Key Points
SFK signaling is sufficient to mediate M fate instruction by CSF-1.
c-Src activity is sufficient to enforce M fate, both in myeloid progenitors and in nonmyeloid cells.
Abstract
Controlled regulation of lineage decisions is imperative for hematopoiesis. Yet, the molecular mechanisms underlying hematopoietic lineage choices are poorly defined. Colony-stimulating factor 1 (CSF-1), the cytokine acting as the principal regulator of monocyte/macrophage (M) development, has been shown to be able to instruct the lineage choice of uncommitted granulocyte M (GM) progenitors toward an M fate. However, the intracellular signaling pathways involved are unknown. CSF-1 activates a multitude of signaling pathways resulting in a pleiotropic cellular response. The precise role of individual pathways within this complex and redundant signaling network is dependent on cellular context, and is not well understood. Here, we address which CSF-1–activated pathways are involved in transmitting the lineage-instructive signal in primary bone marrow-derived GM progenitors. Although its loss is compensated for by alternative signaling activation mechanisms, Src family kinase (SFK) signaling is sufficient to transmit the CSF-1 lineage instructive signal. Moreover, c-Src activity is sufficient to drive M fate, even in nonmyeloid cells.
Introduction
Tightly controlled lineage decisions and their modulation are a prerequisite for normal and emergency hematopoiesis, allowing regulated and demand-driven production of all mature blood cells. Lineage commitment of multipotent cells could either be induced by extrinsic factors such as cytokines, or by intrinsic mechanisms including stochastic regulation of transcription factors or other regulatory molecules. Both extrinsic and intrinsic factors may actively induce lineage commitment (“instruct”) or be permissive (“select”) for one lineage. The instructive vs selective model in orchestrating hematopoietic fate has been intensely debated, especially regarding the role of cytokines. In the selective model, lineage commitment occurs independently of cytokines by a spontaneous stochastic process. The cytokines’ function is then to only provide selective survival and/or proliferation signals after lineage choice.1 The instructive model postulates that cytokines actively drive uncommitted cells toward a particular fate by inducing lineage-instructive signaling.2,3
The hematopoietic cytokine colony-stimulating factor 1 (CSF-1), also known as macrophage (M) colony-stimulating factor, is central to the survival, proliferation, and differentiation of the monocyte/M lineage, which is critical for innate and adaptive immunity and development. CSF-1 can instruct the lineage choice of bipotent bone marrow (BM)-derived granulocyte M progenitors (GMPs) toward an M fate.4 However, the signaling pathways orchestrating this lineage instruction remain elusive. The pleiotropic actions of CSF-1 are mediated through the CSF-1 receptor (CSF-1R), a type III receptor tyrosine kinase. Six tyrosine residues of the CSF-1R cytoplasmic domain have been reported to be phosphorylated upon CSF-1 binding and receptor dimerization (murine: Y559, Y697, Y706, Y721, Y807, and Y974). Two more (Y544 and Y921) have been described to be phosphorylated in an oncogenic receptor form. Once phosphorylated, most of these tyrosines act as docking sites for adaptor proteins initiating downstream signaling events. These include for example, Src family kinase (SFK) signaling via Y559,5 phosphatidylinositol 3-kinase (PI3K)/Akt signaling via Y721,6,7 and mitogen-activated protein kinase (MAPK) signaling via Y6978 (see supplemental Figure 1, available on the Blood Web site).
The exact role of individual pathways in mediating specific CSF-1–induced cellular responses remains poorly understood. Contradictory results have been observed when studying the same CSF-1R residue or signaling pathway,5,9-12 likely due to the use of cell lines, which do not reflect the molecular milieu (eg, signaling components, transcription factors, micro RNAs, and chromatin) of relevant primary cells. Moreover, epigenetic states and intracellular components mediating specific cytokine responses may only be active or present during specific time windows of differentiation. It is therefore crucial to study CSF-1R function in the correct cellular context.
Here, we use a combination of loss- and gain-of-function experiments in combination with single-cell differentiation readouts in primary uncommitted progenitors that physiologically respond to CSF-1. We identify SFK signaling as a sufficient component of CSF-1R signaling to induce M fate in GMPs, and even in nonmyeloid cells.
Methods
Mice
Csf1r+/− mice13 backcrossed on the C57BL/6J CD45.2 background for 10 generations and C57BL/6J-Gpi1a Ptprca KitW-41JJ (c-kitW41/W41 [W41], CD45.1) mice14 were used. Experiments were performed according to Swiss federal law and institutional guidelines of Eidgenössische Technische Hochschule Zurich, and approved by the local Animal Ethics Committee of Basel-Stadt (approval numbers 2655 and 2688).
Isolation and transplantation of fetal liver cells
Csf1r+/− × Csf1r+/− matings were used to generate Csf1r−/− embryos. Single-cell suspensions of individual fetal livers from embryonic day 14.5 (E14.5) embryos were isolated and strained through a 35 μm mesh. After genotyping by flow cytometry, 2 × 107Csf1r−/− fetal liver cells were IV injected into 8- to 10-week-old W41 mice.
Cell cultures and colony assays
Fluorescence-activated cell sorter (FACS)-purified myeloid progenitor cells were cultured in serum-free medium (Stemcell Technologies) containing indicated cytokines. All cytokines used were murine and obtained from PeproTech. Inhibitors used were Ly294002 (50 µM), U0126 (10 μM) (both Cell Signaling Technology), PD0325901 (1 μM) (Selleck), and SU6656 (2 μM) (Calbiochem). Methylcellulose assays were performed with MethoCult M3434 or MethoCult M3234 (Stemcell Technologies). Colonies were scored after 5 to 7 days according to morphology and live in-culture antibody stainings.15 Small colonies consisted of <50 cells. C10 cells were cultured as described.16
Plasmid constructs
Time-lapse imaging and cell tracking
Time-lapse imaging and live in-culture antibody staining to detect differentiated cells was performed as described.4,15 Single-cell tracking was performed using The Tracking Tool.24-27 Pedigrees and cells with lost identity were excluded from analysis. Because sister cells within pedigrees behaved synchronously with regards to differentiation, pedigrees were sometimes only partly tracked (supplemental Figure 5).
Statistical analyses
A Cochran-Mantel-Haenszel test for repeated tests of independence was used to determine the significance of the difference of proportions across replicates. All data are plotted as mean ± standard error of the mean using Prism (GraphPad Software Inc).
Results
Chemical inhibition of signaling pathways is not sufficient to block CSF-1–instructed M differentiation
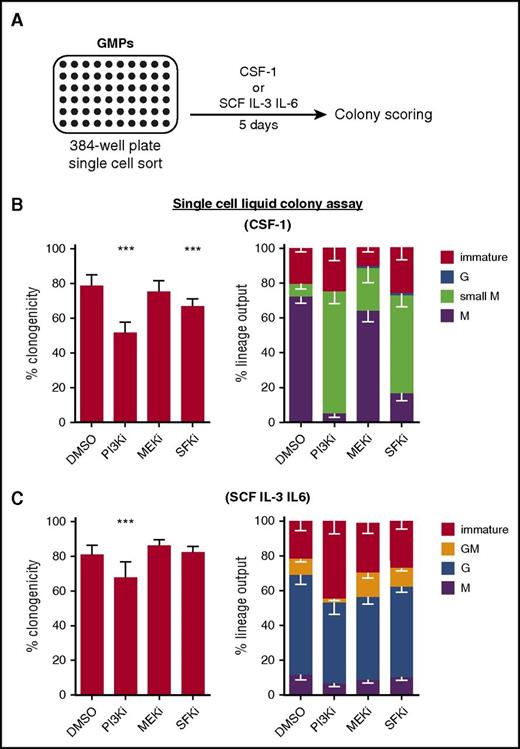
Previous studies have described the involvement of several major signaling pathways, including PI3K/Akt, SFKs, and MAPKs in CSF-1–mediated survival and differentiation.28 To identify CSF-1–induced signaling pathways involved in mediating lineage instruction, we cultured freshly isolated single GMPs with either CSF-1, instructing M-lineage fate, or stem cell factor (SCF) interleukin 3 (IL-3) IL-6, allowing all myeloid lineage fates, in the absence or presence of signaling inhibitors (Figure 1A). Although the PI3K inhibitor (PI3Ki) Ly294002 and the SFK inhibitor (SFKi) SU6656 decreased the clonogenicity of single-plated GMPs in CSF-1 condition (Figure 1B, left), none of the tested inhibitors decreased the total M output (M and small M colonies, consisting of more or less than 50 cells, respectively), as compared with the DMSO control (Figure 1B, right). In the case of PI3Ki and SFKi, and to a lesser extent for the MEK inhibitor (MEKi) (U0126), most CSF-1–induced M colonies were much smaller than in the DMSO control. This suggests that these pathways are mainly involved in survival and proliferation, but are not essential for lineage instruction. In permissive cultures, only PI3Ki decreased the clonogenicity and M output as compared with the control (Figure 1C).
Chemical inhibition of signaling pathways is not sufficient to block CSF-1–mediated lineage instruction. (A) GMPs were sorted as single cells into 384-well plates containing medium with indicated cytokines, 10% FCS, and different signaling inhibitors. Colonies were identified after 5 days of culture according to morphology and lineage-specific markers (F4/80 for M and Ly6G for G). Immature colonies (including small colonies) consisted of cells negative for both markers. (B) Clonogenicity (ie, capability to form colonies existing of ≥2 cells) and lineage output of cells cultured in the presence of CSF-1 and indicated inhibitors (PI3Ki = 50 μM Ly294002; MEKi = 10 μM U0126; and SFKi = 2 μM SU6656). (C) Clonogenicity and lineage output of cells cultured in the presence of SCF, IL-3 and IL-6, and indicated inhibitors (n = 5 experiments). P values are shown as follows: ***P < .001 compared with DMSO control. DMSO, dimethyl sulfoxide; FCS, fetal calf serum.
Chemical inhibition of signaling pathways is not sufficient to block CSF-1–mediated lineage instruction. (A) GMPs were sorted as single cells into 384-well plates containing medium with indicated cytokines, 10% FCS, and different signaling inhibitors. Colonies were identified after 5 days of culture according to morphology and lineage-specific markers (F4/80 for M and Ly6G for G). Immature colonies (including small colonies) consisted of cells negative for both markers. (B) Clonogenicity (ie, capability to form colonies existing of ≥2 cells) and lineage output of cells cultured in the presence of CSF-1 and indicated inhibitors (PI3Ki = 50 μM Ly294002; MEKi = 10 μM U0126; and SFKi = 2 μM SU6656). (C) Clonogenicity and lineage output of cells cultured in the presence of SCF, IL-3 and IL-6, and indicated inhibitors (n = 5 experiments). P values are shown as follows: ***P < .001 compared with DMSO control. DMSO, dimethyl sulfoxide; FCS, fetal calf serum.
These results have indicated that PI3K, SFK, or MEK/extracellular signal-regulated kinase (ERK) signaling are not essential for lineage instruction. However, small molecule inhibitors can have off-target effects, or rather reduce than abolish signaling activity. We therefore used genetic receptor manipulations for complete and specific knockouts of signaling nodes.
Assessing the function of ectopically expressed genes in uncommitted GMPs
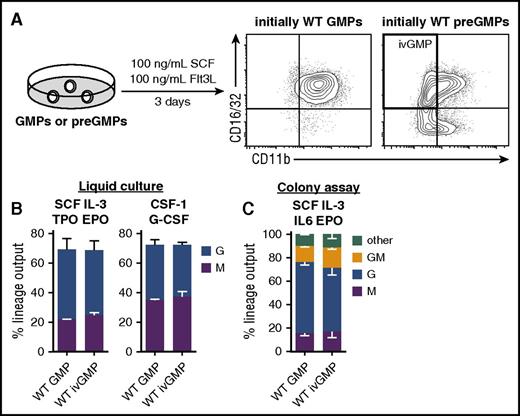
GMPs are a transient bipotent population that quickly differentiates into unipotent cells when isolated from the BM. This impedes the ex vivo genetic manipulation of GMPs before commitment, and hence differentiation occurs. We therefore established an in vitro differentiation system that gives rise to GMPs and simultaneously allows time for genetic manipulation. To this end, immediate precursors of GMPs (preGMPs)29 were FACS-purified (supplemental Figure 2) and cultured in the presence of SCF and Flt3L (Figure 2A). As a control, GMPs were cultured in the same cytokine conditions. When re-analyzing the cells via flow cytometry after 3 days in culture, preGMPs (originally sorted as CD16/32low CD11bneg) gave rise to a CD16/32hi CD11bpos and a CD16/32hi CD11bneg population, with the latter being immunophenotypically identical to BM-derived GMPs (Figure 2A). In contrast, cultured GMPs retained high CD16/32 expression, and virtually all cells expressed CD11b (Figure 2A), which is an early myeloid differentiation marker. The lineage potential of these CD16/32hi CD11bneg in vitro-derived GMPs (ivGMPs) was comparable to freshly isolated GMPs in liquid or semi-solid cultures with different cytokine combinations (Figure 2B-C). This makes ivGMPs an appropriate cellular model system to study the effects of exogenously expressed genes in a primary uncommitted bipotent cellular context.
Derivation and lineage potential of ivGMPs. (A) GMPs or preGMPs isolated from WT BM were cultured in presence of 100 ng/mL SCF and Flt3L for 3 days, and then analyzed for CD16/32 and CD11b expression via flow cytometry. Representative FACS plots are shown. (B) Freshly isolated GMPs and ivGMPs were cultured in either SCF, IL-3, TPO, EPO, 10% FCS or CSF-1, G-CSF, and 10% FCS, and analyzed for M (CD11bpos F4/80pos Ly6Gneg) and G (CD11bpos F4/80neg Ly6Gpos) output via flow cytometry after 4 to 5 days (n = 3 to 6). (C) Freshly isolated GMPs and ivGMPs were cultured in methylcellulose containing SCF, IL-3, IL-6, and EPO (M3434). After 5 to 7 days, colonies were enumerated and identified according to morphology (clonogenicity WT GMP = 48% ± 7.5% SD; ivGMPs = 45% ± 6% SD; n = 3 experiments). No significant difference was detected. EPO, erythropoietin; SD, standard deviation; TPO, thrombopoietin.
Derivation and lineage potential of ivGMPs. (A) GMPs or preGMPs isolated from WT BM were cultured in presence of 100 ng/mL SCF and Flt3L for 3 days, and then analyzed for CD16/32 and CD11b expression via flow cytometry. Representative FACS plots are shown. (B) Freshly isolated GMPs and ivGMPs were cultured in either SCF, IL-3, TPO, EPO, 10% FCS or CSF-1, G-CSF, and 10% FCS, and analyzed for M (CD11bpos F4/80pos Ly6Gneg) and G (CD11bpos F4/80neg Ly6Gpos) output via flow cytometry after 4 to 5 days (n = 3 to 6). (C) Freshly isolated GMPs and ivGMPs were cultured in methylcellulose containing SCF, IL-3, IL-6, and EPO (M3434). After 5 to 7 days, colonies were enumerated and identified according to morphology (clonogenicity WT GMP = 48% ± 7.5% SD; ivGMPs = 45% ± 6% SD; n = 3 experiments). No significant difference was detected. EPO, erythropoietin; SD, standard deviation; TPO, thrombopoietin.
Derivation of CSF-1R–deficient GMPs
During hematopoiesis, GMPs give rise to granulocytes (Gs) and Ms.30 In vitro, GMP lineage choice can be instructed toward Ms or Gs by CSF-1 or G-CSF, respectively.4 They therefore represent an ideal cellular model system to study CSF-1–induced lineage choice and M differentiation. However, GMPs endogenously express the CSF-1R and are therefore unsuitable for studying the effect of exogenously expressed CSF-1R mutants. Hence, we sought to obtain GMPs from a CSF-1R–deficient mouse model. Because C57Bl/6 Csf1r−/− mice rarely survive into adulthood,31 we took advantage of W41 mutant mice, which accept syngenic c-kit+/+ hematopoietic stem and progenitor cells (HSPCs), allowing long-term engraftment and amplification of donor cells without prior irradiation.32,33 In order to test W41 mice as recipients for Csf1r−/− cells, we transplanted adult W41 mice with unfractionated E14.5 Csf1r−/− or wild-type (WT) (Csf1r+/+ littermate control) fetal liver cells (Figure 3A). Donor contribution increased steadily over time irrespective of genotype and with no difference in kinetics (Figure 3B, left). Likewise, no difference was detected in donor contribution to myeloid progenitors in BM (Figure 3B, right), demonstrating that CSF-1R is not required for the engraftment and differentiation of HSPCs in vivo. Csf1r−/− GMPs purified from W41 mice had the same lineage potential as WT GMPs derived from either C57BL/6 mice or from W41 mice transplanted with WT fetal liver cells (Figure 3C, left). As expected, Csf1r−/− GMPs were, however, unable to form colonies in response to CSF-1 (Figure 3C, right).
Derivation and lineage potential of BM-derived Csf1r−/−progenitors. (A-B) W41 (CD45.1) mice were IV transplanted with 2 × 107 E14.5 fetal liver cells of WT or Csf1r−/− (CD45.2) genotype. Donor contribution was checked at various timepoints in recipients’ as follows: (B, left) peripheral blood (n = 3 to 6 per timepoint per genotype) or (B, right) BM (GMPs) (n = 3 per genotype) via flow cytometry. (C) Freshly isolated GMPs from C57Bl/6 (WT), W41 transplanted with WT fetal liver cells (WT [transpl.]), or W41 transplanted with Csf1r−/− fetal liver cells (Csf1r−/−) were cultured in methylcellulose containing either SCF, IL-3, IL-6, and EPO (left) or CSF-1 (right). After 5 to 7 days, colonies were enumerated and identified according to morphology (clonogenicity SCF IL-3 IL-6 EPO: WT = 45% ± 6.4% SD, WT [transpl.] = 47% ± 7.2% SD, Csf1r−/− = 43% ± 4.4% SD; and clonogenicity CSF-1: WT = 46% ± 5.1% SD, WT [transpl.] = 47% ± 5% SD, Csf1r−/− = 0, n = 3 per genotype per condition). P values are shown as follows: ***P < .001 compared with WT. Transpl, transplanted.
Derivation and lineage potential of BM-derived Csf1r−/−progenitors. (A-B) W41 (CD45.1) mice were IV transplanted with 2 × 107 E14.5 fetal liver cells of WT or Csf1r−/− (CD45.2) genotype. Donor contribution was checked at various timepoints in recipients’ as follows: (B, left) peripheral blood (n = 3 to 6 per timepoint per genotype) or (B, right) BM (GMPs) (n = 3 per genotype) via flow cytometry. (C) Freshly isolated GMPs from C57Bl/6 (WT), W41 transplanted with WT fetal liver cells (WT [transpl.]), or W41 transplanted with Csf1r−/− fetal liver cells (Csf1r−/−) were cultured in methylcellulose containing either SCF, IL-3, IL-6, and EPO (left) or CSF-1 (right). After 5 to 7 days, colonies were enumerated and identified according to morphology (clonogenicity SCF IL-3 IL-6 EPO: WT = 45% ± 6.4% SD, WT [transpl.] = 47% ± 7.2% SD, Csf1r−/− = 43% ± 4.4% SD; and clonogenicity CSF-1: WT = 46% ± 5.1% SD, WT [transpl.] = 47% ± 5% SD, Csf1r−/− = 0, n = 3 per genotype per condition). P values are shown as follows: ***P < .001 compared with WT. Transpl, transplanted.
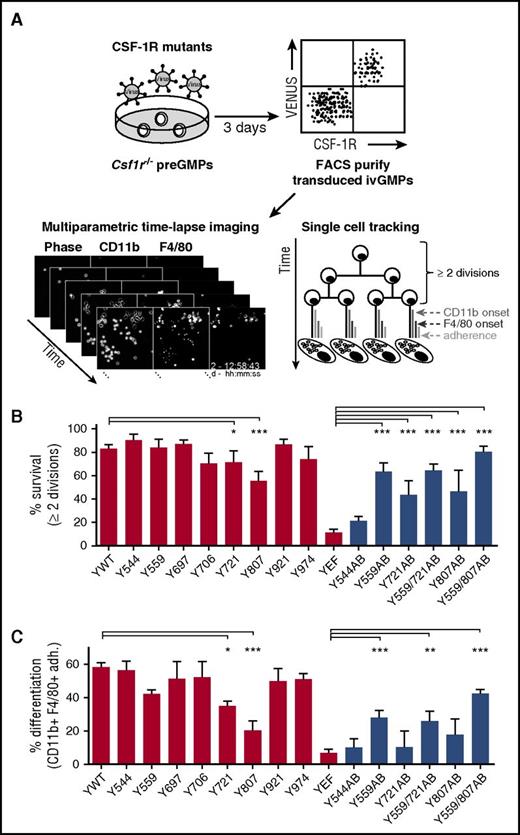
CSF-1R tyrosine residue 559 is sufficient for CSF-1–mediated lineage choice
The CSF-1R has 8 intracellular tyrosine residues that initiate downstream signaling pathways, mediating its pleiotropic effects (supplemental Figure 128 ). To gain insight into the function of individual CSF-1R tyrosines and their activated pathways in GMPs, we reintroduced CSF-1R tyrosine mutants17,18 into Csf1r−/− preGMPs by lentiviral transduction (supplemental Figure 3A-C). We then analyzed by continuous live cell imaging how the lack of tyrosines affected CSF-1–mediated cell fates, including survival, proliferation, and differentiation, of individual ivGMPs (Figure 4A; Video 1).24-26 Transduced ivGMPs expressing similar CSF-1R levels as WT cells were FACS purified to avoid potential overexpression artifacts (supplemental Figure 4A). Transducing Csf1r−/− cells with the WT CSF-1R (Ywt) restored CSF-1–induced M colony formation, demonstrating that CSF-1–mediated survival, proliferation, and differentiation was rescued (supplemental Figure 4B and 5A). Moreover, time-lapse imaging revealed that the rescued cells were instructed toward Ms, with similar differentiation kinetics as compared with published data from BM-derived GMPs4 (data not shown). To reveal the contributions of individual tyrosines to progenitor survival/proliferation, we determined how many transduced starting cells divided at least 2 consecutive times (corresponding to ∼24 hours), after which most GMPs are committed.4 As expected, virtually all cells reconstituted with an CSF-1R lacking all 8 tyrosine residues (YEF) did not divide more than once and died within the first 2 generations (Figure 4B; supplemental Figure 5C). In contrast, the majority of starting cells rescued with the WT CSF-1R (Ywt) survived/proliferated (Figure 4B). Only the mutation of Y721 or Y807 resulted in significantly lower survival in response to CSF-1 as compared with Ywt (Figure 4B; supplemental Figure 6A), which is in agreement with previous studies of CSF-1–mediated survival in Ms.17,28 Although being quite heterogeneous among individual cells, none of the single tyrosine-mutated receptors drastically influenced the average cell-cycle times of undifferentiated cells as compared with Ywt (data not shown). Next, we looked at the efficiency of individual CSF-1R signaling mutants to rescue CSF-1–instructed M differentiation. Colonies that did not form Ms included those that had either died prior to differentiation or produced cells that were not classified as Ms (data not shown). Compared with Ywt, the mutation of Y721 or Y807 resulted in significantly less M differentiation of transduced cells (Figure 4C; supplemental Figure 6B). Whether this decreased differentiation capacity was solely due to decreased survival or additionally to impaired differentiation, cannot be concluded with certainty. However, CSF-1Rs carrying only Y721 (Y721AB) or Y807 (Y807AB) were not sufficient to induce differentiation upon CSF-1 exposure, thus pointing toward a survival-specific effect (Figure 4C).
Time-lapse imaging reveals CSF-1R Y559 to be sufficient for CSF-1–mediated lineage choice. (A) Csf1r−/− preGMPs were FACS purified and transduced with lentiviruses encoding different CSF-1R mutants (supplemental Figure 3B-C). Transduced cells were purified, time-lapse imaged during CSF-1–mediated differentiation (Video 1), and tracked. (B) Percentage of surviving colonies. Dying colonies were defined as those with <2 cell divisions of undifferentiated cells. (C) Percentage of differentiating colonies (colonies with CD11bpos F4/80pos adherent cells at any timepoint) of all starting cells. Displayed Ywt and YEF data were pooled from all experiments. Total movies/colonies analyzed: Ywt (9/247), Y544F (3/55), Y559F (4/75), Y697F (3/45), Y706F (3/52), Y721F (3/81), Y807F (3/141), Y921F (4/114), Y974F (2/51), YEF (4/140), Y544AB (2/64), Y559AB (3/138), Y721AB (2/67), Y559/721AB (4/99), Y807AB (2/72), and Y559/807AB (4/115). For pairwise comparison, see supplemental Figures 6 and 7. Red bars represent single tyrosine mutants and controls, blue bars represent AB mutants. P values (calculated for data pairs) are shown as follows: *P < .05; **P < .01; ***P < .001 compared with Ywt (single tyrosine mutants) or YEF (AB mutants). AB, add back.
Time-lapse imaging reveals CSF-1R Y559 to be sufficient for CSF-1–mediated lineage choice. (A) Csf1r−/− preGMPs were FACS purified and transduced with lentiviruses encoding different CSF-1R mutants (supplemental Figure 3B-C). Transduced cells were purified, time-lapse imaged during CSF-1–mediated differentiation (Video 1), and tracked. (B) Percentage of surviving colonies. Dying colonies were defined as those with <2 cell divisions of undifferentiated cells. (C) Percentage of differentiating colonies (colonies with CD11bpos F4/80pos adherent cells at any timepoint) of all starting cells. Displayed Ywt and YEF data were pooled from all experiments. Total movies/colonies analyzed: Ywt (9/247), Y544F (3/55), Y559F (4/75), Y697F (3/45), Y706F (3/52), Y721F (3/81), Y807F (3/141), Y921F (4/114), Y974F (2/51), YEF (4/140), Y544AB (2/64), Y559AB (3/138), Y721AB (2/67), Y559/721AB (4/99), Y807AB (2/72), and Y559/807AB (4/115). For pairwise comparison, see supplemental Figures 6 and 7. Red bars represent single tyrosine mutants and controls, blue bars represent AB mutants. P values (calculated for data pairs) are shown as follows: *P < .05; **P < .01; ***P < .001 compared with Ywt (single tyrosine mutants) or YEF (AB mutants). AB, add back.
In CSF-1R YEF AB mutants, tyrosine residues are added back to the YEF mutant.18 Single AB mutants Y559AB, Y721AB, and Y807AB partially restored CSF-1–mediated survival, but only Y559AB significantly rescued M differentiation compared with YEF (Figure 4B-C, black bars; supplemental Figure 7A-B). This indicates that CSF-1R Y559 plays a major role in survival- and differentiation-inducing signaling in primary myeloid progenitors. Adding back Y721 to the Y559AB mutant did not increase the number of surviving and differentiating colonies as compared with Y559AB. However, adding back Y807 in addition to Y559 (Y559/807AB) restored Ywt levels of survival and further increased differentiation as compared with Y559AB. Cells rescued with Y559/807AB had similar differentiation kinetics and cycling times as compared with Ywt (13.4 ± 0.2 hours for Ywt and 14.5 ± 0.3 hours for Y559/807AB) (supplemental Figure 5A-B).
Taken together, probing the role of functional CSF-1R tyrosine residues in primary myeloid progenitors revealed Y559 to be sufficient for CSF-1–mediated survival and M differentiation, and hence suggests that lineage-instructive signaling can be activated via this residue. The addition of Y807 to Y559 elevated this effect, whereas Y807 alone only contributed to survival but not differentiation.
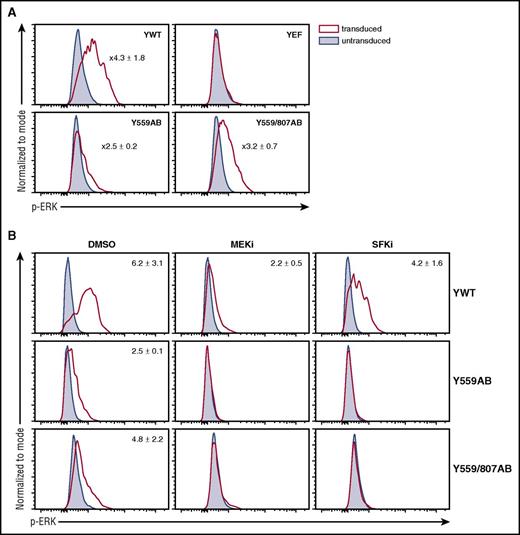
Y559 signals via SFK-MEK/ERK
CSF-1R Y559 is described to activate SFKs,18,34-36 whereas activation loop Y807 is proposed to have a role in CSF-1R structural activation rather than inducing signaling pathways.18 A potential signaling pathway downstream of SFKs involved in lineage choice is the MEK/ERK pathway.37,38 It has previously been shown that Y559 activates SFK and MEK/ERK signaling in Ms.18 To test whether ERK is activated through Y559(/807) in primary progenitors, we performed phospho-flow with Csf1r−/− ivGMPs expressing Ywt, YEF, Y559AB, or Y559/807AB. Stimulation with CSF-1 led to ERK activation with both AB mutants and could be blocked using an SFK inhibitor, demonstrating that Y559 signals through MEK/ERK via SFKs (Figure 5A-B).
Y559 activates ERK via SFK signaling. (A) Csf1r−/− ivGMPs expressing indicated mutants were generated as described in Figure 4. Cells were stimulated with CSF-1 for 5 minutes and then stained against p-ERK. (B) Cells were pre-incubated with either a MEK (10 µM U0126) or SFK (2 µM SU6656) inhibitor for 1 hour prior to CSF-1 stimulation. Representative FACS plots of transduced and untransduced cells within the same sample are shown. Numbers represent average fold values ± SD (n = 3). p-ERK, phosphorylated ERK.
Y559 activates ERK via SFK signaling. (A) Csf1r−/− ivGMPs expressing indicated mutants were generated as described in Figure 4. Cells were stimulated with CSF-1 for 5 minutes and then stained against p-ERK. (B) Cells were pre-incubated with either a MEK (10 µM U0126) or SFK (2 µM SU6656) inhibitor for 1 hour prior to CSF-1 stimulation. Representative FACS plots of transduced and untransduced cells within the same sample are shown. Numbers represent average fold values ± SD (n = 3). p-ERK, phosphorylated ERK.
We performed time-lapse movies with Csf1r−/− ivGMPs expressing Y559/807AB to evaluate whether SFK and MEK inhibitors could also block CSF-1–mediated differentiation. Indeed, the presence of both a MEK or SFK inhibitor significantly decreased M output, further suggesting that Y559 activates an SFK-MEK/ERK signaling axis to induce commitment and differentiation (supplemental Figure 9).
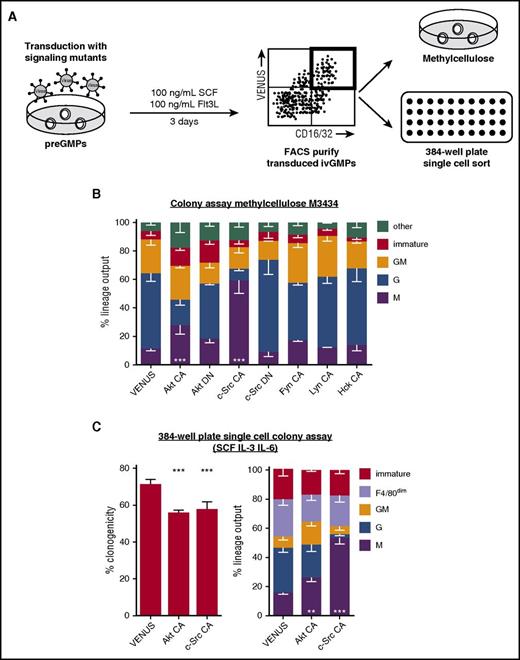
c-Src activity is sufficient to drive M differentiation of GMPs in the absence of CSF-1
To date, no phospho-specific antibodies discriminating individual SFK isoforms exist. The specific SFK(s) activated by CSF-1R can therefore not be determined in individual HSPCs. However, to probe which specific SFK is sufficient to drive M differentiation independently of CSF-1, we overexpressed constitutive active variants of different SFK members (c-Src, Fyn, Lyn, and Hck) and potential downstream signaling target Akt in ivGMPs. Transduced ivGMPs were sorted and their lineage output assessed (Figure 6A). To facilitate unambiguous colony scoring, lineage-specific live antibody staining was applied (supplemental Figure 8). Forced expression of constitutive active c-Src and Akt resulted in significantly increased M output, largely at the expense of granulocytic colonies (Figure 6B-C). The liquid colony assay revealed a decreased clonogenicity of cells transduced with constitutive active forms of either Akt or c-Src (Figure 6C, left). Consequently, selective effects might contribute to the observed lineage skewing. The nature of F4/80dim cells remained inconclusive, but they differed from Ms not only in the expression level of F4/80, but also by their motility (determined by colony spread) and morphology. However, the frequency of these colonies remained constant under all conditions tested. Constitutive active mutants of other SFKs Fyn, Lyn, and Hck did not lead to significantly increased M output, suggesting a specific role of c-Src in M lineage choice and differentiation. Presence of MEK inhibitors did not reduce the elevated M differentiation induced by c-Src (supplemental Figure 10), suggesting the involvement of complementary pathways to induce M lineage choice.
Enforced c-Src activity is sufficient to drive CSF-1–independent M differentiation of GMPs at the expense of granulocytic fate. (A) preGMPs were freshly isolated, and cultured in 100 ng/mL SCF and Flt3L while being transduced with lentiviruses encoding variants of signaling molecules CA or DN. After 3 days, transduced ivGMPs were either sorted and cultured in permissive colony assay medium (M3434, containing SCF, IL-3, IL-6, and EPO), or sorted as single cells into 384-well plates containing SCF, IL-3, IL-6, and 10% FCS. After 5 to 7 days in culture, colonies were enumerated and identified using lineage-specific markers (F4/80 for M and Ly6G for G). Immature colonies consisted of cells negative for both markers. (B) Lineage output in methylcellulose colony assay (clonogenicity VENUS = 49% ± 7.2% SD, Akt CA = 33% ± 7.4% SD, Akt DN = 39% ± 8.4% SD, c-Src CA = 35% ± 9.2%, c-Src DN = 51% ± 7.4% SD, Fyn CA = 45% ± 9.4% SD, Lyn CA = 52% ± 6.4% SD, and Hck CA = 46% ± 7.3% SD; n = 3 to 6 experiments). (C) Clonogenicity and lineage output of single cells cultured in 384-well plates (n = 4 to 5 experiments). P values are shown as follows: **P < .01; ***P < .001 compared with VENUS control (only calculated for M). CA, constitutive active; DN, dominant negative.
Enforced c-Src activity is sufficient to drive CSF-1–independent M differentiation of GMPs at the expense of granulocytic fate. (A) preGMPs were freshly isolated, and cultured in 100 ng/mL SCF and Flt3L while being transduced with lentiviruses encoding variants of signaling molecules CA or DN. After 3 days, transduced ivGMPs were either sorted and cultured in permissive colony assay medium (M3434, containing SCF, IL-3, IL-6, and EPO), or sorted as single cells into 384-well plates containing SCF, IL-3, IL-6, and 10% FCS. After 5 to 7 days in culture, colonies were enumerated and identified using lineage-specific markers (F4/80 for M and Ly6G for G). Immature colonies consisted of cells negative for both markers. (B) Lineage output in methylcellulose colony assay (clonogenicity VENUS = 49% ± 7.2% SD, Akt CA = 33% ± 7.4% SD, Akt DN = 39% ± 8.4% SD, c-Src CA = 35% ± 9.2%, c-Src DN = 51% ± 7.4% SD, Fyn CA = 45% ± 9.4% SD, Lyn CA = 52% ± 6.4% SD, and Hck CA = 46% ± 7.3% SD; n = 3 to 6 experiments). (C) Clonogenicity and lineage output of single cells cultured in 384-well plates (n = 4 to 5 experiments). P values are shown as follows: **P < .01; ***P < .001 compared with VENUS control (only calculated for M). CA, constitutive active; DN, dominant negative.
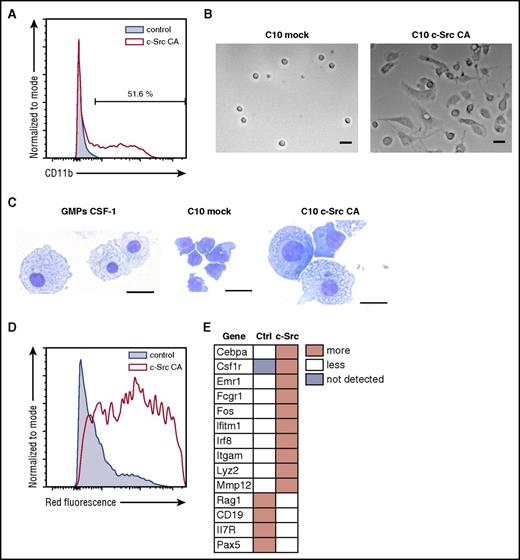
Enforced c-Src activity is sufficient to drive M fate in pre-B cells
We hypothesized that enforced c-Src activity might also be sufficient to induce M fate in other lineages. Consequently, we transduced a B-cell progenitor cell line (C10)16 with constitutive active c-Src. Four to 6 days after transduction, >50% of cells expressed CD11b (Figure 7A) and large adherent cells appeared in culture (Figure 7B). Untransduced or mock-infected cells did not generate CD11bpos or adherent cells (Figure 7A-B). Cytopsin and May-Grünwald Giemsa staining of the adherent cells revealed an increased cytoplasmic-to-nucleus ratio and cytoplasmic vacuoles similar to Ms (Figure 7C). Moreover, adherent cells were phagocytotic (Figure 7D) and cells transduced with constitutive active c-Src upregulated M-associated genes, whereas B-cell–associated genes were downregulated (Figure 7E; supplemental Table 3). This suggests that c-Src signaling activates an M-inducing program that is capable of not only pushing GM-committed progenitors toward Ms, but also of enforcing M fate even in nonmyeloid cells. Of note, forced expression of CSF-1R mutants Y559AB and Y559/807AB in C10 cells also led to upregulation of CD11b, dependent on the presence of CSF-1 (supplemental Figure 11 and data not shown).
Constitutive c-Src signaling is sufficient to trans-differentiate pre-B cells to M-like cells. (A) C10 B-cell progenitors were infected with lentiviruses encoding CA c-Src and analyzed for CD11b expression after 6 days of culture. A representative FACS plot gated on transduced cells is shown (n = 3). (B) Only C10 cells transduced with c-Src CA give rise to adherent cells (right). Note: overgrowing nonadherent cells were washed away before image acquisition. Image was taken 8 days after transduction using a Nikon Eclipse Ti with a ×10 Plan Apochromat objective (NA 0.45) and a Hamamatsu Orca Flash 4.0 camera. (C) May Grünwald-Giemsa staining of adherent C10 cells transduced with c-Src CA reveals M-like morphology. Images were acquired with a Leica DMi1 equipped with a ×40 Plan objective (NA 0.5) and a color camera. Representative images are shown (n = 5 experiments). Scale bars represent 25 μm (B) and 20 μm (C). (D) Only cells transduced with c-Src CA phagocytose fluorescently labeled Escherichia coli. A representative FACS plot is shown (n = 3). (E) Cells transduced with c-Src CA upregulate M genes and downregulate B-cell genes (see supplemental Table 3 for absolute values) (n = 3).
Constitutive c-Src signaling is sufficient to trans-differentiate pre-B cells to M-like cells. (A) C10 B-cell progenitors were infected with lentiviruses encoding CA c-Src and analyzed for CD11b expression after 6 days of culture. A representative FACS plot gated on transduced cells is shown (n = 3). (B) Only C10 cells transduced with c-Src CA give rise to adherent cells (right). Note: overgrowing nonadherent cells were washed away before image acquisition. Image was taken 8 days after transduction using a Nikon Eclipse Ti with a ×10 Plan Apochromat objective (NA 0.45) and a Hamamatsu Orca Flash 4.0 camera. (C) May Grünwald-Giemsa staining of adherent C10 cells transduced with c-Src CA reveals M-like morphology. Images were acquired with a Leica DMi1 equipped with a ×40 Plan objective (NA 0.5) and a color camera. Representative images are shown (n = 5 experiments). Scale bars represent 25 μm (B) and 20 μm (C). (D) Only cells transduced with c-Src CA phagocytose fluorescently labeled Escherichia coli. A representative FACS plot is shown (n = 3). (E) Cells transduced with c-Src CA upregulate M genes and downregulate B-cell genes (see supplemental Table 3 for absolute values) (n = 3).
Discussion
Numerous studies have attempted to elucidate the functional role of individual CSF-1R tyrosine residues and their activated signaling pathways. Using a variety of artificial cellular systems and experimental approaches, these studies have led to contradictory results regarding activated signaling and effects on cellular fates.3 For example, the mutation of Y559 has been described to either increase,5 decrease,11 or not affect proliferation.12 This exemplifies the importance of studying CSF-1R function in the correct cellular context. We therefore established a system to analyze the role of different CSF-1R–activated signaling pathways in uncommitted, primary BM-derived progenitors normally expressing endogenous CSF-1R. The use of unphysiological cell lines and ectopic chimeric receptor expression was thus avoided.
Our approach is based on genetic CSF-1R mutants to deactivate CSF-1–induced signaling pathways instead of less reliable chemical inhibitors. A previous study used a CSF-1R–deficient immortalized M cell line to genetically characterize the function of individual CSF-1R tyrosines.17 However, these mature Ms are not suitable for investigations of CSF-1–mediated lineage choice and the differentiation of uncommitted progenitor cells.
We find that SFK signaling is sufficient to mediate M instruction of primary progenitors by CSF-1. Moreover, c-Src signaling, independently of CSF-1, is sufficient to induce M fate. Previous reports have indicated a role for SFK signaling in CSF-1–mediated M survival and differentiation.11,12,17,39 However, these studies were performed in mature cells, transformed cell lines, or mixed cellular populations such as whole BM, rather than in relevant primary cells. For the first time, we show that this pathway is sufficient for uncommitted primary BM progenitors to be instructed by CSF-1.
Former reports describing CSF-1R functions mainly relied on classical cellular assays to quantify CSF-1–mediated effects on bulk cultures. Besides losing information on cell-to-cell variability, these assays can also obscure contributions of individual CSF-1–affected cell fates to an observed effect. By using live cell imaging, we were able to simultaneously analyze individual cell fate parameters influenced by CSF-1 at the single-cell level, allowing to reveal for example, whether decreased cell numbers are due to more death, reduced proliferation, or a combination of both. Moreover, clonal colony assays showed that some chemical inhibitors have an effect on survival and proliferation, but not on lineage choice. The latter point could otherwise easily be confused with a cell differentiation defect.
In primary myeloid progenitors, single-cell readouts revealed mutations of individual CSF-1R tyrosines to have varying effects on CSF-1–mediated survival, proliferation, and differentiation. As previously described in Ms,17 the mutation of all 8 tyrosine residues results in a receptor that is unable to transmit survival and proliferation signals. Data from individually tracked colonies showed that none of the single CSF-1R tyrosine mutations totally abrogated cell survival, corresponding to previous findings.17 We find that only the mutation of Y721 or Y807 significantly compromised cell survival. The individual mutation of Y559 or of the remaining tyrosines did not have an effect on survival/proliferation. This is surprising, because there are reports indicating a major role of Y559 for proliferation.11,17 However, these studies were done in mature M cells, which divide much less frequently. Possibly, different combinations of pathways induce proliferation in these cells. A hyperproliferative response to CSF-1 when mutating Y559, reported for a myeloid cell line,5 was also not observed in our model. A previous study using the myeloid progenitor cell line FDCP1 showed that the mutation of Y807 abrogated CSF-1–induced differentiation, and at the same time, enhanced proliferation.10 Our data suggests that Y807 is only indirectly involved in differentiation by contributing to cell survival (see section to follow), and we did not detect hyperproliferation upon its mutation.
Adding back Y559 to a fully mutated CSF-1R background was sufficient to significantly restore CSF-1–mediated survival/proliferation and differentiation. Y559, in combination with Y807, fully restored Ywt levels of survival/proliferation and further enhanced differentiation. In mature Ms, adding back either Y559 or Y807 to the CSF-1R is sufficient to restore CSF-1–mediated proliferation but not differentiation (as determined via CD11b expression).18 In contrast to Yu et al,18 Y807AB only partially rescued the survival/proliferation in primary progenitors and we did not observe cytokine-independent proliferation of cells transduced with Y807AB (data not shown). However, we confirm that adding back Y721 or Y807 individually was not sufficient to restore CSF-1 differentiation. Collectively, by using single-cell–based assays and primary cells, our approach confirms some results obtained in other cellular systems. However, some tyrosine functions differ from what has been described in nonprimary cells.
Y559 is the first tyrosine to be phosphorylated in the CSF-1R,17,18,40 and has been proposed to be critical for CSF-1R kinase activity and receptor phosphorylation.5 At the same time, it activates an SFK/c-Cbl ubiquitination pathway that leads to full receptor activation and to ligand-induced receptor internalization and degradation.5,11,17,40 Several reports have described phosphorylated Y559 as the only CSF-1R SFK binding site.34-36 No other molecules have been reported to bind Y559. Y807 is located in the CSF-1R activation loop and has been implicated in the activation of CSF-1R kinase activity along with Y559.18 To date, no protein has been described to directly interact with the phospho-Y807 site. Y807 increases the survival and differentiation-transmitting capacity of the CSF-1R alongside Y559, reminiscent of what has been described previously.11,18 Y807 could either improve survival through its role in receptor activation and thereby make cells permissive to differentiate through an Y559-mediated mechanism, or contribute to differentiation signaling itself. Because Y807 on its own is not sufficient to rescue differentiation, and an SFK inhibitor was sufficient to significantly decrease Y559-/807-mediated rescue of survival and differentiation, we believe that the main role of Y807 in the context of the Y559/807AB mutant is to improve full functional activation of the receptor, and through that survival, rather than actively contributing to differentiation-inducing signaling.
Consequently, our data indicate that CSF-1 survival and differentiation signaling can be transmitted through SFKs, with SFK signaling being sufficient for CSF-1–mediated lineage instruction. Thus, this approach described by us is suitable for the elucidation of the relevant transcriptional programs involved in lineage instruction. However, the observation that the Y559 mutation does not significantly reduce M output also suggests that: (1) SFKs are not essential for this process, and (2) that the signaling pathways involved can also be activated through other compensating tyrosine residues. Upon CSF-1 induction, SFKs have been reported to activate several major downstream signaling pathways, including MAPKs (ERK1/2 and p38), PI3K/Akt, phospholipase γ, STATs, and nuclear factor-κB.11,12,18,39,41,42 We here show that in primary progenitors, Y559 activates MEK/ERK signaling via SFKs, and that both SFK and MEK inhibitors decrease the capacity of the Y559/807AB mutant to rescue M differentiation. However, we cannot exclude that in this context SFKs activate other pathways as well. Interestingly, a previous study has suggested an SFK to MEK/ERK signaling axis to be involved in the CSF-1–mediated proliferation and differentiation of BM cultures.39
Due to the lack of phospho-specific antibodies against individual SFK members, activation of relevant SFKs cannot be directly shown. However, forced activity of c-Src but not of other SFK members such as Fyn, Lyn, and Hck, resulted in a marked skewing of G to M lineage choice in GMPs. Although it cannot be excluded that expression of constitutively active c-Src may have a different effect than activated endogenous c-Src, this strongly suggests that c-Src is the relevant SFK involved in M lineage choice. MEK inhibitors did not block the effect of constitutive active c-Src in GMPs, suggesting that compensating signaling pathways are activated via c-Src. Forced c-Src activity also induced differentiation of B-cell progenitors into M-like cells, which is reminiscent of spontaneous M differentiation of oncogene-transformed pre–B-cell lines.43-45 Insertion of the CSF-1R into a pre–B-cell line is sufficient to induce M differentiation in the presence of CSF-1.46 This suggests that c-Src signaling mimics CSF-1–induced M differentiation and makes c-Src an interesting candidate to identify M lineage-instructive transcription factors. Noteworthy, c-Src- and other SFK-deficient mice do not show an M phenotype,47 agreeing with our observation that loss of CSF-1–mediated SFK signaling by mutation of Y559 is not sufficient to abrogate M differentiation.
Constitutive active Akt also led to an increased M output of GMPs. In mice, constitutive active Akt has been shown to induce myeloid expansion in the spleen in vivo and Akt was suggested to play a role in human myeloid lineage decisions.48,49 This suggests that PI3K/Akt-signaling might be a complementary pathway involved in CSF-1–induced differentiation, which may also be activated by SFKs in GMPs.
Collectively, our results demonstrate that CSF-1–instructed differentiation of primary GMPs to Ms can occur through SFK-activated signaling, involving downstream activation of the MEK/ERK pathway. Moreover, c-Src activity alone is sufficient to induce M differentiation in myeloid and even nonmyeloid cells. At the same time, Y559/SFK signaling is not essential for M lineage instruction by CSF-1, and can be compensated for by other CSF-1R tyrosine residues. This demonstrates that several CSF-1R–activated signaling axes are integrated into a complex system controlling lineage choice, as has also been shown for CSF-1 control of M proliferation.11,18
The online version of this article contains a data supplement.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Acknowledgments
The authors thank Christian Raithel, Angelika Ziegler, Sandra Ammersdoerfer, and Bianca Vogel for technical support, and Thomas Graf for providing the C10 cell line. Constitutive active forms of c-Src, Lyn, and Fyn were generously provided by Owen N. Witte; and constitutive active Hck was a gift from Glenn Scholz.
This work was supported by grants from the Swiss National Science Foundation; SystemsX.ch (T.S., S.S., and O.H); and the National Institutes of Health, National Cancer Institute (CA26504 and PO1 CA100324) (E.R.S.).
Authorship
Contribution: M.E. planned and performed experiments, collected and analyzed data, and wrote the manuscript; A.S. provided lentiviral vectors; E.R.S. provided the Csf1r+/− mice, the Csf1r constructs, and reviewed the manuscript; P.S.H. provided technical support; and T.S. designed and supervised the study, developed and maintained long-term bioimaging with D.L. and K.D.K., single-cell tracking with O.H., analysis software with S.S., and wrote the manuscript with M.E.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Timm Schroeder, Department of Biosystems Science and Engineering, Eidgenössische Technische Hochschule Zurich, Mattenstrasse 26, 4058 Basel, Switzerland; e-mail: timm.schroeder@bsse.ethz.ch.

![Figure 3. Derivation and lineage potential of BM-derived Csf1r−/− progenitors. (A-B) W41 (CD45.1) mice were IV transplanted with 2 × 107 E14.5 fetal liver cells of WT or Csf1r−/− (CD45.2) genotype. Donor contribution was checked at various timepoints in recipients’ as follows: (B, left) peripheral blood (n = 3 to 6 per timepoint per genotype) or (B, right) BM (GMPs) (n = 3 per genotype) via flow cytometry. (C) Freshly isolated GMPs from C57Bl/6 (WT), W41 transplanted with WT fetal liver cells (WT [transpl.]), or W41 transplanted with Csf1r−/− fetal liver cells (Csf1r−/−) were cultured in methylcellulose containing either SCF, IL-3, IL-6, and EPO (left) or CSF-1 (right). After 5 to 7 days, colonies were enumerated and identified according to morphology (clonogenicity SCF IL-3 IL-6 EPO: WT = 45% ± 6.4% SD, WT [transpl.] = 47% ± 7.2% SD, Csf1r−/− = 43% ± 4.4% SD; and clonogenicity CSF-1: WT = 46% ± 5.1% SD, WT [transpl.] = 47% ± 5% SD, Csf1r−/− = 0, n = 3 per genotype per condition). P values are shown as follows: ***P < .001 compared with WT. Transpl, transplanted.](https://ash.silverchair-cdn.com/ash/content_public/journal/blood/129/12/10.1182_blood-2016-05-714329/4/m_blood714329f3.jpeg?Expires=1769164436&Signature=qVkrew-6Hk8n67uGcFMvftapiHRZMQ0bFz3BhzQScB7JZoTDCMHX4q~bg9VdPjqLo8JLHySEwT9nFDW5CtSct8Z~FmWHVkC1vpiwR4tozLliNSpF6P6v1y9kYdY8zTaxg8CqUzlLz2K-tsF7CWT1cGK0CekRWIS0GCWEQHN1E2vxkOddbK9~ouJjzWP2ZAp2gUrMzlH31NKVGvV0PEvyDnW8GhNrK4etTRyUqt~8CBo1Tq1mtQJ5Lhf77E2t0tG7W4GbomMqShRvZxzOa~D0iMIqJ5N7SABDqMBeTsI-cu0r-ELt-9MEmzfnDA4pSED-CmfM3kcFVNnXlCR~UnJRKA__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)