Abstract
Background: Cytogenetic abnormalities are identified at the time of diagnosis in approximately 80% of patients with acute lymphoblastic leukemia (ALL). In acute myelogenous leukemia (AML), the detection of persistent abnormal cytogenetics at complete remission (ACCR) is associated with shorter relapse-free survival (RFS) and overall survival (OS) compared to patients with normal cytogenetics at CR (NCCR). However, the incidence and prognostic significance of ACCR in adult patients with ALL is unknown.
Methods: We evaluated 324 adult patients with ALL and abnormal cytogenetics at baseline who were treated on frontline induction chemotherapy protocols between 6/2001 and 3/2015 and achieved complete remission (CR) or CR without platelet recovery (CRp). Bone marrow specimens for cytogenetic and minimal residual disease (MRD) assessment were obtained at the time of CR. Cytogenetic abnormalities were classified according to standard conventions (Moorman et al., Blood 2007;109:3189-97). MRD by multi-parameter flow cytometry (MFC) was assessed with a sensitivity of 0.01% using a 15-marker, 4-color panel in the first half of the study period; subsequently a 6-color panel was used.
Results: 272 patients (84%) had cytogenetic analysis performed at the time of remission and were evaluable for this analysis. Median age was 48 years (range, 16-84) and median WBC at presentation was 11.6 x109/L (range, 0.4-629.4 x109/L). A hyperCVAD backbone was used for induction in 237 patients (87%) and augmented BFM was used in 35 (13%). All patients with Philadelphia positive (Ph+) ALL were treated with a tyrosine kinase inhibitor added to the chemotherapy regimen. 245 patients (90%) had pre-B ALL, 14 (5%) had pre-T ALL and 13 (5%) had Burkitt or Burkitt-like leukemia. Cytogenetics at baseline were Ph+ in 119 (44%), hyperdiploid in 31 (11%), complex in 18 (7%), MLL rearranged in 16 (6%), hypodiploid in 9 (3%) and miscellaneous in 79 (29%). Median time to CR was 23 days (range 14 to 84 days).
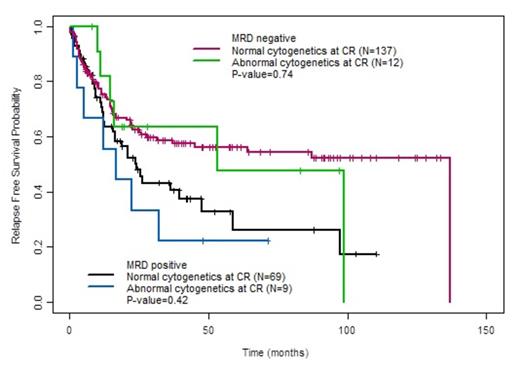
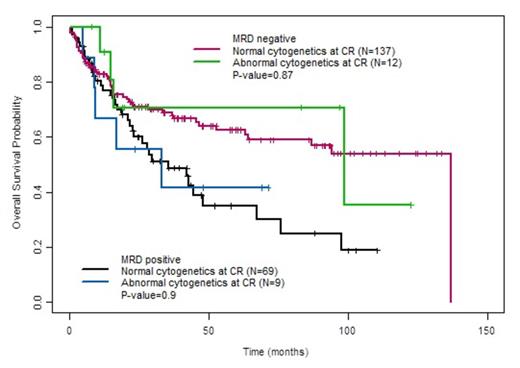
Among the 272 patients, ACCR was observed in 26 (9.6%). Baseline characteristics associated with ACCR were Ph+ ALL (62% of ACCR group vs. 42% of NCCR group, P=0.055) and longer mean time to CR (29.8 ± 15.6 days for ACCR group vs. 26.0 ± 9.7 days for NCCR group, P=0.07). Median RFS was 22.4 months (range, 12.3 months to not reached) for patients with ACCR vs. 47.7 months (range, 29.5 to 125 months) in those patients with NCCR (P=0.31). Median OS did not differ between the ACCR (98.7 months [range, 17.1 months to not reached]) and NCCR groups (67.3 months [range, 46.6 months to not reached], P=0.86). There was also not a significant difference in RFS or OS between the ACCR and NCCR groups when only Ph+ patients were evaluated. Among the 227 patients evaluable for MRD by MFC, MRD positivity at CR was observed in 78 patients (34%) and was highly associated with shorter RFS and OS (P<0.01 for both). The specificity of ACCR for detecting MRD (as assessed by MFC) was 43%, and there was overall poor correlation between these two methods for the detection of residual disease (P=0.47). When patients were stratified by MRD status as assessed by MFC, the presence of absence of persistent cytogenetic abnormalities did not add additional prognostic information (Fig. 1 and 2).
Conclusions: There is poor association between MRD assessment by MFC and the presence or absence of cytogenetic abnormalities at CR in adult patients with ALL. Although ACCR has prognostic significance in AML, ACCR is not associated with adverse outcomes in ALL and therefore should not be used to guide prognostication or therapeutic decisions.
Relapse-free survival of patients with and without cytogenetic abnormalities at CR, stratified by MRD status
Relapse-free survival of patients with and without cytogenetic abnormalities at CR, stratified by MRD status
Overall survival of patients with and without cytogenetic abnormalities at CR, stratified by MRD status
Overall survival of patients with and without cytogenetic abnormalities at CR, stratified by MRD status
Faderl:Celgene Corp.: Other: Advisory Board. Burger:Pharmacyclics LLC, an AbbVie Company: Research Funding. Konopleva:Novartis: Research Funding; AbbVie: Research Funding; Stemline: Research Funding; Calithera: Research Funding; Threshold: Research Funding. Cortes:Pfizer: Consultancy, Research Funding; BMS: Consultancy, Research Funding; Teva: Consultancy, Research Funding; Novartis: Consultancy, Research Funding; ARIAD Pharmaceuticals Inc.: Consultancy, Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal