Abstract
Introduction:
pPCLis a rare form of multiple myeloma (MM) with very poor outcome. Current strategies including novel agents and transplantation (auto or allo-HSCT) may improve survival but the prognosis remains poor. The IFM recently published results from a prospective study incorporating tandem auto/allo-HSCT or auto/auto and maintenance after alternate induction (doxorubicin-bortezomib-cyclophosphamide and dexamethasone: PAD/VCD) in 40 pPCL patients. We report here outcomes of patients who effectively underwent second transplant (auto or allo) with long-term follow-up.
Methods:
24 over 40 patients actually received second transplant:allo-HSCT (as per protocol) forpatients< 60 years-old with a donor, second auto-HSCT for others. At time of second transplant, response rates were: sCR (n=1), CR (n=8), VGPR (n=10), PR (n=5) (cf table).
7 patients received high-dose melphalan (HDM)/auto-HSCT followed by maintenance consisting in Bortezomib-Lenalidomide-Dex (VRD) every 3 months and Lenalidomideduring other months for 1 year. 17 others patients underwent allo-HSCT: syngenic(n=1) or reduced intensity conditioning (RIC)-Allo (n=16). The conditioning regimens for allo were: fludarabin-busulfan-antithymoglobulin(n=13), fludarabin-melphalan(n=1), bortezomib-fludarabin-melphalan(n=2) and HDM for syngenic(n=1). They were grafted with peripheral blood stem cells except 1 who received bone marrow and 1 cord blood unit, from related (n=9) or HLA-matched unrelated donors (n=8): median infused CD34 cells was 6.8 106/kg [3-8.5]; all pts receivedciclosporine+/-methotrexate as GvHD prophylaxis.
Results:
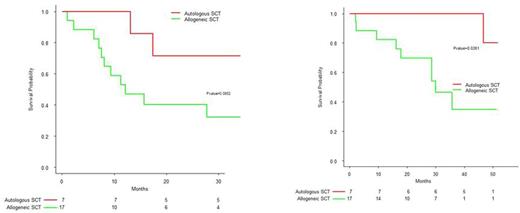
As of June 2016, median follow-up from diagnosis was44.7months[41.0;57.6], 10 patientswerestillalive.MedianPFS for allo and auto ptswas18.5months[17.8;-] and 50.1months[25.6;-]respectively ;medianOSwas39.3months[26.6;-] and notreached,respectively.Inlandmarkanalysisfromdate of second transplant, PFSwas12.2 months [8.1 ;-] for allo pts and 40.0months[17.7;-] for auto pts (p=0.085); OSwas30.0months[28.6;-] and notreached,respectively(p=0.026). 2 allo ptsdiedbefored100evaluation ; 18 patients (75%)werein VGPR orbetter : sCR=2, RC=8, VGPR=8 ; 4 patients (17%)werein PR.
For allo-HSCT : 6patients developed acute GvHD which responded to steroid in 5 cases and 1 was steroid-resistant and responded secondary to anti-IL2Rα antibody;5 patients experienced chronic GVHD (mild [n=4], extensive [n=1]).3 patients received 3 months after allo as immunomodulation, donor lymphocyte infusions plusLenalidomide-Cyclophosphamide-Fludarabin(n=1), Bortezomib (n=1) and Lenalidomide (n=1): one patient in PR achieved CR and the two others in VGPR maintained their response.Day 100 treatment-related mortality was 12%: 2 patients had Epstein-Barr virus reactivation, with 1 experiencing neuromeningeal relapse of pPCL and 1 concomitant disseminated toxoplasmosis.
For auto-HSCT: as per protocol, 6 received a second HDM/auto, and 1 declined. Consolidation with VRD/Len was initiated in these 7 patients: 4 receiving the planned 1 year of treatment and 3 discontinuing after 4, 8, and 10 months as a result of prolonged cytopenia.
Conclusion:
This is the first large prospective trial for pPCL patients including tandem auto/auto-HSCT or tandem auto/RIC-allo after induction including proteasome inhibitor. This approach is feasible, and induces high response rate but relapses remain frequent especially after RIC-allo. Tandem auto/auto plus maintenance may be superior to tandem auto/alloregarding PFS and OS. In a future trial, we will investigate tandem auto/auto after induction incorporating novel proteasome inhibitor, anti-CD38 monoclonal antibody and Lenalidomide, and followed by consolidation and prolonged maintenance.
Table: characteristics of Patients
Roussel:amgen: Consultancy; Janssen: Consultancy; celgene: Consultancy. Karlin:janssen-cilag: Consultancy, Honoraria; amgen: Consultancy, Honoraria; celgene: Consultancy, Honoraria; Bristol: Consultancy; takeda: Consultancy. Hulin:Janssen: Honoraria; Bristol: Honoraria; Amgen: Honoraria; takeda: Honoraria; celgene: Honoraria. Macro:sanofi: Consultancy; Novartis: Honoraria; Bristol: Consultancy; Amgen: Consultancy, Honoraria; Janssen: Consultancy, Honoraria; Celgen: Consultancy, Honoraria. Belhadj:janssen: Consultancy; novartis: Consultancy. Chaleteix:Amgen: Honoraria; Janssen: Honoraria. Leleu:TEVA: Membership on an entity's Board of Directors or advisory committees; Novartis: Honoraria; LeoPharma: Honoraria; Pierre Fabre: Honoraria; Amgen: Honoraria; Bristol-Myers Squibb: Honoraria; Takeda: Honoraria; Celgene: Honoraria; Janssen: Honoraria. Garderet:Takeda: Consultancy; Amgen: Consultancy; BMS: Consultancy, Honoraria; Novartis: Consultancy. Choquet:Janssen: Consultancy; Celgene: Consultancy. Fornecker:Roche: Consultancy; Takeda: Consultancy; Gilead: Consultancy; Mundipharma: Speakers Bureau. Facon:Novartis: Consultancy; Amgen: Consultancy, Speakers Bureau; Millenium/Takeda: Consultancy; Celgene: Consultancy, Speakers Bureau; Janssen: Consultancy, Speakers Bureau; Bristol: Consultancy; Karyopharm: Consultancy. Moreau:Bristol-Myers Squibb: Honoraria; Janssen: Honoraria, Speakers Bureau; Takeda: Honoraria; Novartis: Honoraria; Amgen: Honoraria; Celgene: Honoraria.
Author notes
Asterisk with author names denotes non-ASH members.


This feature is available to Subscribers Only
Sign In or Create an Account Close Modal