status (active vs resolved), underscores the need for continued improvement in strategies for management of GVHD in this high-risk population.
HSCT conditioning and transplant-related complications can modify the risk of organ compromise3,7,30-37 Nearly one-third of BMTSS patients in the current study reported grades 3-5 endocrine complications, with 1 in 4 reporting gonadal failure; the prevalence of gonadal failure approached 50% for female BMTSS survivors. This finding is in contrast to female CCSS patients, where the prevalence was only 4%. BMTSS patients did not appear to be at increased risk of cardiovascular disease, compared with the CCSS patients. However, BMTSS patients had a 13-fold increase in risk for cardiovascular complications compared with sibling controls. One possible explanation for this finding is that, as seen in previous studies,38,39 the risk for late-occurring cardiovascular complications after HSCT may be largely because of pre-HSCT therapeutic exposures, with little additional risk from conditioning-related exposures or GVHD. Our regression analysis had adjusted for the relevant pre-HSCT therapeutic exposures (anthracyclines, chest radiation), potentially tempering the risk for cardiovascular complications among BMTSS survivors, compared with the CCSS survivors.
Both BMTSS and CCSS groups rely on self-report for measuring chronic health conditions. Thus, this study summarizes the prevalence of and risk factors for chronic health conditions diagnosed by our health care system and communicated to cancer survivors and their siblings and is therefore a reflection of the patient's health insurance coverage, access to care, awareness among the health care providers of the risk of long-term complications, and communication of these outcomes to their patients. The authors of previous studies have shown excellent correlation between medical records and self-reported outcomes, including GVHD,17,18 and we feel confident that self-report can be used effectively to describe post-treatment complications that are diagnosed as part of routine healthcare delivery.10,13,16,40
Detailed therapeutic exposures were abstracted from medical records, including exposures before HSCT for the BMTSS cohort. For the BMTSS cohort, we recognize that there may be differences in the extent of pre-HSCT treatment because a proportion of the HSCT survivors underwent transplantation shortly after induction therapy, whereas for others, HSCT was offered after relapse of their primary malignancy. As a result, all relevant pre-HSCT therapeutic information (chemotherapy and radiation) was included in the regression model in addition to treatment era, diagnosis, disease status at HSCT, length of follow-up, and sociodemographics comparing the burden of morbidity between BMTSS and CCSS survivors. Even with adjustment for these factors, the potential could exist for residual confounding, which may influence the results. However, to describe the contribution of HSCT to the overall disease burden, we were careful to maximize comparability in the inclusion criteria between the CCSS (5+ year survivors from diagnosis) and BMTSS cohort (survival of 5+ years from diagnosis; HSCT within 3 years of diagnosis; an additional 2+ years of follow-up after HSCT). This possibly limits the generalizability of the absolute burden of morbidity experienced by HSCT survivors to those with the aforementioned inclusion criteria but ensures that the primary goals for the study were addressed.
Finally, we acknowledge that treatment-related exposures for the 2 cohorts may not reflect the recent therapeutic options available for children undergoing HSCT41,42 or receiving conventional therapy.43-45 That being said, the increasing reliance on alternative stem cell sources for HSCT coupled with better integration of risk-adapted therapy for conventionally treated patients,2,42 make it likely that the magnitude of risk for adverse health-related outcomes will continue to remain greater for HSCT survivors. Future studies with adequate lengths of follow-up will be able to evaluate the modifying effect of contemporary treatment strategies on the long-term burden of treatment-related chronic disease.
Keeping these issues in mind, we believe this study provides a global assessment of the burden of morbidity in survivors of childhood HSCT and finds that, compared with conventionally treated cancer patients and sibling controls, HSCT survivors are at increased risk for adverse health-related outcomes. Unrelated HSCT recipients are at greatest risk; conditioning with TBI and active chronic GVHD are critical modifiers of risk of chronic health conditions, and adverse health status, respectively. Taken together, this study identifies vulnerable subpopulations for targeted surveillance, as well as multidisciplinary management of patients with chronic GVHD.
Presented in part at the 52nd annual meeting of the American Society of Hematology, Orlando, FL, December 4-7, 2010.
An Inside Blood analysis of this article appears at the front of this issue.
The online version of this article contains a data supplement.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Acknowledgments
This study was supported in part by grants from the National Institutes of Health (R01 CA078938, to S.B.; P01 CA 30206, to S.J.F.; U24-CA-55727, to L.L.R.), Leukemia & Lymphoma Society Scholar Award for Clinical Research (2191-02, to S.B.), and the American Lebanese Syrian Associated Charities (St Jude Children's Research Hospital).
National Institutes of Health
Authorship
Contribution: S.H.A. designed the research, collected and assembled the data, analyzed and interpreted the data, and wrote the paper; C.-L.S. and F.L.W. analyzed and interpreted the data; T.K., W.L., and C.A.S. designed the research and collected andassembled data; L.F., J.B.T., G.M., and L.R.D. collected and assembled the data; M.A., K.S.B., J.R., and S.J.F. provided study patients and collected and assembled data; M.M.H. and K.C.O. designed the research, provided study materials, and contributed to the writing of the paper; L.L.R. is the principal investigator of the CCSS, designed the research, analyzed and interpreted the data, and contributed to the writing of the paper; and S.B. is the principal investigator of the BMTSS, designed the research, analyzed and interpreted the data, and contributed to the writing of the paper.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Smita Bhatia, MD, MPH, City of Hope, 1500 E Duarte Rd, Duarte, CA 91010-3000; e-mail: sbhatia@coh.org.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal