In this issue of Blood, Camus et al describe how microparticles derived from sickle erythrocytes can deliver heme to vascular endothelial cells, leading to their activation and injury, and promote vasoocclusion in sickle cell disease (SCD).1
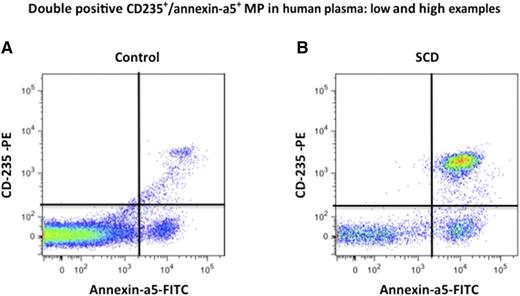
Erythrocyte MP quantification in human plasma by fluorescence-activated cell sorter (FACS). Erythrocyte MPs were quantified as annexin-a5+/CD235+ double-positive events by FACS in control and sickle cell disease plasma vs calibrated fluorescent microbeads body. (A-B) Examples of stained annexin-a5+ and CD235a+ events in plasma: (A) control plasma and (B) SCD plasma. Modified from supplemental Figure 1 in the article by Camus et al that begins on page 3805.
Erythrocyte MP quantification in human plasma by fluorescence-activated cell sorter (FACS). Erythrocyte MPs were quantified as annexin-a5+/CD235+ double-positive events by FACS in control and sickle cell disease plasma vs calibrated fluorescent microbeads body. (A-B) Examples of stained annexin-a5+ and CD235a+ events in plasma: (A) control plasma and (B) SCD plasma. Modified from supplemental Figure 1 in the article by Camus et al that begins on page 3805.
From the time of Herrick’s description of the oddly shaped red blood cells (RBCs) of Walter Clement Noel in Chicago, investigators have puzzled over how the sickle RBC leads to hemolysis, organ dysfunction, and pain. Ham and Castle,2 in 1940, postulated that it was the consequence of a vicious cycle of deoxygenation and sickling initiated by increased viscosity, the end point being vascular occlusion by erythrostasis. After defining the molecular basis of the disease and the modulating effects of hemoglobin F on polymerization, the field turned to the role of the vasculature based on the pioneering work of Hebbel on sickle RBC adhesion to endothelial cells.3 Subsequently, the roles of inflammation, oxidative stress, nitric oxide biology, red cell hydration, blood cell adhesion, endothelial injury, etc have been examined to understand the pathophysiology.
Hemolysis with the daily release of free hemoglobin (Hb) bathing the vasculature associated with concomitant anemia, decreased levels of the hemoglobin/heme binding proteins, haptoglobin, and hemopexin is critical in understanding the vasculopathy of SCD.4 The mechanism of Hb toxicity is the pro-oxidative reactivity of Hb in plasma and within tissues after extravasation. Hb reactions with nitric oxide and with physiologic oxidants (eg, hydrogen peroxide and lipid peroxides) are the most extensively studied. Another mechanism of Hb toxicity is through release of hemin from methemoglobin (Hb[Fe3+]). Hemin release allows transfer of the reactive hydrophobic porphyrin to cell membranes or plasma proteins. The high affinity (Kd < 10−13) plasma heme binding protein hemopexin can prevent heme-mediated toxicity, but in SCD, hemopexin levels are depleted.5 Because of heme’s hydrophobicity, lower-affinity binding proteins (eg, albumin), lipoproteins, and cell membranes bind heme. Ultimately, heme is delivered to cells where it can selectively bind to receptors, transcription factors, and enzymes; be redox active; and alter cell activation state, gene transcription, and metabolism.6 In SCD mouse models, hemoglobin and heme induce vascular stasis and injury through interactions with Toll-like receptor 4 (TLR-4), inducing cell signaling, oxidant production, nuclear factor-κB activation, and adhesion molecule and tissue factor expression. TLR-4 inhibitors, adhesion molecule blockade, hemopexin, and antioxidants can diminish these effects.7
However, with all the heme scavengers in plasma, is there really ever “free heme” available to interact with the endothelium to promote vascular stasis? Camus et al show that microparticles (MPs) derived from sickle RBCs laden with heme are an important delivery vehicle.1 MPs, which express phosphatidylserine (PS), are 3- to 10-fold higher in SCD patients during steady state than in healthy control subjects (see figure) and further increased up to 3-fold during vasoocclusive crisis. SCD MPs are mostly of RBC origin and may be produced through accelerated aging of sickle cells during oxygenation/deoxygenation cycles, under the influence of stress factors and matrix proteins, or during hemolysis in capillary beds. MPs from human SCD plasma or generated in vitro from sickle and control RBCs contain heme. In SCD, MPs might carry up to 10 μM hemoglobin, and 20% to 35% consisted of methemoglobin. In vitro, SCD RBCs induced to vesiculate contained twice as much heme as controls. Synthetic phospholipid vesicles containing PS and calcium, which mimic RBCs, avidly bind heme, but this could be reduced by the addition of hemopexin. The heme-laden RBC MPs transferred heme to endothelial cells (human umbilical vein endothelial cells) and triggered endothelial reactive oxygen species and apoptosis blocked by apocynin, hemopexin, TLR-4 blockade, or an antibody to annexin-5. In mouse mesenteric vessels, the MPs from a sickle mouse reduced acetylcholine-induced vasodilation, and hemopexin coincubation reversed this effect. Finally, heme-laden mouse SCD MPs infused into a sickle mouse altered kidney blood flow, and the histology showed congestion consistent with vasoocclusion.
This report provides an elegant and thorough review of the role of RBC MPs in the pathophysiology of SCD. Specifically, it adds to the evidence that hemolysis is pivotal to vasoocclusion and vasculopathy in SCD. In thinking about approaches to SCD, ideally gene therapy or hematopoietic stem cell transplantation would eliminate the hemolysis and vasoocclusion. Preventing polymerization, for example, by elevating hemoglobin F or minimizing hemolysis, has been lifesaving. However, if these measures are not complete, dealing with the proinflammatory, proadhesive, procoagulant, pro-occlusive consequences of hemolysis requires detoxification of heme through heme oxygenase, antioxidants, etc, or clearing the hemoglobin/heme with haptoglobin/hemopexin. To break the vicious cycle of erythrostasis and sickling proposed by Ham and Castle, these and other therapies should be considered based on the studies presented by Camus et al.
Conflict-of-interest disclosure: G.M.V. receives research funding from Seattle Genetics and Biogen Idec.