Abstract
The mechanisms of hematopoietic progenitor cell egress and clinical mobilization are not fully understood. Herein, we report that in vivo desensitization of Sphingosine-1-phosphate (S1P) receptors by FTY720 as well as disruption of S1P gradient toward the blood, reduced steady state egress of immature progenitors and primitive Sca-1+/c-Kit+/Lin− (SKL) cells via inhibition of SDF-1 release. Administration of AMD3100 or G-CSF to mice with deficiencies in either S1P production or its receptor S1P1, or pretreated with FTY720, also resulted in reduced stem and progenitor cell mobilization. Mice injected with AMD3100 or G-CSF demonstrated transient increased S1P levels in the blood mediated via mTOR signaling, as well as an elevated rate of immature c-Kit+/Lin− cells expressing surface S1P1 in the bone marrow (BM). Importantly, we found that S1P induced SDF-1 secretion from BM stromal cells including Nestin+ mesenchymal stem cells via reactive oxygen species (ROS) signaling. Moreover, elevated ROS production by hematopoietic progenitor cells is also regulated by S1P. Our findings reveal that the S1P/S1P1 axis regulates progenitor cell egress and mobilization via activation of ROS signaling on both hematopoietic progenitors and BM stromal cells, and SDF-1 release. The dynamic cross-talk between S1P and SDF-1 integrates BM stromal cells and hematopoeitic progenitor cell motility.
Introduction
Motility is a key feature of hematopoietic stem and progenitor cells (HSPCs). These cells are continuously released at basal levels from the bone marrow (BM) reservoir to the circulation during steady state homeostasis together with maturing leukocytes, and at increased rates on stress situations, such as bleeding or inflammation.1,2 The complex process of HSPC trafficking is orchestrated by various cytokines, chemokines, proteolytic enzymes, and adhesion molecules3-5 through a dynamic interplay between the immune and nervous systems within the bone microenvironment.1,2,6-8 HSPC mobilization can be clinically induced by a variety of cytokines and drugs, such as granulocyte colony stimulating factor (G-CSF, the most commonly used agent),9,10 sulfated polysaccharides,11,12 and recently also by AMD3100.13,14 Repetitive G-CSF administrations cause mobilization by inducing proliferation and differentiation of HSPC, thus increasing their pool size, accompanied by reduced retention in the BM microenvironment.15 The chemokine stromal cell–derived factor-1 (SDF-1, also termed CXCL12), which is the most powerful chemoattractant of both human and murine HSPCs,16,17 and its major receptor CXCR4 are key players in HSPC mobilization.10,12,13,18-21 SDF-1 is transiently increased in the murine BM during G-CSF stimulation followed by its down-regulation at both protein18,22 and mRNA23 levels, reaching a nadir at the peak of HSPC mobilization.18 The intensified SDF-1/CXCR4 interactions induce enhanced production of reactive oxygen species (ROS) through activation of the HGF/c-Met pathway, further facilitating HSPC motility.24 In contrast to G-CSF, AMD3100 is a rapid mobilizing agent that is administrated only once.10 This agent enhances CXCR4-dependent HSPC recruitment to the circulation by triggering SDF-1 secretion from BM CXCR4+ stromal cells followed by its release from the BM to the circulation, leading to urokinase plasminogen activator (uPA), MMP-9, and reactive oxygen species (ROS) activation.13,25-27
Sphingosine-1-phosphate (S1P) is a bioactive lipid implicated in many biologic processes, including cell migration, survival, proliferation, and angiogenesis, as well as immune and allergic responses.28 S1P is generated from sphingosine by sphingosine kinases (Sphks), and can either be converted back to sphingosine by specific S1P phosphatases (Sgpps), or degraded by S1P lyase (Sgpl).29 Although most cells can synthesize S1P, its levels are very high in the blood and lymph circulations compared with solid tissues, because of its production by mature red blood cells29-31 and activated platelets.32 S1P stimulates distinct pathways such as Rho GTPase, phospholipase C, Ras, MAP kinase, and PI3K signaling pathways depending on the expression patterns of its receptors.31 One of these receptors, S1P1, is involved in lymphocyte egress from lymphoid organs32,33 as well as from the BM34,35 into circulatory fluids via a gradient of S1P. Interestingly, S1P can directly act as a chemoattractant for HSPCs in a dose-dependent manner.36,37 Accordingly, HSPC egress from extramedullary tissues depends on S1P1 up-regulation and migration toward higher S1P concentrations in the lymph and blood circulations.38 Intriguingly, the HSPC chemotactic activity of the plasma was almost completely abolished after inactivation of bioactive lipids present in the plasma, suggesting a crucial role for S1P as a chemoattractant for BM-residing HSPCs.37 Accordingly, S1P was shown to regulate HSPC AMD3100 and G-CSF–induced mobilization,37,39 although no mechanism was yet described. Notably, S1P also induces chemotaxis of osteoclast precursors to the circulation demonstrating an osteoporotic phenotype in S1P1 conditional deficient mice because of increased retention of mature osteoclasts adjacent to the bone surface.40 Recent publications show elevated S1P levels during stress, such as irradiation leading to HSPC homing.41
In this study, we demonstrated that inhibition of the S1P/S1P1 axis reduced steady-state egress as well as rapid AMD3100-induced and repeated G-CSF–induced progenitor cell mobilization. In addition, S1P levels were transiently increased in the circulation during AMD3100 and G-CSF–induced mobilization, accompanied by enhanced levels of progenitors expressing S1P1 in the BM via mTOR signaling. S1P induced SDF-1 secretion from stromal cells including Nestin+ mesenchymal stem cells (MSCs), and in turn SDF-1 was released from the BM to the circulation, thus further regulating progenitor cell mobilization. The elevated S1P levels in the murine BM led to enhanced ROS production in both primitive hematopoietic Sca-1+/c-Kit+/Lin− (SKL) cells, thus enhancing their migratory potential, and in BM stromal cells leading to SDF-1 secretion. These dynamic changes induce progenitor cells to pave their way from the BM into the circulation, as part of host defense and repair mechanisms.
Methods
Mice and in vivo protocols
All experiments in mice were approved by the institutional animal care and use committee (IACUC) of the Weizmann Institute. C57BL/6 and B6.SJL mice were purchased from Harlan. Sphk1 knockout (KO) mice and S1P1loxP/loxP conditional KO mice (both on C57BL/6 background) were kindly provided by Prof Richard L. Proia (National Institutes of Health). S1P1loxP/loxP conditional KO mice were crossed with Mx1-Cre mice (The Jackson Laboratory) and we used S1P1loxP/loxP/Mx1-Cre+ (S1P1 KO) and S1P1loxP/loxP/Mx1-Cre− (wild-type [WT] counterparts). Mice were mobilized by G-CSF (Filgastrim, Amgen; 300 μg/kg) as previously described,24 or by AMD3100 (Sigma-Aldrich; 5 mg/kg) as previously described.25 The inhibitor of S1P receptors, FTY720 (Cayman Chemicals), was injected intraperitoneally (1 mg/kg) alone for 1.5 hours or 24 hours before sacrificing the mice, together with AMD3100 (30 minutes before AMD3100 administration) or together with G-CSF (once a day for 5 days). The inhibitor of S1P lyase activity, 4-deoxypyridoxine (DOP, Sigma-Aldrich) was administrated to mice in the drinking water (30 mg/L) together with glucose (10 g/L) for 3 days and compared with control mice, which received glucose alone. Rapamaycin (Wyeth Europa), the inhibitor of mTOR, was subcutaneously injected (480 μg/kg) and Akt inhibitor (Triciribine; Calbiochem) was intraperitoneally injected (1 mg/kg), both coadministrated with G-CSF once a day for 5 days as indicated. White blood cell (WBC) counts in the circulation were determined after red blood cell lysis.
Colony-forming assay
Peripheral blood (PB) mononuclear cells were isolated by Ficoll separation and were seeded (2 × 105 cells/mL) in semisolid cultures as previously described.18 Colonies, reflecting colony-forming units cells (CFU-C), were scored 7 days after plating and presented as CFU-C per 1 milliliter of blood.
Flow cytometry
Phenotypes of murine cells were determined by flow cytometry analysis in FACSCalibur (BD Biosciences) using CellQuest Pro Version 5.2 software (BD Biosciences), as previously described.42 Levels of SKL cells were determined as previously described.24 Expression of S1P1 on BM cells was detected using polyclonal rabbit anti–mouse S1P1 Ab (Abcam) followed by donkey anti–rabbit-PE Ab (Jackson ImmunoResearch Laboratories). Signaling lymphocyte activation molecule (SLAM) staining was performed with anti-CD41/CD48 (eBioscience) and anti-CD150 (eBioscience). The fluorescence of oxidized hydroethidine (Molecular Probes), which is the probe for ROS detection24 as well as CD34− (eBioscience) detection on SKL cells were determined by LSRII Flow Cytometer (BD Biosciences) and analyzed by FACSDiva 6.1.3 (BD Biosciences). Staining was performed at 4°C for 30 minutes followed by washing with phosphate-buffered saline (PBS).
Immunofluorescence staining of Nestin-GFP cells
Bones from Nestin-GFP knockin mice,43 kindly provided by Prof Grigori Enikolopov (Cold Spring Harbor Laboratory, NY), were flushed with freshly prepared medium: αMEM (Sigma-Aldrich), supplemented with 20% preselected fetal calf serum (FCS), 5% of l-glutamine/penicillin/streptomycin, and 5mM sodium-pyruvate (Biologic Industries). Total BM cells were seeded in 24-well plates (Falcon) with cover slips inside for 4-7 days with no medium change. The cells were pretreated with either: 1μM S1P (Cayman Chemicals) or NaOH (vehicle) for 1 hour, 1μM FTY720 (Cayman Chemicals) for 1 hour or for 30 minutes before AMD3100 addition, 1mM buthionine-sulfoximine (BSO; Sigma-Aldrich) for 2 hours, 100 ng/mL AMD3100 for 1 hour, or 2mM NAC (N-acetyl-L-cysteine, Sigma-Aldrich) together with S1P. The cells were fixated by 4% paraformaldehyde and permeabilized by triton after staining with anti–SDF-1 (K15C ab INRA, Paris). Stained Nestin-GFP cells were visualized and photographed using an Olympus BX51 microscope and Olympus DP71 camera at a magnitude of ×60.
Statistical analysis
Statistical significance was determined by 2-tailed Student t test using Excel X software (Microsoft).
Additional methods are listed in supplemental Methods (available on the Blood Web site; see the Supplemental Materials link at the top of the online article).
Results
Hematopoietic progenitor cell egress is regulated by the chemotactic lipid S1P
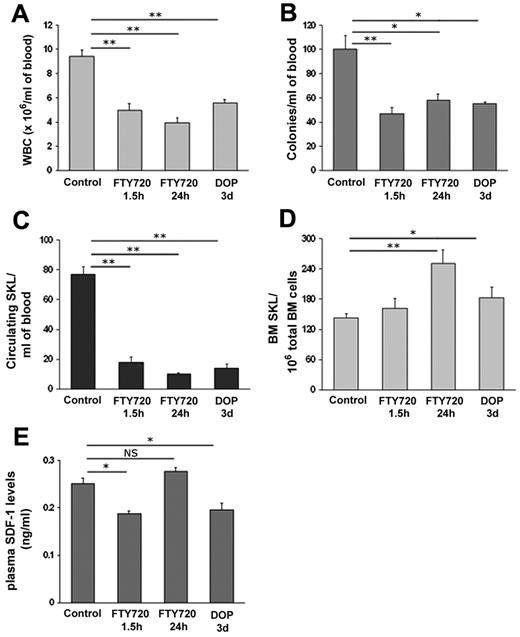
To identify the role of S1P and its receptors in the homeostatic release of hematopoietic progenitor cells (HPCs), 2 different strategies were applied. To desensitize S1P receptors, mice were injected with FTY720, which was previously shown to internalize most S1P receptors, including S1P1 in leukocytes.44 To interfere with the gradient of S1P between the blood and different tissues, mice were treated with 4-deoxypyridoxine (DOP), which inhibits the activity of the degrading enzyme, S1P lyase, and therefore increases S1P concentrations in the BM (supplemental Figure 1), but not in the blood.45 We observed a significant decrease in WBC counts in the peripheral blood (PB) of C57BL (Figure 1A) and B6/SJL (supplemental Figure 2A) mice 1.5 hours and 24 hours after a single injection of FTY720 or after 3 days of treatment with DOP in the drinking water (Figure 1A, supplemental Figure 2A). In accordance, reduced numbers of immature CFU-C (Figure 1B, supplemental Figure 2B) as well as primitive SKL cells (Figure 1C, supplemental Figure 2C) were detected in the PB of these mice. To further investigate reduced HPC egress by interfering with the S1P/S1P1 axis, we analyzed SKL cells in the BM and found increased levels in mice treated either with FTY720 for 24 hours or with DOP and only a very slight increase in mice treated with FTY720 for 1.5 hours (Figure 1D) along with unchanged cell cycle status (supplemental Figure 3). SDF-1 release from the BM to the circulation was previously shown to guide the way of progenitors to the blood during homeostasis and rapid AMD3100-induced mobilization25,27 and high circadian SDF-1 levels in the BM correlate with reduced homeostatic HSPC egress.8 Therefore, we examined functional SDF-1 levels in the plasma of mice and observed reduced levels in mice treated with DOP for 3 days or FTY720 for 1.5 hours (Figure 1E), correlating with decreased HPC egress and suggesting diminished release of SDF-1 from the BM to the blood. Although the egress of HPC continued to be reduced, we found increased functional SDF-1 levels in the plasma after treatment with FTY720 for 24 hours (Figure 1E), suggesting involvement of additional signaling pathways such as serine protease uPA, ROS, and BM functional SDF-1 levels, which were revealed and are discussed later in this paper. Together, these results indicate that the S1P axis plays a substantial role in steady state egress of HPCs from the BM to the circulation via SDF-1 release.
Hematopoietic progenitor cell egress is regulated by the chemotactic lipid S1P. (A-E) Mice were injected with PBS or administered with glucose in the drinking water (control), injected with FTY720 (single intraperitoneal injection 1.5 hours and 24 hours before sacrifice) or administered with DOP (together with glucose for 3 days in the drinking water). The numbers of WBC (A), CFU-C (B) and primitive SKL cells (C) per milliliter of blood were analyzed. BM SKL numbers per 1 × 106 total BM cells (D) are shown. SDF-1 ELISA levels in the plasma (E) are determined. Indicated values are presented as mean ± SE in 3 independent experiments, a total of 6 mice in each group (*P < .05, **P < .01).
Hematopoietic progenitor cell egress is regulated by the chemotactic lipid S1P. (A-E) Mice were injected with PBS or administered with glucose in the drinking water (control), injected with FTY720 (single intraperitoneal injection 1.5 hours and 24 hours before sacrifice) or administered with DOP (together with glucose for 3 days in the drinking water). The numbers of WBC (A), CFU-C (B) and primitive SKL cells (C) per milliliter of blood were analyzed. BM SKL numbers per 1 × 106 total BM cells (D) are shown. SDF-1 ELISA levels in the plasma (E) are determined. Indicated values are presented as mean ± SE in 3 independent experiments, a total of 6 mice in each group (*P < .05, **P < .01).
The S1P axis regulates AMD3100-induced rapid mobilization of HPCs via enhanced SDF-1 release
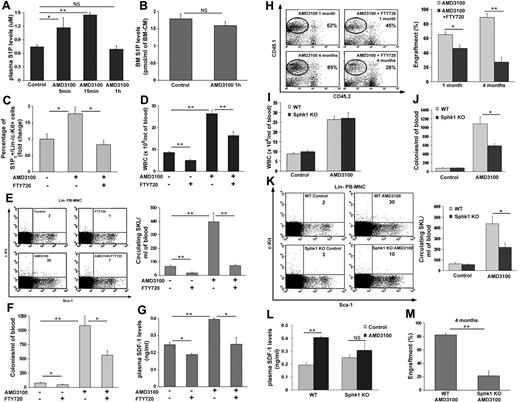
Next, we tested S1P regulation of rapid AMD3100-induced mobilization, and measured the concentration of S1P in the plasma of AMD3100 mobilized mice as well as the changes in S1P1 expression. We found transiently increased S1P levels in the plasma 5 minutes and an even higher increase 15 minutes after AMD3100 injection (Figure 2A), which were back to baseline in the plasma after 1 hour of AMD3100 administration (Figure 2A). At 1 hour after AMD3100 injection, S1P levels in the BM were also normal (Figure 2B). Moreover, an increase in the percentage of S1P1 expressing BM c-Kit+/Lin− (KL) progenitor cells 1 hour after AMD3100 treatment was detected, which was blocked by cotreatment with AMD3100 and FTY720 (Figure 2C). This combined treatment was accompanied by decreased WBCs (Figure 2D) as well as primitive SKL cell numbers (Figure 2E), and CFU-C (Figure 2F) in the circulation, thus correlating with the decreased expression of S1P1 on progenitor cells in the BM. Because AMD3100 induces SDF-1 secretion in the BM and increases its release to the blood,25,27 we investigated the cross-talk between S1P/S1P1 and enhanced SDF-1 release during rapid AMD3100-induced mobilization. We found that coadministration of AMD3100 and FTY720 decreased functional SDF-1 levels in the plasma 1 hour after AMD3100 treatment (Figure 2G) compared with mice treated only with AMD3100. Of interest, we detected no changes between AMD3100 and AMD3100 and FTY720 cotreated mice in the ability of BM-MNCs to migrate toward a gradient of SDF-1 in vitro (supplemental Figure 4). To further investigate the reduced progenitor rapid mobilization on blockage of S1P receptors, we transplanted whole blood from AMD3100 or AMD3100 and FTY720 treated donors and found decreased short and long term repopulation levels in recipients of AMD3100 and FTY720 pretreated blood compared with AMD3100 alone (Figure 2H). To further determine the involvement of S1P in rapid mobilization, we used a model of Sphingosine kinase 1 genetically deficient mice (Sphk1 KO) with reduced levels of S1P in the circulation,46 thus mimicking low S1P conditions. During steady-state, Sphk1 KO mice share differences in several hematopoietic populations but have no changes in BM SKL levels compared with their WT counterparts (supplemental Table 1). Sphk1 KO mice were therefore treated with AMD3100, and although no difference in the numbers of WBCs in the PB was observed (Figure 2I), we found lower numbers of immature CFU-C (Figure 2J) and primitive SKL cells (Figure 2K) in the blood in comparison with AMD3100-treated WT mice. Moreover, the rates of SDF-1 elevation in the plasma were lower in AMD3100-treated versus untreated Sphk1 KO mice compared with AMD3100-treated versus untreated WT mice (Figure 2L). Finally, we found significantly reduced long-term repopulation levels of whole blood from Sphk1 KO mice compared with WT mice, both pretreated with AMD3100 (Figure 2M). These results reveal involvement of the S1P axis in rapid HPC mobilization by increasing SDF-1 release from the BM to the plasma.
The S1P/S1P1 axis regulates rapid AMD3100-induced mobilization of progenitor cells via enhanced SDF-1 release. (A) S1P ELISA plasma levels in PBS (control) and AMD3100 injected mice (single subcutaneous injection and evaluation after 5 minutes, 15 minutes or 1 hour) are shown. (B) S1P BM concentration in PBS (control) and AMD3100 injected mice (single subcutaneous injection and evaluation after 1 hour) are presented. (C-H) Mice were injected with PBS (control), FTY720 (single intraperitoneal injection and evaluation after 1.5 hours), AMD3100 (single subcutaneous injection and evaluation after 1 hour) or AMD3100+FTY720 (single intraperitoneal injection of FTY720 30 minutes before a single subcutaneous injection of AMD3100 and evaluation 1 hour after AMD3100 injection). The percentage of BM S1P1+/c-Kit+/Lin− cells (C) is shown. The numbers of WBCs (D), primitive SKL cells (E) Left, representative FACS plots of Lin− PB-MNC. The numbers in the top right quadrant of each plot represent SKL cells per 1 × 106 PB-MNC. (E) Right, total PB-MNC cell analysis and CFU-C (F) per milliliter blood are shown. Plasma SDF-1 ELISA levels (G) and the level of donor chimerism 1 month and 4 months after transplantation of whole pretreated blood (H) Left, representative FACS plots of total BM cells. The numbers in the top right quadrant of each plot represent % of engraftment. (H) Right, total engraftment analysis are presented. (I-M) WT and Sphk1 deficient mice (Sphk1 KO) were injected with PBS (control) or AMD3100 (single subcutaneous injection and evaluation after 1 hour). The numbers of WBCs (I), CFU-Cs (J), and primitive SKL cells (K) Left, representative FACS plots of Lin− PB-MNC. The numbers in the top right quadrant of each plot represent SKL cells per 1 × 106 PB-MNC. (K) Right, total PB-MNC cell analysis per milliliter of blood were analyzed. Plasma SDF-1 ELISA levels (L) and the level of donor chimerism 4 months after transplantation of whole pretreated blood (M) are shown. Indicated values are presented as mean ± SE in 3 independent experiments, a total of 6 mice in each group. Repopulation experiments were performed with a total of 4-7 mice per each group (*P < .05, **P < .01).
The S1P/S1P1 axis regulates rapid AMD3100-induced mobilization of progenitor cells via enhanced SDF-1 release. (A) S1P ELISA plasma levels in PBS (control) and AMD3100 injected mice (single subcutaneous injection and evaluation after 5 minutes, 15 minutes or 1 hour) are shown. (B) S1P BM concentration in PBS (control) and AMD3100 injected mice (single subcutaneous injection and evaluation after 1 hour) are presented. (C-H) Mice were injected with PBS (control), FTY720 (single intraperitoneal injection and evaluation after 1.5 hours), AMD3100 (single subcutaneous injection and evaluation after 1 hour) or AMD3100+FTY720 (single intraperitoneal injection of FTY720 30 minutes before a single subcutaneous injection of AMD3100 and evaluation 1 hour after AMD3100 injection). The percentage of BM S1P1+/c-Kit+/Lin− cells (C) is shown. The numbers of WBCs (D), primitive SKL cells (E) Left, representative FACS plots of Lin− PB-MNC. The numbers in the top right quadrant of each plot represent SKL cells per 1 × 106 PB-MNC. (E) Right, total PB-MNC cell analysis and CFU-C (F) per milliliter blood are shown. Plasma SDF-1 ELISA levels (G) and the level of donor chimerism 1 month and 4 months after transplantation of whole pretreated blood (H) Left, representative FACS plots of total BM cells. The numbers in the top right quadrant of each plot represent % of engraftment. (H) Right, total engraftment analysis are presented. (I-M) WT and Sphk1 deficient mice (Sphk1 KO) were injected with PBS (control) or AMD3100 (single subcutaneous injection and evaluation after 1 hour). The numbers of WBCs (I), CFU-Cs (J), and primitive SKL cells (K) Left, representative FACS plots of Lin− PB-MNC. The numbers in the top right quadrant of each plot represent SKL cells per 1 × 106 PB-MNC. (K) Right, total PB-MNC cell analysis per milliliter of blood were analyzed. Plasma SDF-1 ELISA levels (L) and the level of donor chimerism 4 months after transplantation of whole pretreated blood (M) are shown. Indicated values are presented as mean ± SE in 3 independent experiments, a total of 6 mice in each group. Repopulation experiments were performed with a total of 4-7 mice per each group (*P < .05, **P < .01).
Dynamic changes in S1P levels in the BM and PB regulate G-CSF–induced mobilization
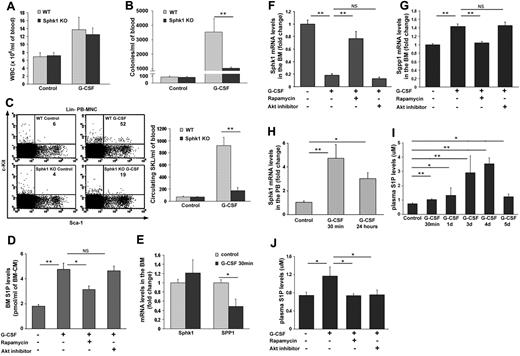
Next, we examined G-CSF–induced mobilization in Sphk1 and S1P1 deficient mice as well as in combination with FTY720 administration. G-CSF was injected to Sphk1 KO mice and although there was no difference in mature WBC numbers (Figure 3A), we observed a significant reduction in immature CFU-C (Figure 3B) as well as in primitive SKL cells (Figure 3C) in the circulation compared with their WT counterparts. These results suggest that S1P is involved in HPC mobilization induced by G-CSF and indeed we found elevated S1P concentrations in the BM during G-CSF administration (Figure 3D). To decipher the mechanism underlying these changes, we examined the PI3K pathway. Previously, we have shown that inhibition of mTOR by Rapamycin reduced G-CSF–induced HPC mobilization.24,47 We found that injection of Rapamycin together with G-CSF blocked the increase in BM S1P levels (Figure 3D). Interestingly, inhibition of the upstream activator of mTOR in the PI3K pathway, namely Akt, had no impact on G-CSF–mediated BM S1P elevation (Figure 3D). Nevertheless, mice treated with G-CSF together with an Akt inhibitor also showed abrogated mobilization (data not shown). These results suggest that the increase in S1P levels in the BM is mTOR-dependent and Akt-independent. Because it is known that S1P levels are regulated by sphingosine kinases (Sphk) and sphingosine phosphatases (Sgpp),29 we measured their transcription levels 30 minutes after a single G-CSF injection and found increased Sphk1 (Figure 3E) and decreased Sgpp1 (Figure 3E) levels in total BM cells, suggesting an overall increase of S1P levels in the BM shortly after G-CSF administration. However, measuring transcription levels of these enzymes after 5 days of G-CSF administration revealed decreased Sphk1 (Figure 3F) and increased Sgpp1 (Figure 3G) levels, which were blocked by cotreatment with Rapamycin, but not with Akt inhibitor (Figure 3F-G). These results suggest a negative feedback loop responsible for restoring the normal levels of S1P in the BM. S1P plasma levels were previously reported to be augmented after G-CSF–induced mobilization.37 Accordingly, we observed increased levels of Sphk1 mRNA in PB MNC 30 minutes after a single G-CSF injection, which were still higher but to a lower extent after 24 hours (Figure 3H). These results suggest increased plasma S1P levels, although S1P is mainly produced by erythrocytes and platelets in the blood.29-31 To further characterize changes in S1P plasma levels during G-CSF administration, we measured its levels at several time points and found an increase as soon as 30 minutes after a single G-CSF injection with a peak at day 4 of consecutive G-CSF administrations (Figure 3I). Interestingly, although high S1P levels were detected at day 5 of G-CSF administrations compared with untreated mice, they were still much lower than on day 4 (Figure 3I), thus hinting again on the existence of a negative feedback loop to restore S1P normal levels also in the plasma. In accordance, we found that S1P levels in the blood are increased in an Akt/mTOR-dependent manner (Figure 3J). Our results suggest that the dynamic changes in BM and plasma S1P levels during G-CSF administration contribute to HPC mobilization.
Dynamic changes in S1P concentrations in the BM and PB regulate G-CSF–induced mobilization. (A-C) WT and Sphk1 deficient mice were injected with PBS (control) or G-CSF (5 consecutive subcutaneous injections and evaluation 3-5 hours after the last injection). The numbers of WBCs (A), CFU-Cs (B), and primitive SKL (C). Left, representative FACS plots of Lin− PB-MNC. The numbers in the top right quadrant of each plot represent SKL cells per 1 × 106 PB-MNC.Right, total PB-MNC cell analysis per milliliter of blood are shown. (D,F,G,J) Mice were injected with PBS (control), G-CSF (5 consecutive subcutaneous injections and evaluation 3-5 hours after the last injection), G-CSF+Rapamycin (5 consecutive subcutaneous injections of both agents) or G-CSF+Akt inhibitor (5 consecutive intraperitoneal injections together with G-CSF). MS quantification of S1P levels in the BM condition medium (BM-CM; D) is shown. (E) Quantitative Real-Time PCR analysis of Sphk1 (S1P producing enzyme) and Sgpp1 (S1P degrading enzyme) mRNA levels in PBS (control) or G-CSF (single subcutaneous injection and evaluated after 30 minutes) total BM cells are presented. Quantitative Real-Time PCR analysis of Sphk1 (F) and Sgpp1 (G) mRNA levels in total BM cells are shown. (H) Quantitative Real-Time PCR analysis of Sphk1 mRNA levels in mice treated with PBS (control) or G-CSF (single subcutaneous injection and evaluated after 30 minutes or 24 hours) in PB-MNC are presented. (I) Plasma S1P ELISA levels in PBS (control) or G-CSF (single subcutaneous injection and evaluated after 30 minutes or 4 hours and 3, 4, or 5 consecutive subcutaneous injections and evaluation 3-5 hours after the last injection) are presented. (J) Plasma S1P ELISA levels are shown. Indicated values are presented as mean ± SE in 3 independent experiments, a total of 6 mice in each group (*P < .05, **P < .01).
Dynamic changes in S1P concentrations in the BM and PB regulate G-CSF–induced mobilization. (A-C) WT and Sphk1 deficient mice were injected with PBS (control) or G-CSF (5 consecutive subcutaneous injections and evaluation 3-5 hours after the last injection). The numbers of WBCs (A), CFU-Cs (B), and primitive SKL (C). Left, representative FACS plots of Lin− PB-MNC. The numbers in the top right quadrant of each plot represent SKL cells per 1 × 106 PB-MNC.Right, total PB-MNC cell analysis per milliliter of blood are shown. (D,F,G,J) Mice were injected with PBS (control), G-CSF (5 consecutive subcutaneous injections and evaluation 3-5 hours after the last injection), G-CSF+Rapamycin (5 consecutive subcutaneous injections of both agents) or G-CSF+Akt inhibitor (5 consecutive intraperitoneal injections together with G-CSF). MS quantification of S1P levels in the BM condition medium (BM-CM; D) is shown. (E) Quantitative Real-Time PCR analysis of Sphk1 (S1P producing enzyme) and Sgpp1 (S1P degrading enzyme) mRNA levels in PBS (control) or G-CSF (single subcutaneous injection and evaluated after 30 minutes) total BM cells are presented. Quantitative Real-Time PCR analysis of Sphk1 (F) and Sgpp1 (G) mRNA levels in total BM cells are shown. (H) Quantitative Real-Time PCR analysis of Sphk1 mRNA levels in mice treated with PBS (control) or G-CSF (single subcutaneous injection and evaluated after 30 minutes or 24 hours) in PB-MNC are presented. (I) Plasma S1P ELISA levels in PBS (control) or G-CSF (single subcutaneous injection and evaluated after 30 minutes or 4 hours and 3, 4, or 5 consecutive subcutaneous injections and evaluation 3-5 hours after the last injection) are presented. (J) Plasma S1P ELISA levels are shown. Indicated values are presented as mean ± SE in 3 independent experiments, a total of 6 mice in each group (*P < .05, **P < .01).
Elevated BM S1P1 levels during G-CSF–induced mobilization
We found that the expression of the S1P1 receptor was increased by 2.5-fold on BM-MNCs of mice treated with G-CSF compared with control mice (Figure 4A). As shown in Figure 4B, G-CSF administration is also accompanied by a 4 fold increase in the percentage of S1P1+/c-Kit+/Lin− cells in the BM that is reduced on cotreatment with G-CSF and FTY720. In light of these results, we hypothesized that analogous to S1P, increased expression of S1P1 during G-CSF–induced mobilization might be regulated via the mTOR pathway. Indeed, concomitant administration of Rapamycin (but not Akt inhibitor) during G-CSF treatment abolished the increase in the percentage of S1P1+/c-Kit+/Lin− cells in the BM (Figure 4B). Coadministration of FTY720 and G-CSF for 5 consecutive days, resulted in reduced numbers of WBCs (Figure 4C), decrease in CFU-C as well as primitive SKL cell (Figure 4D) numbers in the blood compared with G-CSF treatment alone. To further investigate the reduced SKL cell mobilization by coadministration of G-CSF and FTY720, we analyzed primitive SKL cells in the BM and found increased levels in mice treated with both agents compared with mice treated with G-CSF alone (Figure 4E). Finally, to examine the role of S1P1, the major S1P receptor in HPC mobilization, we used a model of hematopoietic-specific S1P1 conditional deficient mice40 (S1P1 KO). S1P1 KO and WT mice were treated with either AMD3100 or G-CSF and though no difference in the numbers of WBC in the PB was observed (Figure 4F), we found a reduction in primitive SKL cell (Figure 4G) and immature CFU-C (Figure 4H) numbers in the blood in both treatments separately. These findings clearly show that repetitive G-CSF stimulations resulted in enhanced BM levels of HPC expressing S1P1 that presumably migrate toward the higher S1P levels in the blood as part of the mobilization process.
Elevated BM S1P1 levels during G-CSF–induced mobilization. (A-E) Mice were injected with PBS (control), G-CSF (5 consecutive subcutaneous injections and evaluation 3-5 hours after the last injection), G-CSF+FTY720 (5 consecutive intraperitoneal injections of both agents), G-CSF+Rapamycin (5 consecutive subcutaneous injections of both agents) or G-CSF+Akt inhibitor (5 consecutive intraperitoneal injections together with G-CSF). A representative histogram (A left) and a total cell analysis (A right) of S1P1 receptor expression on BM MNC are presented. The percentage of S1P1+/c-Kit+/Lin− cells in the BM: (Bi) representative FACS plots of Lin− BM-MNC with the percentage of positive cells from 1 experiment written in each plot and (Bii) total BM-MNC cell analysis are shown. The numbers of WBCs (C), primitive SKL cells (D bars) and CFU-Cs (D dots) per milliliter of blood are shown. BM SKL numbers per 1 × 106 total BM cells (E) are presented. (F-H) WT and S1P1 conditional deficient mice (S1P1 KO) were injected with PBS (control), AMD3100 (single subcutaneous injection and evaluation after 1 hour) or G-CSF (5 consecutive subcutaneous injections and evaluation 3-5 hours after the last injection). The numbers of WBCs (F), primitive SKL cells (G), and CFU-Cs (H) per milliliter of blood are shown. Indicated values are presented as mean ± SE in 3 independent experiments, a total of 6 mice in each group (*P < .05, **P < .01).
Elevated BM S1P1 levels during G-CSF–induced mobilization. (A-E) Mice were injected with PBS (control), G-CSF (5 consecutive subcutaneous injections and evaluation 3-5 hours after the last injection), G-CSF+FTY720 (5 consecutive intraperitoneal injections of both agents), G-CSF+Rapamycin (5 consecutive subcutaneous injections of both agents) or G-CSF+Akt inhibitor (5 consecutive intraperitoneal injections together with G-CSF). A representative histogram (A left) and a total cell analysis (A right) of S1P1 receptor expression on BM MNC are presented. The percentage of S1P1+/c-Kit+/Lin− cells in the BM: (Bi) representative FACS plots of Lin− BM-MNC with the percentage of positive cells from 1 experiment written in each plot and (Bii) total BM-MNC cell analysis are shown. The numbers of WBCs (C), primitive SKL cells (D bars) and CFU-Cs (D dots) per milliliter of blood are shown. BM SKL numbers per 1 × 106 total BM cells (E) are presented. (F-H) WT and S1P1 conditional deficient mice (S1P1 KO) were injected with PBS (control), AMD3100 (single subcutaneous injection and evaluation after 1 hour) or G-CSF (5 consecutive subcutaneous injections and evaluation 3-5 hours after the last injection). The numbers of WBCs (F), primitive SKL cells (G), and CFU-Cs (H) per milliliter of blood are shown. Indicated values are presented as mean ± SE in 3 independent experiments, a total of 6 mice in each group (*P < .05, **P < .01).
S1P directly induces SDF-1 secretion from BM Nestin+ stromal precursors
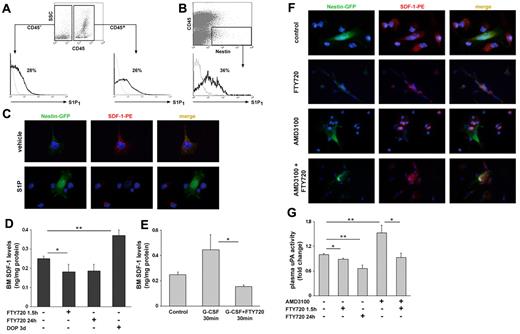
Because we found that inhibition of S1P receptors by FTY720 reduces SDF-1 release to the blood during AMD3100 administration (Figure 2G), we went on to study S1P regulation of SDF-1 secretion from BM stromal cells in vitro. We found that S1P1 is expressed by CD45+ hematopoietic as well as by CD45− stromal cells in the BM (Figure 5A). Several nonhematopoietic BM stromal cell types were shown to participate in HSC regulation and in particular Nestin+ reticular MSCs.48 These supportive stromal cells form BM niches for primitive SLAM+ HSPCs, are located near BM sinuses in contact with primitive hematopoietic cells and exhibit the highest levels of SDF-1.48 Importantly, we found that BM Nestin+/CD45− MSCs also express cell surface S1P1 (Figure 5B). To examine whether S1P induces SDF-1 secretion from stromal BM cells, we seeded total BM cells from Nestin-GFP mice in cultures and pretreated them with S1P or its vehicle. We found that S1P treatment directly induced SDF-1 secretion from Nestin+ MSCs in vitro, because of the lack of intracellular SDF-1 content in S1P, but not vehicle treated cells (Figure 5C). Accordingly, we examined functional BM SDF-1 levels in mice treated with DOP or FTY720 and observed elevated BM SDF-1 levels in DOP mice, whereas reduced levels in FTY720 (1.5 hours and 24 hours) treated mice (Figure 5D). To study the role of S1P in SDF-1 secretion during mobilization, we tested BM SDF-1 levels shortly after a single G-CSF administration, because we previously published that each G-CSF injection causes a transient increase in BM SDF-1 levels.18 We detected a milder increase in SDF-1 BM levels 30 minutes after coadministration of G-CSF and FTY720 compared with G-CSF alone (Figure 5E), which suggests involvement of S1P in SDF-1 secretion during G-CSF–induced mobilization. To test a role for S1P also in SDF-1 secretion during rapid mobilization, we treated total BM cells from Nestin-GFP mice in vitro with FTY720, AMD3100 or both. As shown in Figure 5F, AMD3100 but not FTY720 eliminated SDF-1 intracellular content, whereas cotreatment of AMD3100 and FTY720 blocked the secretion of intracellular SDF-1 content from Nestin+ MSCs. We have previously shown that AMD3100-induced mobilization is dependent on uPA activation by SDF-1.25 Therefore, we examined S1P regulation of uPA and found that desensitization of S1P receptors by FTY720 reduced uPA (Figure 5G) activity and cotreatment of AMD3100 and FTY720 reduced AMD3100-induced activation of uPA (Figure 5G). Together, these results reveal that S1P directly induces in vitro SDF-1 secretion and release from BM stromal cells, including Nestin+ MSCs, thus activating circulating uPA that participate in rapid HSPC mobilization.
S1P induces SDF-1 secretion from BM Nestin+ stromal stem and progenitor cells during mobilization. (A) A representative histogram of the percentage of S1P1 positive cells within the CD45+ and CD45− populations in steady-state conditions. (B) A representative histogram of the percentage of S1P1 positive cells within the Nestin+/CD45− population in steady-state conditions. (C) Immunofluorescence staining of total BM cells from Nestin-GFP mice for SDF-1 expression on stimulation with 1μM S1P/vehicle for 1 hour. (D,G) Mice were injected with PBS or administered with glucose in the drinking water (control), injected with FTY720 (single intraperitoneal injection 1.5 hours and 24 hours before sacrifice) or administered with DOP (together with glucose for 3 days in the drinking water). (D) SDF-1 ELISA levels in BM fluids are shown. (E) SDF-1 ELISA levels in the BM fluids of PBS (control), G-CSF (single subcutaneous injection and evaluated after 30 minutes) or G-CSF+FTY720 (coinjection of subcutaneous G-CSF and intraperitoneal FTY720 and evaluated after 30 minutes) mice are presented. (F) Immunofluorescence staining of total BM cells from Nestin-GFP mice for SDF-1 expression on stimulation with PBS, 100 ng/mL AMD3100 for 1 hour, 1μM FTY720 for 1.5 hours alone or 30 minutes before AMD3100 addition. (G) The levels of uPA activity are shown. Indicated values are presented as mean ± SE in 2-3 independent experiments, a total of 4-6 mice in each group (*P < .05, **P < .01).
S1P induces SDF-1 secretion from BM Nestin+ stromal stem and progenitor cells during mobilization. (A) A representative histogram of the percentage of S1P1 positive cells within the CD45+ and CD45− populations in steady-state conditions. (B) A representative histogram of the percentage of S1P1 positive cells within the Nestin+/CD45− population in steady-state conditions. (C) Immunofluorescence staining of total BM cells from Nestin-GFP mice for SDF-1 expression on stimulation with 1μM S1P/vehicle for 1 hour. (D,G) Mice were injected with PBS or administered with glucose in the drinking water (control), injected with FTY720 (single intraperitoneal injection 1.5 hours and 24 hours before sacrifice) or administered with DOP (together with glucose for 3 days in the drinking water). (D) SDF-1 ELISA levels in BM fluids are shown. (E) SDF-1 ELISA levels in the BM fluids of PBS (control), G-CSF (single subcutaneous injection and evaluated after 30 minutes) or G-CSF+FTY720 (coinjection of subcutaneous G-CSF and intraperitoneal FTY720 and evaluated after 30 minutes) mice are presented. (F) Immunofluorescence staining of total BM cells from Nestin-GFP mice for SDF-1 expression on stimulation with PBS, 100 ng/mL AMD3100 for 1 hour, 1μM FTY720 for 1.5 hours alone or 30 minutes before AMD3100 addition. (G) The levels of uPA activity are shown. Indicated values are presented as mean ± SE in 2-3 independent experiments, a total of 4-6 mice in each group (*P < .05, **P < .01).
S1P regulates HPC mobilization and SDF-1 secretion from BM stromal cells, including Nestin+ MSCs, via ROS signaling
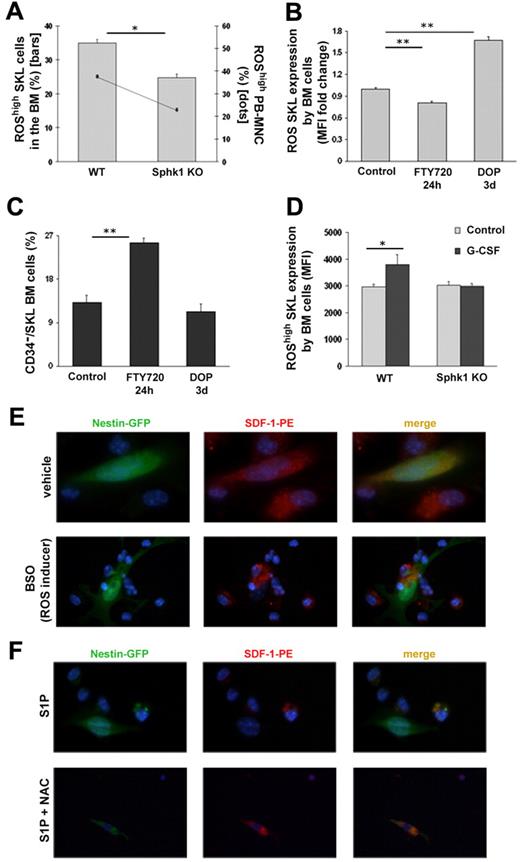
Next, we focused on ROS, which was previously shown to be elevated and essential for G-CSF–induced mobilization and to increase HSPC motility.24 We found that Sphk1 KO mice have lower levels of ROShigh/SKL cells in the BM (Figure 6A), but not lower intensity of homeostatic ROS (Figure 6D) as well as lower levels of ROShigh/ MNC cells in the plasma (Figure 6A). Correspondingly, desensitization of S1P receptors by FTY720 24 hours administration reduced ROS expression by primitive BM SKL cells (Figure 6B). Moreover, increasing BM S1P levels by DOP led to enhanced ROS levels in primitive BM SKL cells (Figure 6B) and reduced egress of HPCs (Figure 1A-D), most probably because of the increased levels of SDF-1 and S1P in the BM of these mice (Figure 5D, supplemental Figure 1). ROShigh expression by HPCs was shown to correlate with enhanced motility and myeloid differentiation, whereas ROSlow expression in these cells is related to their quiescent state and higher long repopulating ability.49,50 We studied the levels of CD34−/SKL cells, previously determined to be quiescent long term repopulating cells, on desensitization of S1P receptors expression or increased S1P BM levels. As shown in Figure 6C, the percentage of CD34−/SKL cells was increased on treatment with FTY720 and decreased on administration of DOP, corresponding to the levels of ROShigh/SKL cells in the BM (Figure 6B). Collectively, these results suggest that the more quiescent primitive cells in the BM are less motile because of lower levels of ROS activity. Interestingly, G-CSF–induced mobilization in Sphk1 KO mice was not accompanied by elevated ROShigh expression levels in primitive SKL cells compared with WT mice (Figure 6D). Because inhibition of ROS signaling by NAC during AMD3100-induced mobilization resulted in reduced SDF-1 release to the plasma,25 we hypothesized that ROS induces SDF-1 secretion from BM stromal cells. Therefore, we treated total BM cells from Nestin-GFP mice in vitro with the ROS inducer BSO and found no SDF-1 intracellular content compared with its vehicle, suggesting that ROS signaling induced SDF-1 secretion (Figure 6E). Consequently, in vitro treatment of total BM cells from Nestin-GFP mice with S1P resulted in SDF-1 secretion, which was blocked on cotreatment with S1P and NAC (Figure 6F). These observations reveal that S1P induced ROS signaling in both BM HPCs and BM stromal cells, resulting in increased HPC motility and increased SDF-1 secretion during mobilization, followed by its release from the BM to the blood circulation. Importantly, ROS elevation is essential for the mobilization process, as ROS inhibition by NAC reduces both G-CSF and AMD3100-induced HSPC mobilization,24,25 but its effect is insufficient because enhanced S1P gradient to the circulation is also required.
S1P regulates progenitor cell mobilization and SDF-1 secretion by Nestin+ stromal stem and progenitor cells via ROS signaling. (A) The percentages of ROShigh/SKL cells in the BM (bars) and ROShigh/MNC in the PB (dots) of WT and Sphk1 KO mice are shown. (B-C) Mice were injected with PBS or administered with glucose in the drinking water (control), injected with FTY720 (single intraperitoneal injection 24 hours before sacrifice) or administered with DOP (together with glucose for 3 days in the drinking water). Fold change of mean fluorescence intensity of ROS/SKL (B) or the percentage of CD34−/SKL (C) cells in the BM are shown. (D) Mean fluorescence intensity of ROShigh in SKL cells of WT and Sphk1 KO mice that were injected with PBS (control) or G-CSF (5 consecutive subcutaneous injections and evaluation 3-5 hours after the last injection) is shown. (E) Immunofluorescence staining of total BM cells from Nestin-GFP mice for SDF-1 expression on stimulation with 1mM BSO (ROS inducer)/vehicle for 2 hours. (F) Immunofluorescence staining of total BM cells from Nestin-GFP mice for SDF-1 expression on stimulation with 1μM S1P alone or together with 2mM NAC for 1 hour. Indicated values are presented as mean ± SE in 3 independent experiments, a total of 4-6 mice in each group (*P < .05, **P < .01).
S1P regulates progenitor cell mobilization and SDF-1 secretion by Nestin+ stromal stem and progenitor cells via ROS signaling. (A) The percentages of ROShigh/SKL cells in the BM (bars) and ROShigh/MNC in the PB (dots) of WT and Sphk1 KO mice are shown. (B-C) Mice were injected with PBS or administered with glucose in the drinking water (control), injected with FTY720 (single intraperitoneal injection 24 hours before sacrifice) or administered with DOP (together with glucose for 3 days in the drinking water). Fold change of mean fluorescence intensity of ROS/SKL (B) or the percentage of CD34−/SKL (C) cells in the BM are shown. (D) Mean fluorescence intensity of ROShigh in SKL cells of WT and Sphk1 KO mice that were injected with PBS (control) or G-CSF (5 consecutive subcutaneous injections and evaluation 3-5 hours after the last injection) is shown. (E) Immunofluorescence staining of total BM cells from Nestin-GFP mice for SDF-1 expression on stimulation with 1mM BSO (ROS inducer)/vehicle for 2 hours. (F) Immunofluorescence staining of total BM cells from Nestin-GFP mice for SDF-1 expression on stimulation with 1μM S1P alone or together with 2mM NAC for 1 hour. Indicated values are presented as mean ± SE in 3 independent experiments, a total of 4-6 mice in each group (*P < .05, **P < .01).
Discussion
Previous results have demonstrated that HSPCs from different extramedullary nonlymphatic tissues egress to the blood by up-regulating S1P1 expression, exerting chemotaxis toward higher concentrations of S1P in the circulation.38 Moreover, S1P was reported to induce in vitro chemotaxis of HSPCs via S1P136 and was characterized as a major chemoattractant in the plasma that directs HSPC egress37 and mobilization from the BM to the circulation.37,39 However, no mechanism was identified to explain how S1P regulates HSPC trafficking in vivo. In this study, we identified for the first time a cross talk between S1P and SDF-1 via ROS signaling, which is essential for HPC egress, AMD3100 and G-CSF–induced mobilization.
It has been shown in vitro that SDF-1 and S1P can act synergistically to increase primitive BM SKL cell migration51 and that overexpression of S1P1 on immature human CD34+ cells decreases their in vitro migration potential toward SDF-1 and in vivo homing because of reduced expression of CXCR4,52 implying a potential cross talk between S1P and SDF-1. Our data show that short-term inhibition of the S1P/S1P1 axis during steady state conditions or AMD3100 administration caused reduced release of SDF-1 to the plasma. We also identified S1P to directly induce SDF-1 secretion from BM stromal cells, including Nestin+ MSCs,48 which suggests that modulation of SDF-1 secretion reduced progenitor egress and mobilization on alteration of the S1P axis.
The PI3K/mTOR/Akt pathway was previously shown to be activated by S1P and its receptors and was demonstrated to have an essential role in G-CSF–induced mobilization,47 through activation of HGF/c-Met and mTOR pathways, leading to ROS production.24 Our data showed that S1P is required for ROS activation in primitive SKL cells thus leading to increased cell motility as previously published.24 Importantly, we demonstrated here for the first time that ROS signaling has an important role also in BM stromal cells, including Nestin+ MSCs, inducing SDF-1 secretion, consequently facilitating HPC egress and mobilization. Accordingly, our previous publication demonstrates that inhibition of ROS signaling by NAC reduces SDF-1 secretion during AMD3100-induced mobilization.25
In steady-state conditions, S1P levels are high in the circulation compared with other tissues29-31 ; however, there is controversy regarding S1P levels in the plasma during stress-induced mobilization. Although one study reports increased S1P plasma levels after G-CSF-induced mobilization,37 another study claims no changes in S1P plasma levels after 1 hour of AMD3100 injection or after 5 days of G-CSF–induced mobilization using a different protocol.39 Indeed, 1 hour after AMD3100 administration we also detected no changes in plasma S1P levels. Nevertheless, we observed a transient elevation in plasma S1P levels in mice as early as 5 minutes and higher levels 15 minutes after a single injection of AMD3100, which were restored to normal levels at 1 hour after the injections during the peak of HSPC mobilization. Moreover, we found augmented S1P plasma levels also after treatment with G-CSF, confirming previous observations.37 Interestingly, S1P plasma levels started rising as soon as 30 minutes after a single G-CSF administration, reaching a peak at day 4 of treatment. We postulate that the discrepancies between our observations and the ones from the recent publication are because of different protocols (we injected G-CSF for 5 days and killed mice 3-4 hours after the last injection in contrast to the other group, which injected it twice daily for 4 days and killed on the 5th day). Together, it seems that S1P plasma levels rose before progenitor cell egress, but are required for HSPC egress and mobilization as evident from various genetic models as well as FTY720 and DOP treatments. It has been shown that such elevations may be because of the activation of the complement cascade and interactions of the membrane attack complex (MAC) with erythrocytes.37 Nevertheless, the augmented S1P levels were transient and were restored to normal homeostatic levels with time. In addition, at the peak of G-CSF–induced mobilization, we still detected a 2-fold increase of S1P levels in the BM, similar to the 2-fold increase in the plasma at this time point. Therefore, because S1P concentrations in solid tissues are low compared with the blood and lymph circulations,29 we believe that the gradient toward the blood is maintained as in steady state and perhaps is even augmented at earlier time points of G-CSF administration.
Moreover, S1P role in G-CSF–induced mobilization of HSPCs is recently questionable.39 We agree that desensitization of S1P receptors by FTY720 does not reduce the frequency of HPC in the circulation (data not shown); however, total progenitor numbers are significantly reduced (Figure 4D).
Finally, our data reveal not only a cross talk between S1P and SDF-1, but also a requirement for an active role of both factors during progenitor cell egress and mobilization. Interestingly, we found that FTY720 treatment for 24 hours augmented SDF-1 plasma levels, although HPC egress continued to be reduced similar to 1.5 hours after FTY720 treatment. Examination of SDF-1 levels in the BM revealed reduction in levels of SDF-1 once again similar to 1.5 hours after FTY710 treatment, suggesting a different source of SDF-1 secretion, such as circulating platelets. Nevertheless, we examined additional signaling pathways and found reduced uPA activity and ROS signaling 24 hours after FTY720 treatment, which point out the requirement for S1P signaling in the process of HPC egress. Moreover, DOP treated mice, which have increased BM ROS levels, also exhibited reduced progenitor egress. We suggest that because of increased functional BM S1P and SDF-1 levels, progenitors are retained in the BM, resulting in reduced egress to the blood. These observations suggest that S1P and SDF-1 levels are both required for balanced HPC egress and mobilization, thus inhibition of one axis impacts on the other and therefore abrogate the whole process.
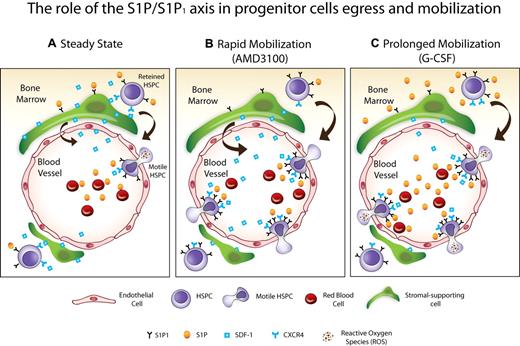
In conclusion, we demonstrated the dynamic regulation of a novel cross-talk involved in HPC trafficking. Our model suggests that the dynamic gradients of S1P between the blood and the BM in steady state egress and AMD3100-induced mobilization can recruit progenitors from the BM directly through chemotaxis and indirectly by SDF-1 secretion and release to the circulation (Figure 7). By creating WT into Sphk1 KO chimeric mice and vice versa, future studies which are beyond the scope of this paper, will reveal the direct contribution of S1P to HSPC egress and mobilization and indirect via BM stromal cells, which both functionally express S1P receptors. During G-CSF–induced mobilization, S1P levels in the circulation are transiently increased along with elevation in the number of BM S1P1 expressing progenitor cells. In addition, S1P and HGF24 activated ROS signaling in primitive BM SKL cells during G-CSF–induced mobilization to further increase their motility (Figure 7) and in stromal cells to facilitate SDF-1 secretion and release. In this study, we extended the paradigm of SDF-1 mediated egress and mobilization and added a new player with chemotactic abilities for HPC that has a cross talk with SDF-1 via activation of ROS signaling. Regulation of the S1P/S1P1 axis may be used to improve clinical mobilization and transplantation protocols.
Scheme of the proposed model. (A) Steady-state, the gradient of S1P between the PB and the BM facilitates progenitor egress via S1P1 mediated SDF-1 release. (B) Rapid mobilization (AMD3100), progenitor cells express elevated levels of S1P1 in the BM, which increases their mobilization via the S1P gradient toward the blood. In addition, S1P increases SDF-1 release from the BM to the PB, thus enhancing progenitor cell trafficking. (C) Prolonged mobilization (G-CSF), S1P levels are increased in the BM and PB along with augmentation of the S1P1 expressing progenitor cells. Moreover, ROS signaling is increased by S1P and HGF24 on primitive SKL cells, enhancing progenitor cell motility, and on BM stromal cells, inducing SDF-1 secretion and release. All these events facilitate progenitor cell mobilization.
Scheme of the proposed model. (A) Steady-state, the gradient of S1P between the PB and the BM facilitates progenitor egress via S1P1 mediated SDF-1 release. (B) Rapid mobilization (AMD3100), progenitor cells express elevated levels of S1P1 in the BM, which increases their mobilization via the S1P gradient toward the blood. In addition, S1P increases SDF-1 release from the BM to the PB, thus enhancing progenitor cell trafficking. (C) Prolonged mobilization (G-CSF), S1P levels are increased in the BM and PB along with augmentation of the S1P1 expressing progenitor cells. Moreover, ROS signaling is increased by S1P and HGF24 on primitive SKL cells, enhancing progenitor cell motility, and on BM stromal cells, inducing SDF-1 secretion and release. All these events facilitate progenitor cell mobilization.
The online version of this article contains a data supplement.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Acknowledgments
The authors thank Prof Richard L. Proia for kindly providing the Sphk1 genetically deficient (Sphk1 KO) and S1P1 conditional deficient (S1P1 KO) mice, and for fruitful discussions; Prof Grigori Enikolopov for kindly providing the Nestin-GFP mice; Vladislav Krupalnik, Giulia Caglio, and Dr Gabriele D'Uva for excellent technical assistance; and Dr Jonathan Canaani for critically reviewing the paper.
This study was supported in part by the Israeli Science Foundation (544/09) and the European Union (Advance Cell–based Therapies for the Treatment of Primary Immunodeficiency HEALTH-F5-2010-261387).
Authorship
Contribution: K.G. designed the research, designed and performed experiments, collected and analyzed the data, and wrote the paper; Y.V., A.L., T.I., and S.C.-G. helped with designing and performing in vivo experiments; A.K. designed, performed, and analyzed ELISA; O.K. helped with designing experiments and colony assays; C.K. performed in vivo experiments for the measurements of S1P in the BM sup; A.S. helped with in vitro experiments; Y.O., K.L., and S.S. performed in vivo experiments; A.J.M. performed MS measurements of S1P in BM sup; M.Z.R. critically revised the paper; and T.L. supervised the study, including experiment design and writing of the paper.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Tsvee Lapidot, Immunology Dept, The Weizmann Institute of Science, 234 Herzl Street, Rehovot 76100, Israel; e-mail: tsvee.lapidot@weizmann.ac.il.