Abstract
Background: Previous studies using next-generation sequencing (NGS) have led to the identification of a number of genes mutated frequently in CLL. Recent publications focus on the most recurrently mutated genes (TP53, SF3B1 and NOTCH1) which tend to be mutually exclusive. Large series of untreated patients have shown that these mutations have a prognostic impact. Relapse may be associated with more frequent mutational events. Further investigation of relapsed CLL genomes within a clinical trial setting using a comprehensive NGS gene panel is required.
Methods: Using targeted NGS we determined the mutational spectrum of 118 refractory/relapsing CLL patients enrolled in one French and two UK prospective trials (ICLL01 from the French intergroup GCFLLC/MW-GOELAMS, NCRNCLL201, NCRNCLL202 respectively). Eighty percent of patients had an unmutated IGHV status and 21 (18%) patients carried a 17p deletion. Sequencing libraries were composed of a panel of nine recurrently mutated genes in CLL (i.e.TP53, SF3B1, ATM, NOTCH1, XPO1, SAMHD1, MED12, BIRC3 and MYD88) and run on the Illumina MiSeq instrument (Illumina Inc). On average 14.1 M reads were obtained per run of which 96.8% were identified reflecting an acceptable signal to noise ratio. Yield was 4.1 Gb and 95.9% of reads were above Q30 across 6 MiSeq runs. Data was analysed using our in-house bioinformatics pipeline consisting of a combination of two different aligners (Custom Amplicon Alignment, Illumina Inc and Stampy, Wellcome Trust centre for Human Genetics), two variant callers (GATK, Broad Institute and Platypus, Wellcome Trust centre for Human Genetics) and a stringent filtering process in order to detect SNVs and indels with a variant allele frequency down to 7%.
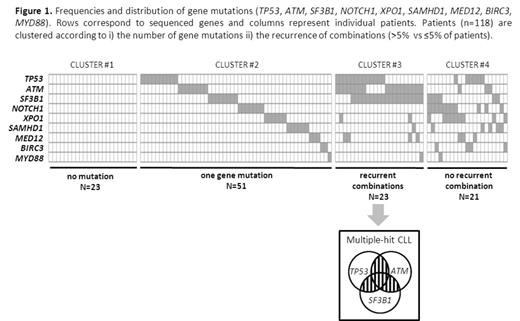
Results: We identified a total of 196 mutations (mean=1.7/sample) in 95 (80%) patients: 138 missense mutations, 41 substitutions/indels, 12 nonsense and 5 splicing mutations. TP53, SF3B1 and ATM mutations occurred frequently in 29 (24.6%), 33 (28%) and 29 (24.6%) patients, respectively. Eighteen (15.3%) patients harbored a NOTCH1 mutation matching the range of reported frequency. Mutations in the other genes sequenced were distributed as follows: XPO1 mutations in 17 (14.4%), SAMHD1 mutations in 12 (10.2%), MED12 mutations in 10 (8.5%), BIRC3 mutations in 6 (5.1%) and MYD88 mutations in 3 (2.5%) patients. Twenty-three (20%) patients did not have any mutations present (Figure 1, cluster #1). A total of 51 (43%) patients had one gene mutated (Figure 1, cluster #2) and the remaining 44 (37%) patients had two or more genes mutated (Figure 1, clusters #3 & #4). Recurrent combinations of mutations (affecting more than 5% of patients) were found in a group of 23 (20%) patients. These combinations of mutations comprised of at least two of the following genes: TP53, SF3B1 and ATM (Figure 1, cluster #3, so called multiple-hit (MH) profile). Remarkably, mutations in these 3 genes were found significantly more frequently associated than in isolation. We then investigated the potential clinical relevance of the MH profile. This profile was associated with poorer ORR than the remaining cohort (43% vs 80%, P<.0001). None of the patients with a MH profile achieved CR compared to 24% for the remaining patients (P=.006). MH patients have also shorter median PFS of 12 months compared to 19 months in cluster #1, 23 months in cluster #2 and 18 months in cluster #4 (P=.03). Multivariate analysis for PFS including relevant factors such as fludarabine-refractory' and TP53 disruption confirmed the adverse prognostic value related to the MH profile (HR=3.194 [95%CI=1.493-6.835], P=.003). Interestingly, among the TP53-disrupted patients, the MH profile retained its prognostic impact with a median PFS of 11 months for those with mut-SF3B1 and/or mut-ATM versus 22 months for those with wt-SF3B1 and wt-ATM (P=.022).
Conclusion: The mutational landscape of relapsing CLL is marked by a group of patients with combined mutations of the TP53, ATM and SF3B1 genes (multiple-hit profile) and is associated with an adverse prognostic impact. In addition to TP53 and SF3B1, ATM should be sequenced at relapse to predict outcome and guide subsequent therapeutic intervention. Further studies are required to confirm these findings and to understand the subclonal distribution of these mutations.
Hillmen:Pharmacyclics, Janssen, Gilead, Roche: Honoraria, Research Funding. Tournilhac:mundipharma: Honoraria, Other, Research Funding; GSK: Honoraria, Other, Research Funding; Roche: Honoraria, Other, Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal