Abstract
Acute myeloid leukemia (AML) with t(8;21) is a heterogeneous disease. The dynamic patterns of AML1-ETO transcript levels during treatment and clinical outcome of patients vary greatly. Our AML05 trial revealed that minimal residual disease (MRD)status and treatment strategy were independent risk factors for outcome and that risk stratification treatment directed by MRD may improve the outcome of t(8;21) AML in CR1 (Blood 2013; 121:4056). As for pretreatment parameters, c-KIT mutation is a well-established adverse predictor on survival. However, a subset of t (8;21) AML patients without c-KIT mutation still showed poor clinical outcome. The prognostic value of WT1 transcript levels at diagnosis in AML has been investigated and the results were controversial. We wondered if WT1 expression associated with outcome in t (8;21) AML patients.
A total of 101 patients were included. They all were eligible cases who enrolled into AML05 trial from June 2005 to December 2012, and had available bone marrow samples at diagnosis. After 1 or 2 induction therapy and 2 cycles of intermediate-dose cytarabine-based consolidation therapy, fifty-seven patients continued cytarabine-based consolidation chemotherapy or received autologous-hematopoietic stem-cell transplantation (auto-SCT) and were defined as CT group, the remaining 44 patients received allogeneic SCT (allo-SCT) and defined as SCT group.WT1 and ABL transcript were tested by real time quantitative PCR, and WT1 transcript levels were calculated as WT1copies/ABL copies in percentage. The upper limit of normal bone marrows (NBMs) was 0.5%. c-KIT mutations in exon 8 and 17 were screened by direct sequencing.
The median follow-up time was 25 (6-93) months for 73 alive patients. The cumulative incidence of relapse (CIR) at 3 years was 35.3%. The 3-year disease free survival (DFS) and overall survival (OS) rates were 57.2% and 62.8%, respectively. The median WT1 transcript levelsssof all patients were 9.1% (0.02%-99.3%). c-KIT mutation was detected in 31 patients. Receiver operating characteristics (ROC) curves revealed that WT1 transcript levels of 5.0% (1-log increase compared to the upper limit of NBM) were the best cutoff values to discriminate patients with different outcome. WT1 transcript levels of ≤5.0% were significantly associated with c-KIT mutation (23/42 vs 8/59, P<0.001), but didn't related to other pretreatment parameters (all P>0.05). In CT group, patients with WT1≤5% (n=19) had significantly higher CIR rate at 3-year, lower 3-year DFS and OS rate than those with WT1>5% (n=38), respectively (89.5% vs 27.9%, P<0.0001; 10.5% vs 66.1%, P<0.0001; 28.5% vs 66.5%, P=0.0062). Furthermore, patients with WT1≤5% and c-KIT mutation (-) had similar CIR, DFS and OS rate to both patients with WT1≤5% and c-KIT mutation (+) and patients with WT1>5% and c-KIT mutation (+) (all P >0.05, Figure 1). Therefore, they were merged into one group (n=24). Thus, patients with WT1≤5% and/or KIT mutation (+) had significantly higher CIR rate at 3-year, lower 3-year DFS and OS rate than those with WT1>5% and c-KIT mutation (-) (n=33) in CT group, respectively (89.4% vs 27.4%,P<0.0001; 10.6% vs 72.6%, P<0.0001; 26.1% vs 72.2%, P=0.0013). For patients ith WT1≤5%, allo-SCT significantly lowered CIR rate at 3-year, improved 3-year DFS rate and tended to improve OS rate compared to chemotherapy/auto-SCT by landmark analysis, respectively (11.5% vs 88.2%, P<0.0001; 65.4% vs 11.8%, P=0.0001; 60.6% vs 32.6%, P=0.10. Figure 2). Multivariate analysis revealed that WT1 transcript levels (≤5% vs >5%) and treatment (chemotherapy/auto SCT vs allo-SCT) instead of other pretreatment parameters were independent prognostic factors for relapse (hazard ratio (HR) 0.20, 95% CI 0.093¨C0.44; 0.096, 95% CI 0.033¨C0.28. all P<0.0001) , DFS (HR 0.22, 95% CI 0.11¨C0.44; 0.23, 95% CI 0.11¨C0.49. all P<0.0001) and OS (HR 0.30, 95% CI 0.14¨C0.66, P=0.003; 0.37, 95% CI0.16¨C0.82, P=0.014).
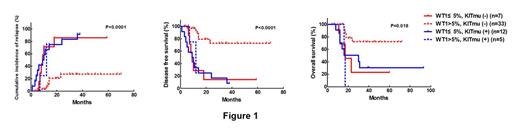
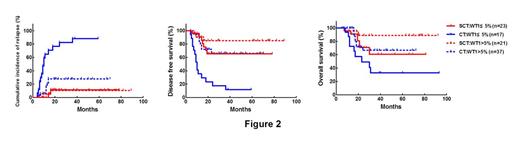
Less than 1-log increase of WT1 transcript levels at diagnosis is a strong predictor on poor outcome in patients with t (8;21) AML, and allo-SCT could significantly improve outcome of such patients.
Bejing Municipal Science & Technology Commission(Z111107067311070) and Nature Science Foundation of China (81170483).
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal