Key Points
Local immunotherapy induced systemic responses in patients with disseminated FL.
Clinical responses correlated with systemic antitumor T-cell immunity.
Abstract
Advanced stage follicular lymphoma (FL) is incurable by conventional therapies. In the present pilot clinical trial, we explored the efficacy and immunogenicity of a novel in situ immunotherapeutic strategy. Fourteen patients with untreated or relapsed stage III/IV FL were included and received local radiotherapy to solitary lymphoma nodes and intranodal injections of low-dose rituximab (5 mg), immature autologous dendritic cells, and granulocyte-macrophage colony-stimulating factor at the same site. The treatment was repeated 3 times targeting different lymphoma nodes. Primary end points were clinical responses and induction of systemic immunity. Five out of 14 patients (36%) displayed objective clinical responses, including 1 patient with cutaneous FL who showed regression of skin lesions. Two of the patients had durable complete remissions. Notably, the magnitude of vaccination-induced systemic CD8 T-cell–mediated responses correlated closely with reduction in total tumor area (r = 0.71, P = .006), and immune responders showed prolonged time to next treatment. Clinical responders did not have a lower tumor burden than nonresponders pretreatment, suggesting that the T cells could eliminate large tumor masses once immune responses were induced. In conclusion, the combined use of 3 treatment modalities, and in situ administration in single lymphoma nodes, mediated systemic T-cell immunity accompanied by regression of disseminated FL. The trial was registered at www.clinicaltrials.gov as #NCT01926639.
Introduction
Follicular lymphoma (FL) is the most common subtype of indolent non-Hodgkin lymphoma and is often diagnosed in advanced stage.1 Although immunotherapy with anti-CD20 monoclonal antibodies has had a major impact on the prognosis in FL over the past 10 to 15 years,2-4 most patients eventually relapse and die of their disease. Therapeutic vaccines targeting the immunoglobulin idiotype in FL initially seemed promising,5-7 but subsequent phase 3 trials failed to document convincing clinical benefit.8-10 One of the studies, however, could report improvement in disease-free survival (P = .045),10 although some have argued that trial design issues may have influenced these results.11 It has been shown that patients can respond to idiotype vaccines and mount antitumor T-cell responses despite treatment with chemotherapy and rituximab prior to vaccination, whereas humoral antitumor responses are typically delayed until B-cell recovery.12 A recent study suggests, however, that idiotype vaccines may be more effective when administered to treatment-naive patients.13 There is at present no clear evidence that early aggressive therapies impact long-term prognosis in FL.14 Thus, a watch-and-wait strategy for nonsymptomatic disease is still the standard of care for many patients and creates a window of opportunity for testing of new immunostimulatory therapies.
FL is an inherently radiosensitive tumor. Early stage disease can be cured by radiotherapy alone, and low-dose radiotherapy of only 4 Gy has proved highly effective for eradication of lymphoma at local sites.15 Evidence has emerged that ionizing irradiation induces immunogenic cell death, setting the stage for systemic antitumoral immune responses.16 Furthermore, radiotherapy leads to cellular and molecular reprogramming of the tumor microenvironment, which promotes the maturation of dendritic cells (DCs) into effective antigen-presenting cells.17 Local radiotherapy followed by direct injections of DCs into tumors might therefore facilitate efficient processing and presentation of tumor antigens to T cells.18 Furthermore, there is increasing evidence that therapeutic antibodies, similar to radiotherapy, mediate indirect antitumor effects through the induction of adaptive immune responses,19,20 and a combination of these 3 therapeutic modalities might therefore prove beneficial.
In the present clinical trial, we hypothesized that 8-Gy irradiation to solitary lymphoma nodes combined with sequential intratumoral injections of low-dose rituximab and immature autologous DCs would induce systemic immune-mediated regression of lymphoma lesions at distant sites in patients with stage III/IV FL.
Patients and methods
Study population, diagnostic workup, and follow-up
Patients 18 years or older with untreated or relapsed biopsy-confirmed nonsymptomatic FL grades 1 to 3A and stage III/IV were enrolled. Patients had measurable disease present at other sites than those used for biopsy and radiation. Other inclusion criteria were as follows: World Health Organization performance status 0 to 1, neutrophils >1.0/µL, platelets >50/µL, life expectancy >6 months, and signed written informed consent. Patients were ineligible if they had progressive lymphoma in need of standard therapy, known central nervous system involvement of lymphoma, HIV infection or another chronic infection, or history of autoimmune disease or were pregnant. Patients underwent standard laboratory and clinical workup, including computed tomography (CT) of neck/thorax/abdomen, positron emission tomography (PET)/CT, and bone marrow biopsy and aspirates for flow cytometry. A surgical biopsy was performed for cryopreservation of single-cell suspensions of viable lymphoma cells for later immune monitoring. Mononuclear cells (MNCs) from peripheral blood were collected and cryopreserved prior to start of treatment and at posttreatment visits at 2, 4, and 8 months. Baseline imaging and laboratory studies were repeated during follow-up. For total tumor area calculations by CT, results were displayed as the fold-change of cross products of all lesions with largest diameter of 1.5 cm or more, excluding irradiated lesions. Each patient was scored at time of best response. Measurements were performed by 2 separate expert lymphoma radiologists who were blinded for the results of the immune response monitoring of study patients. One patient with primary cutaneous FL was evaluated by photographic documentation. The protocol was approved by Norwegian Medicines Agencies and Ethics Committee. Informed consent was obtained from all patients in accordance with the Declaration of Helsinki.
Production of immature DCs
Peripheral MNCs were collected by leukapheresis, and monocytes were separated and cultured for generation and cryopreservation of immature DCs in accordance with good manufacturing practice as previously described.21 The quality control of the immature DCs consisted of sterility tests, phenotyping, and viability testing.21
Antibodies and immune response measurements by flow cytometry
The antibodies were in-house conjugated to Pacific blue (PB) (anti-CD8 [Hit8a]) or to Alexa Fluor 647 (anti-CD107a [H4A3], anti-CD107b [H4B4], and anti-CD20), all from BD Biosciences. Anti-CD8 (SK1) PE or PB, anti-IFN-γ PE, anti-CD3 (SK7) PerCP-Cy5.5, and anti-CD127 (eBioRDR5) PE-Cy7 were from eBiosciences. Anti-FoxP3 (259D/C7) Alexa Fluor 647, anti-CD4 (L200) PerCP, and anti-CD45RA (HI100) APC-H7 were from BD Biosciences. Anti-CD25 (4E3) PE was from Miltenyi Biotec, Germany. Cells were analyzed on a FACS LSR II, and data analysis was performed with FACS DiVa software (BD Biosciences) or FlowJo (Tree Star).
Antitumor T-cell responses among MNCs harvested from peripheral blood samples were measured by flow cytometry following 5 days of coculture with autologous lymphoma cells (1 × 105 MNCs and FL cells at a ratio of 1:1) in serum-free X-vivo 20 medium (BioWhittaker, Lonza, Walkersville, MD). For measurements of proliferation, MNCs were labeled with carboxyfluorescein succinimidyl ester (CFSE; Invitrogen, Molecular Probes) at 1-μM concentration prior to coculture. At harvest, cells were stained with anti-CD20 to exclude the remaining FL cells, and with anti-CD3 and anti-CD8. T-cell proliferation in MNC samples drawn prior to start of therapy and at posttreatment visits was measured as frequencies of CFSElow events among CD20– and CD3+CD8+ or CD3+CD8– events (CD4+ T cells). For measurements of degranulation (CD107a/b) and production of interferon (IFN) γ, cocultures were restimulated on day 5 with 105 anti-CD20-labeled tumor cells for 5 hours at 37°C in the presence of monensin (final concentration 10 μM) and brefeldin A (final concentration 10 μg/mL). Cells were subsequently stained with anti-CD107a/b, anti-CD3, and anti-CD8, and in some samples also with anti-IFN-γ following fixation and permeabilization, as previously described.22,23 As positive controls for T-cell responses, MNCs were stimulated with either phythohemagglutinin or MNCs from a third-party donor, which resulted in similarly strong responses in MNCs sampled before and after vaccination. Staining of regulatory T cells (Treg’s) was performed according to the manufacturer’s protocol, using Human FoxP3 Buffer Stain from BD Biosciences.
Treatment schema
Low-dose rituximab (5 mg in 1 mL saline) was injected in a solitary palpable lymphoma node or lesion on days 1 and 3. A single dose of 8-Gy radiotherapy targeting the same site was administered on day 2. In the majority of cases, an electron beam field was applied with 1-cm margin. Immature DCs (5-10 × 107 in 1 mL of saline) were injected in the irradiated lesion on days 4 and 5. Additionally, granulocyte-monocyte colony-stimulating factor (50 µg in 1 mL of saline) was administered subcutaneously close to the lesion on days 4 and 5, to potentially facilitate retention of the cells and promote efficient uptake of tumor material. The intranodal injections were guided by ultrasound and performed by a radiologist to ensure correct administration. This schedule was performed 3 times: weeks 1, 3, and 5, targeting a different lymphoma lesion each week. Restaging was done 2, 4, 8, and 12 months after the start of therapy; then every sixth month the second year; and then annually until 5 years or until need of systemic treatment.
Response criteria and statistics
Evaluation of response was performed according to the International Working Group (IWG) criteria of 199924 and 2007,25 excluding irradiated lesions. In this pilot clinical trial, the aim was to detect immune responses and/or clinical responses in a clinically relevant proportion of the patients. Assuming that 50% in the relevant patient group responded, we would with 14 patients detect a response in at least 4 patients (29%) with a probability >90%. Progression-free survival was calculated for all patients from date of inclusion to progression or relapse or death of any cause. Time to next treatment was calculated from date of inclusion until start of new systemic conventional therapy. Survival analyses were performed according to the Kaplan-Meier method,26 and differences between subgroups were analyzed by the log-rank test. Pearson’s statistics was used to analyze correlations between clinical and immunologic end points.
Results
Patient characteristics
A total of 14 patients with stage III/IV FL grades 1 to 3A were enrolled and treated according to the protocol from 2009 to 2012 (Table 1). The majority had not received any previous therapies (11/14), and all had SD not requiring standard therapy at inclusion. Median age of the study population was 59 years (range 33-81). Patients had disseminated disease and enlarged lymph nodes or lesions >1.5 cm at multiple sites available for radiotherapy and local injections. All patients received radiation and intranodal immunotherapy targeting single lymph nodes at 3 different sites. A median of 6 untreated nodal areas with lesions of >1.5 cm in largest diameter was used for monitoring of clinical responses (range 3-8).
Patients’ characteristics and systemic response to local immunotherapy
| Patient . | Age . | Disease grade . | Stage . | FLIPI . | Previous treatment . | Months since diagnosis or treatment . | Response IWG 199924 . | Response IWG 200725 . |
|---|---|---|---|---|---|---|---|---|
| 1 | 59 | FL 2 | IVA | 2 | None | 13 | PD | PD |
| 2 | 66 | FL 2 | IVA | 4 | None | 5 | CR | CR |
| 3 | 43 | FL 1 | IVA | 2 | None | 3 | SD | SD |
| 4 | 55 | FL 2 | IVA | 2 | None | 6 | SD | SD |
| 5 | 72 | FL 1 | IVA | 2 | None | 6 | CR | PR |
| 6 | 54 | FL 2 | IIIA | 2 | None | 6 | SD | SD |
| 7 | 62 | FL 2 | IVA | 4 | Zevalin | 35 | SD | SD |
| 8 | 62 | FL 2 | IVA | 3 | Radiotherapy | 37 | PR | PR |
| 9 | 40 | FL 3A | IVA | 1 | Rituximab | 33 | PR* | PR* |
| 10 | 81 | FL 1 | IVA | 3 | None | 3 | SD | SD |
| 11 | 53 | FL 2 | IVA | 2 | None | 9 | PD | PD |
| 12 | 58 | FL 2 | IVA | 2 | None | 2 | SD | SD |
| 13 | 66 | FL 1 | IVA | 3 | None | 12 | SD | SD |
| 14 | 33 | FL 1 | IVA | 2 | None | 13 | PR | PR |
| Patient . | Age . | Disease grade . | Stage . | FLIPI . | Previous treatment . | Months since diagnosis or treatment . | Response IWG 199924 . | Response IWG 200725 . |
|---|---|---|---|---|---|---|---|---|
| 1 | 59 | FL 2 | IVA | 2 | None | 13 | PD | PD |
| 2 | 66 | FL 2 | IVA | 4 | None | 5 | CR | CR |
| 3 | 43 | FL 1 | IVA | 2 | None | 3 | SD | SD |
| 4 | 55 | FL 2 | IVA | 2 | None | 6 | SD | SD |
| 5 | 72 | FL 1 | IVA | 2 | None | 6 | CR | PR |
| 6 | 54 | FL 2 | IIIA | 2 | None | 6 | SD | SD |
| 7 | 62 | FL 2 | IVA | 4 | Zevalin | 35 | SD | SD |
| 8 | 62 | FL 2 | IVA | 3 | Radiotherapy | 37 | PR | PR |
| 9 | 40 | FL 3A | IVA | 1 | Rituximab | 33 | PR* | PR* |
| 10 | 81 | FL 1 | IVA | 3 | None | 3 | SD | SD |
| 11 | 53 | FL 2 | IVA | 2 | None | 9 | PD | PD |
| 12 | 58 | FL 2 | IVA | 2 | None | 2 | SD | SD |
| 13 | 66 | FL 1 | IVA | 3 | None | 12 | SD | SD |
| 14 | 33 | FL 1 | IVA | 2 | None | 13 | PR | PR |
CR, complete response; FLIPI, FL prognostic index; PD, progressed disease; PR, partial response; SD, stable disease.
Cutaneous lymphoma not evaluable by IWG 1999 or 2007.
Adverse events
Adverse events were monitored and scored in agreement with the National Cancer Institute Common Terminology Criteria for Adverse Events for toxicity grading. The treatment was well tolerated, and side effects were limited to mild fever and flulike symptoms grade 1 in some patients in conjunction with the rituximab injection. Autoimmune toxicities were not observed. All patients showed a reduction of peripheral blood B cells posttreatment, presumably caused by a systemic effect of rituximab.
Clinical responses and correlated systemic antitumor T-cell responses
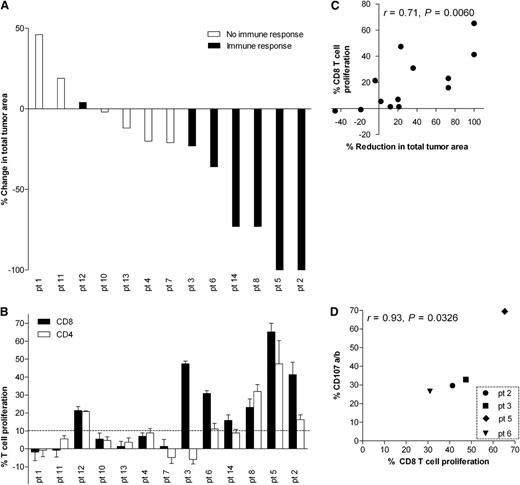
In Figure 1A, a watershed diagram shows change in total tumor areas at time of best clinical response in 13 of 14 patients, excluding the patient with primary cutaneous FL. When excluding irradiated lesions, the overall objective response rate was 31%, with 2 CRs and 2 PRs, according to the IWG response criteria of 199924 (Table 1). When including the patient with primary cutaneous FL, who had regression of disseminated skin lesions, the overall response rate was 36%. According to the revised response criteria of 2007,25 including PET/CT, 1 of the 2 patients in CR who had a minor metabolic uptake by PET/CT was scored as PR. Seven patients had SD, and 2 PD (Table 1). In all 6 patients with the largest reduction in total tumor area, robust immune responses were induced postvaccination (Figure 1A-B). Moreover, we observed a strong correlation between the percentage reduction in total tumor area and the magnitude of CD8 T-cell antitumor responses (Figure 1C, r = 0.71, P = .006).
Intranodal vaccination induces clinical responses and correlated T-cell responses. (A) Watershed diagram of change in total tumor area at time of best clinical response compared with baseline and associated positive (black bars) or negative (white bars) CD8 T-cell immune responses, as defined in panel B. (B) Percent CD8 (black bars) or CD4 (white bars) T cells proliferating in response to autologous tumor. Time point for best response after treatment (2, 4, or 8 months) following subtraction of baseline values is shown, with positive immune responses defined as 10% or higher (dotted line). CFSE-labeled MNCs were cocultured with autologous tumor cells (1:1 ratio) and proliferation measured on day 5 as CSFElow events among gated CD3+CD20– events that were either CD8+ or CD8–. Error bars indicate standard deviation of triplicates. (C) Correlation between percent reduction in tumor area and percent CD8 proliferation at time point for best response. (D) Correlation between CD8 T-cell proliferation (%CSFElow) and degranulation (% expressing CD107a/b) at time point for best response following restimulation with autologous tumor cells for the 4 patients for which both assays were performed (baseline values subtracted).
Intranodal vaccination induces clinical responses and correlated T-cell responses. (A) Watershed diagram of change in total tumor area at time of best clinical response compared with baseline and associated positive (black bars) or negative (white bars) CD8 T-cell immune responses, as defined in panel B. (B) Percent CD8 (black bars) or CD4 (white bars) T cells proliferating in response to autologous tumor. Time point for best response after treatment (2, 4, or 8 months) following subtraction of baseline values is shown, with positive immune responses defined as 10% or higher (dotted line). CFSE-labeled MNCs were cocultured with autologous tumor cells (1:1 ratio) and proliferation measured on day 5 as CSFElow events among gated CD3+CD20– events that were either CD8+ or CD8–. Error bars indicate standard deviation of triplicates. (C) Correlation between percent reduction in tumor area and percent CD8 proliferation at time point for best response. (D) Correlation between CD8 T-cell proliferation (%CSFElow) and degranulation (% expressing CD107a/b) at time point for best response following restimulation with autologous tumor cells for the 4 patients for which both assays were performed (baseline values subtracted).
Of the 13 patients evaluable for immunomonitoring, 7 (54%) showed strong antitumor T-cell responses in posttreatment peripheral blood samples. In the majority of cases, there was an increase in the proliferation of CD8+ as well as CD4+ T cells. However, CD8 responses were generally stronger (Figure 1B). In all cases, T-cell proliferation against tumor seen in peripheral blood MNCs sampled before the start of treatment was low, and all values for antitumor responses after the start of therapy were reported following subtraction of antitumor responses before therapy (Figure 1B [proliferation] and Figure 1D [degranulation]). Notably, tumor cells sampled before therapy were used for stimulation of all MNCs, and no serum or cytokines were added to minimize background proliferation. MNCs cultured in medium only showed negligible proliferation (supplemental Figure 1A available on the Blood Web site). The majority of tumor-reactive CD8 cells underwent 6 or more divisions during the 5-day culture period, as shown for T cells sampled after treatment from 1 of the complete responders (supplemental Figure 1B). Figure 1D shows that there was a strong correlation between CD8 proliferation and degranulation in the 4 patients in which both assays were applied (r = 0.93, P = .0326). We furthermore assessed the frequencies of FoxP3-positive Treg’s among peripheral blood CD4 T cells from 1 of the complete responders (patient 2) and 2 nonresponders (patients 10 and 11) sampled before treatment and at 2 months after start of therapy (supplemental Figure 2). Although the data represent very few patients, it is worth noting that the complete responder had lower levels of Treg’s than the nonresponders at both time points (FoxP3+CD127– cells in CD4 T cells) and also the largest reduction in activated Treg’s (FoxP3hiCD45RA–) after treatment relative to before (supplemental Figure 2B-C).
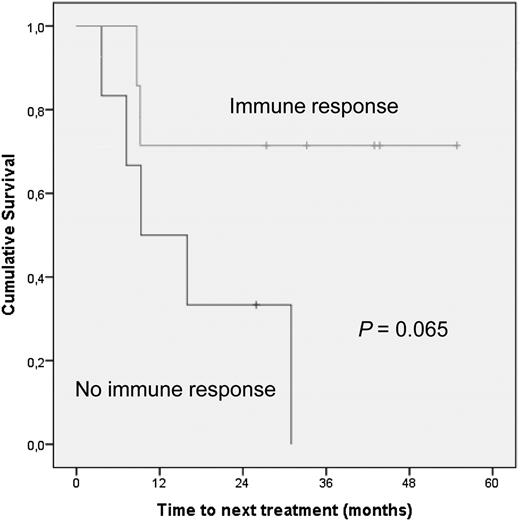
The median time to progression among the patients who mounted antitumor immune responses was not significantly longer than for the nonresponders. However, only 2 out of 7 patients (29%) who developed T-cell responses have received subsequent conventional systemic treatments, compared with 6 of 7 nonresponders (86%). Hence, despite the limited size of our study, there was a trend toward prolonged time to next treatment of the group that had detectable T-cell responses, relative to those without a T-cell response (Figure 2, P = .065). Prognostic factors, such as age, stage, and FLIPI score were not predictive of clinical response or immune response (not shown). Total tumor area at baseline did not differ in immune responders as compared with nonresponders (2-sided Student t test P = .859). Among patients who developed a T-cell response, there was no difference in total tumor area at inclusion between those achieving an objective clinical response and those who did not (2-sided Student t test P = .667).
Patients with detectable immune responses show prolonged time to next treatment compared with patients without immune responses.
Patients with detectable immune responses show prolonged time to next treatment compared with patients without immune responses.
Characteristics of the clinical responders
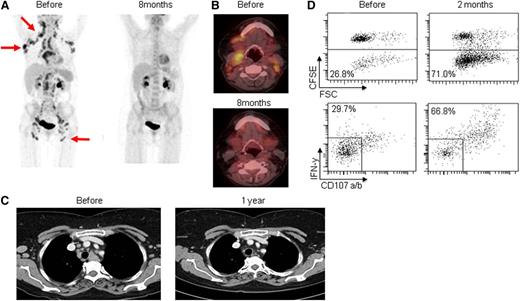
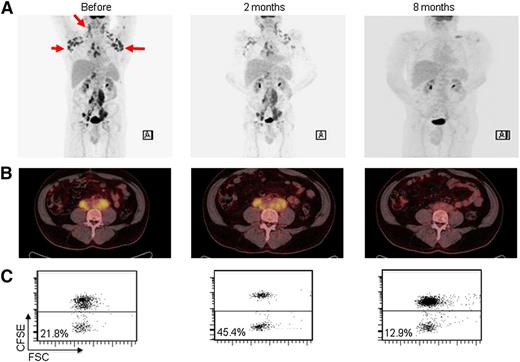
Patient 2 had stage IVA FL and obtained a clinical response that developed gradually, with a decreased metabolic uptake visible at 4 months and complete PET/CT negativity at 8 months after start of therapy (Figure 3A-C). Before treatment was initiated, there was a 40% infiltration of FL in the bone marrow, assessed by immunohistologic examination. Bone marrow involvement persisted at 8 months but had decreased to 10%. At 1 year, there was no sign of FL by histologic examination or minimal residual disease assessment by diagnostic flow cytometry. The clinical response was associated with antitumor T-cell responses found at 2 and 4 months manifested by vaccine-induced increase in CD4 and CD8 T-cell proliferation as well as degranulation (CD107a/b) and IFN-γ production by CD8 T cells (Figure 3D). The patient is still in complete remission with a follow-up of 54 months. Patient 5 had stage IIIA FL and achieved CR by CT at 8 months. The PET/CT confirmed a substantial response with only minor metabolic uptake (Figure 4A). The patient developed sustained antitumor CD8 and CD4 T-cell responses (Figure 4B-C) present at 2, 4, and 8 months. He remained in CR for 2 years, at which time he relapsed with morphologically verified FL at 2 sites. Both nodes were re-treated with the same approach, and the patient achieved a second CR that lasted for 14 months until he relapsed at 1 site. At 42 months follow-up, he is still not in need of additional treatment. Patient 8 had stage IV FL and achieved best response (PR) at 8 months. As for previous responders, PET/CT improved from 4 to 8 months (Figure 5A-B). Lymphoma infiltration in the bone marrow was not detectable at 8 months. A CD8 T-cell response was observed at 2 months only (Figure 5C), consistent with a limited immunologic control and potentially explaining the relapse occurring at 12 months. The patient was re-treated, but a second response was not observed, and the disease slowly progressed, albeit with no need for further therapy at 19 months. Patient 14 had stage IV FL and a more rapid response with PR as assessed by CT at 4 months and also marked improvement by PET/CT. A T-cell response was detected at 2 months. The patient relapsed at 8 months. He was treated with single-agent rituximab and achieved CR. Patient 9 had cutaneous FL and displayed regression of the majority of skin lesions after vaccination, as evaluated by photographic documentation. He stayed in partial remission for 2 years before relapse. This patient is still not in need of further therapy at 29 months.
Complete clinical response and T-cell responses in patient number 2. (A-B) PET/CT scans taken before start of treatment and at 8 months. (C) CT from before treatment and at 1 year. (D) Flow cytometric measurements of antitumor CD8+ T-cell responses in peripheral blood drawn before treatment and at 2 months postvaccination, following 5 days of coculture with autologous T cells. Upper dot plots: Percent proliferating cells, measured as CD3+CD8+CD20– events showing low CFSE fluorescence. Lower dot plots: On day 5, cocultures of MNCs and tumor cells were restimulated with autologous tumor cells for 5 hours before measuring degranulation (CD107 a/b expression) and secretion of IFN-γ. Numbers represent percent responding cells.
Complete clinical response and T-cell responses in patient number 2. (A-B) PET/CT scans taken before start of treatment and at 8 months. (C) CT from before treatment and at 1 year. (D) Flow cytometric measurements of antitumor CD8+ T-cell responses in peripheral blood drawn before treatment and at 2 months postvaccination, following 5 days of coculture with autologous T cells. Upper dot plots: Percent proliferating cells, measured as CD3+CD8+CD20– events showing low CFSE fluorescence. Lower dot plots: On day 5, cocultures of MNCs and tumor cells were restimulated with autologous tumor cells for 5 hours before measuring degranulation (CD107 a/b expression) and secretion of IFN-γ. Numbers represent percent responding cells.
Complete clinical response and vigorous T-cell responses in patient number 5. (A) PET/CT scans before treatment and at 4 and 8 months. Proliferation (B) and degranulation (C) of CD8+ T cells is shown in samples taken before treatment and at 4 and 8 months, analyzed as described in the legend of Figure 1.
Complete clinical response and vigorous T-cell responses in patient number 5. (A) PET/CT scans before treatment and at 4 and 8 months. Proliferation (B) and degranulation (C) of CD8+ T cells is shown in samples taken before treatment and at 4 and 8 months, analyzed as described in the legend of Figure 1.
Partial clinical response and T-cell response in patient number 8. (A-B) PET/CT scans before treatment and at 4 and 8 months. (C) CD8 T-cell proliferation monitored before treatment and at 2 and 8 months, as described in the legend of Figure 1.
Partial clinical response and T-cell response in patient number 8. (A-B) PET/CT scans before treatment and at 4 and 8 months. (C) CD8 T-cell proliferation monitored before treatment and at 2 and 8 months, as described in the legend of Figure 1.
Discussion
Here, we demonstrate that an in situ strategy combining sequential treatment with local radiotherapy and intranodal injections of low-dose rituximab and autologous DCs was safe and effectively induced clinical responses in patients with considerable tumor burden and disseminated FL. Notably, clinical responses were invariably accompanied by systemic antitumor T-cell responses. Thus, 2 patients with continuous CR had strong T-cell responses detected in continuous peripheral blood samples drawn posttreatment, whereas the patients who achieved PR had transient T-cell responses preceding the PR. The reduction in tumor area correlated strongly with the magnitude of antitumor CD8 T-cell reactivity, indicating that the locally administrated therapy evoked systemic antitumor immunity acting at distant sites.
Vaccine formulations based on systemic injection of DCs loaded with lymphoma cell preparations ex vivo27-29 or tumor-derived proteoliposomes30 have shown some promise, including objective clinical responses linked to increased antitumor reactivity of tumor-infiltrating T cells.29 However, no systemic antitumor T-cell responses were detected. Furthermore, clinical and immune responses were restricted to patients without high tumor burden or bulky disease. In contrast, we were able to detect systemic antitumor T-cell immunity in all responding patients, regardless of total tumor area at baseline, and the magnitude of CD8 T-cell responses was highly correlated to reduction in total tumor area.
A potentially important aspect of the current protocol is the route of vaccination. In murine models, intratumoral injections of DCs alone or in combination with either chemotherapy, local radiation, or radionuclides have been shown to induce tumor regression and antitumor immunity.18,31,32 Intratumoral administration of autologous DCs was tested previously in human cancer patients and was shown to be safe.33,34 In a study with a similar strategy to ours, patients with relapsed indolent B-cell lymphoma received repeated intranodal injections with the toll-like receptor agonist cytosine guanine dinucleotide following 4-Gy local irradiation.35 Clinical responses were inversely correlated to the ability of tumor cells to induce Treg’s among autologous CD4 T cells ex vivo. Tumor-reactive CD8 T cells were demonstrated in some patients but not significantly correlated to clinical response. This group also explored the cytosine guanine dinucleotide/radiation strategy in patients with mucosis fungoides, and some patients had clinical responses at untreated sites.36 The immunized sites showed a reduction in CD25+FoxP3+ Treg’s with a trend toward greater reduction in responders. Of note, we observed depletion of FoxP3hiCD45RAlow-positive activated Treg’s37 in peripheral blood posttreatment in 1 of the complete responders, unlike in 2 nonresponders.
We chose immature DCs because of their superior ability to acquire antigens compared with mature DCs.38 Local radiation induces inflammatory signals and immunogenic cell death that has been shown to facilitate maturation of the DCs.17 Upon maturation, the DCs upregulate HLA and costimulatory molecules and secrete cytokines that promote the presentation of antigens to T cells.38,39 Furthermore, it has been shown that DCs administered directly into tumors migrate to draining lymph nodes more effectively than when injected in the skin,40,41 thereby paving the way for systemic antitumor immunity.
There is evidence that intranodal injections of rituximab can be effective at local sites,42 and that binding to lymphoma cells will enhance Fc-receptor-mediated fagocytosis by DCs.43,44 Furthermore, targeting of antibody-coated tumor cells to DCs may improve the cross presentation of tumor antigens to CD8 T cells by an Fc-dependent mechanism that occurs after uptake of tumor.20 We thus hypothesized that rituximab would promote the uptake of irradiated tumor cells and presentation of tumor-associated antigens by the injected DCs. Furthermore, rituximab has been shown to increase the sensitivity of lymphoma cells for external beam radiation.45 The question can, however, be raised whether regression of lymphoma at distant sites in our study could be because of a direct systemic effect of rituximab. We believe that this is unlikely because the total dose of 30 mg of rituximab administered in lymph nodes is ∼100-fold less than normally used for single-agent rituximab therapy. Few studies have been conducted that might shed light on possible systemic effects after local treatment with rituximab. In a case report with intranodal injections of rituximab in a patient with cutaneous B-cell lymphoma, efficacy was noted even for untreated cutaneous lesions.46 However, the cumulative dose of rituximab administered was as high as 300 mg/m2. In another small study on indolent cutaneous B-cell lymphoma, 4 out of 6 patients who were treated with intratumoral injections of rituximab and responded locally had recurrence of lymphoma at distant sites after a median of 6 months.47 Hence, even though the total dose of rituximab used for local treatment was 5- to 10-fold higher than in our study, it did not protect against short-term systemic relapse. Finally, the slow kinetics of the clinical responses in our study, which typically peaked at 8 to 12 months after start of treatment, was unlike the response patterns normally observed after rituximab therapy.48 Even though we cannot completely exclude some systemic effect of locally administered rituximab, the strong correlation between clinical responses and antitumor T-cell reactivity observed suggests a predominantly immune-mediated mechanism.
In conclusion, our sequential in situ vaccine strategy showed clinical efficacy in patients with disseminated FL. Importantly, we were able to show a strong correlation between the magnitude of systemic antitumor T-cell responses and clinical responses, suggesting that T-cell–mediated attack may be responsible for the tumor reduction. The targets of the T-cell responses remain to be identified, but lack of autoimmune side effects suggests specificity for B cells. The presented strategy represents a promising platform for further development of even more effective immunotherapeutic approaches in FL.
The online version of this article contains a data supplement.
There is an Inside Blood Commentary on this article in this issue.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Acknowledgments
The authors thank Ron Levy (Stanford University Medical Center) for valuable advice and interest in the project, Karl-Johan Malmberg (Oslo University Hospital) for critically reviewing the manuscript, and Johannes Landskron (The Biotechnology Centre of Oslo, University of Oslo) for expert advice with regard to Treg analysis.
This work was supported by grants from the Norwegian Cancer Society, the Regional Health Authorities South-Eastern Norway, K. G. Jebsen Foundation, the Research Council of Norway, and Oslo University Hospital Radiumhospitalet.
Authorship
Contribution: A.K., H.H., and J.O. were responsible for conception and design of the study; all authors performed collection and assembly of data; A.K., S.K., M.W., and J.O. performed data analysis and interpretation; A.K. and J.O. wrote the manuscript; and all authors participated in final approval of the manuscript.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Arne Kolstad, Department of Oncology, Oslo University Hospital, Radiumhospitalet, Ullernchausséen 70, N-0310 Oslo, Norway; e-mail: arnek@ous-hf.no.


This feature is available to Subscribers Only
Sign In or Create an Account Close Modal