To the editor:
Approximately 3% to 5% of patients with acute promyelocytic leukemia (APL) will have an extramedullary relapse in their lifetimes, most commonly in the central nervous system (CNS).1 CNS relapse can be isolated or associated with a bone marrow (BM) relapse. The most appropriate clinical management remains controversial.2 The major obstacle to all treatments is the need for the therapeutic drugs to penetrate the blood-brain barrier (BBB). Although arsenic trioxide (ATO) and all-trans retinoic acid (ATRA) are among the frontline medications for APL intramarrow treatment, these water-soluble medicines have limited ability to cross the BBB and cannot reach therapeutically effective levels in the cerbrospinal fluid (CSF). With regular oral doses, the ATO level in CSF has been reported to reach only 17.7% of the corresponding levels in plasma.3 Previously, we investigated the potential efficacy of combination therapy with mannitol and ATO using a rabbit model, and we showed that the approach caused a transient increase in the BBB permeability for ATO, thereby increasing the ATO concentration in CSF.4 Our experiments using human cortical neurons revealed a differential tolerance of APL blasts to different concentrations of ATO.5,6 We also identified a safe range of ATO concentrations in the human CNS.7,8 The results from these collective studies allowed an ATO concentration in CSF that was both safe and therapeutic to be achieved.9
Here, we describe our efforts to extend the application of our method to additional patients. This study was registered at www.isrctn.org (#ISRCTN94954912). The study protocol was reviewed and approved by the Harbin Medical University Medical Ethics Committee, and signed informed consent was obtained from all patients or their legal guardians. Seventeen CNS APL patients diagnosed between 2000 and 2010 were included in this study (10 males and 7 females, between 6 and 50 years old). Expression of the PML/RARα gene [or t(15;17)(q22;q21) transcripts] was detected in all patient CSF. For all patients, the induction treatment was started immediately on diagnosis. The daily protocol was as follows: a bolus infusion of 125 mL of 20% mannitol mixed in 100 mL normal saline (NS) (for children ≤15 years old: 50 mL mannitol and 50 mL NS) was administered intravenously at a flow rate of 12 ∼20 mL/min (∼8-11 minutes total), followed by a slow intravenous infusion of 125 mL of 20% mannitol plus 7.0 mg/m2/day ATO (for children ≤15 years: 50 mL mannitol and 0.16 mg/kg/day ATO) in 500 mL NS at a flow rate of 1.0 mL/min (∼9-10.5 hours total). Patients were instructed to rest in bed during the entire procedure. Urine flow was measured to ensure maintenance of a rate of at least 30 to 50 mL/h. Daily infusions were continued until the patient’s CSF was found to be free of APL blasts/promyelocytes. The level of elemental arsenic metabolites was measured in each patient’s CSF and blood samples, which had been taken immediately after every other day’s infusion. Consolidation therapy started 2 weeks after the induction therapy and was repeated at 2-week intervals. A total of 3 cycles of consolidation therapy was given to all the patients who achieved normal CSF morphology with induction treatment. For each cycle of consolidation, daily ATO and mannitol infusion (administered as described for the induction treatment) continued for 14 consecutive days. Absence of PML/RARα transcripts in CSF and BM (defined as complete molecular remission [CMR]) was achieved prior to commencement of lifetime maintenance therapy. The first year after CMR, a 14-day cycle administration of ATO and mannitol was repeated at 1-month intervals. In the second year, a protocol identical to that of year 1 was instituted, but using a 3-month interval regimen. From the third year on, the protocol was administered at 6-month intervals and maintained for life. PML/RARα levels were monitored at 6-month intervals. If a relapse was identified, another cycle of remission induction was started. Clinical monitoring and supportive treatment followed our departmental guidelines.10
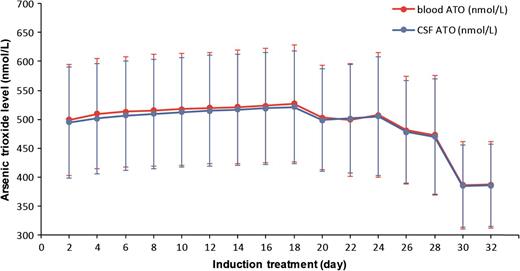
In 16 of the 17 patients examined, abnormal blasts/promyelocytes from CSF were eliminated in 18 to 32 days (median, 24 days) after the start of induction treatment. All the patients tolerated the induction well. There were no complaints of side effects associated with the use of mannitol. There were no documented cases of acute arsenic poisoning. No patients discontinued use of ATO during the induction. There were some cases of mild liver enzyme changes and mild nonhematologic toxicity associated with ATO, but in all instances, these abnormalities resolved with the use of hydroxyurea and dexamethasone for 3 to 5 days. There were no significant differences in the level of arsenic metabolites between children and adults. The correlation between parallel blood and CSF arsenic levels was significant (r = 0.998). Over the course of the entire induction treatment process, the concentrations of arsenic in the blood and CSF were fairly stable in each patient (Figure 1). For each individual, the arsenic level in CSF was ∼99.7% of those in the paired blood samples. However, arsenic levels in different individuals were highly variable in blood and CSF. Of the 16 patients who responded to induction therapy, all achieved CMR after the first consolidation cycle. Nine of these patients retained their CMR status during the follow-up period (median, 92 months; range, 70-125 months). Among the other 7 patients, there were 1 to 3 relapses in BM and/or CNS after their first CMR. All 7 of these patients responded well to ATO and mannitol during reinduction: 5 of them achieved CMR again and stayed free of relapse; 2 died after withdrawing treatment 3 to 5 months after their second or third remission. One 6-year-old female patient was nonresponsive to induction treatment after 49 days and later received ATRA and ATO alternative therapy. She died 15 months after the start of therapy. Eventually, the mean 5-year overall survival rate for all 17 patients was 82.4%. The mean 5-year event-free survival rate for all patients was 52.9%. At the time of the last follow-up appointment, all patients remained in generally good health without signs of chronic arsenic intoxication or second malignancy.
Elemental arsenic levels in patient-paired blood and CSF samples measured during induction treatment.
Elemental arsenic levels in patient-paired blood and CSF samples measured during induction treatment.
Authorship
Acknowledgments: The authors thank Dr Ellen Wertheimer for editing assistance. This project is supported by China National Natural Science Foundation grant 81070439 and China 863 Projects Foundation grant 2012AA020903.
Contribution: H.W. and J.Z. designed the study and wrote the manuscript; F.C., J.L., L.L., Y.L., C.S., W.L., D.L., Y.Z., Z.Z., X.L., R.M., and B.Y. performed the research; and H.Z. and B.Y. read and analyzed the data.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Jin Zhou, Department of Hematology, First Affiliated Hospital, Harbin Medical University, Harbin, Heilongjiang 150001, China; e-mail: zhoujin1111@126.com.