TO THE EDITOR:
Myelodysplastic syndromes (MDSs) are hematopoietic stem cell disorders characterized by ineffective hematopoiesis and high rates of leukemic transformation. The only curative treatment is stem cell transplantation; therefore, new therapies are needed.1 Recent studies suggest that enhanced Toll-like receptor (TLR) signaling may contribute to the pathogenesis of MDS.2-10 Specifically, the expression of TLR2 is markedly elevated in the CD34+ cells of MDS patients compared with healthy controls,3,11 and TLR2 stimulation of CD34+ cells in vitro impairs erythroid differentiation.3 Based on these data, a clinical trial using a TLR2 antagonist as a second-line treatment of patients with lower risk MDS is ongoing (OPN-305, Opsona Therapeutics). However, despite growing interest in TLR2 inhibition as a therapeutic strategy, the role of TLR2 in MDS is not clear. Moreover, increased expression of TLR2 is actually associated with lower risk disease and improved overall survival,3,11 raising the question of whether TLR2 may have a protective role in MDS.
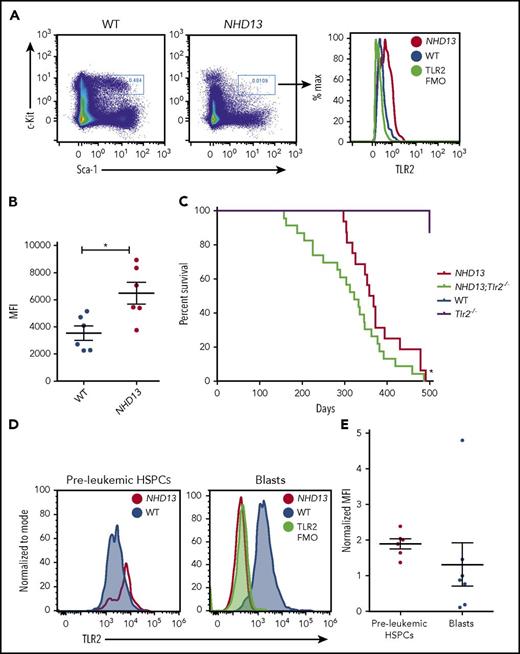
To elucidate the contribution of TLR2 signaling to MDS pathogenesis, we used a well-characterized mouse model of MDS (expressing the NUP98-HOXD13 fusion from the hematopoietic Vav-1 promoter, also known as NHD13 mice). These mice exhibit many features of human MDS, including bone marrow dysplasia, cytopenias, and increased apoptosis of hematopoietic stem and progenitor cells (HSPCs) and die of acute leukemia or severe cytopenias.12-15 Notably, similar to the CD34+ cells of patients with MDS, the HSPCs (lineage− c-Kit+ Sca-1+ [KSL] cells) of NHD13 mice display elevated TLR2 expression and signaling compared with wild-type (WT) controls (Figure 1A-B; supplemental Figure 1A-C, available on the Blood Web site). In this study, the NHD13 mice were crossed to mice lacking TLR2, and the effects of TLR2 loss on the development of cytopenias, leukemogenesis, and survival were assessed. In addition, the contribution of TLR2 to premalignant HSPC cycling, apoptosis, and DNA damage accumulation were determined (see supplemental Materials for a description of the methods used).
Loss of TLR2 accelerates leukemogenesis in the NHD13 mice. (A) Representative flow plots of TLR2 surface expression on the bone marrow lineage− c-Kit+ Sca-1+ (KSL) cells of WT and NHD13 mice. A TLR2 FMO control was included as a negative control. These data are quantified in panel B, which shows the MFI values for each of the samples analyzed (n = 6 mice/group, age 6-8 weeks). (C) Kaplan-Meier survival curve of NHD13;Tlr2+/+ (n = 16), NHD13;Tlr2−/− (n = 23), Tlr2−/− (n = 16), and WT mice (n = 7). *P = .03 by the Gehan-Breslow-Wilcoxon test and .07 by log-rank (Mantel-Cox) test comparing NHD13;Tlr2−/− with NHD13;Tlr2+/+. (D) TLR2 surface expression was assessed by flow cytometry on bone marrow KSL cells of 6- to 8-week-old (preleukemic) WT vs NHD13 mice (left) and the bone marrow blast cells of leukemic NHD13 mice compared with the c-Kit+ cells of healthy WT controls (right). A TLR2 FMO was included as a negative control. Data for each mouse analyzed are plotted in panel E, with each data point representing the MFI of the NHD13 cells (KSL cells or blasts) normalized to the MFI of WT cells (KSL cells or c-Kit+ cells for preleukemic HSPCs and blasts, respectively) run at the same time. n = 6-7 mice/group. Error bars represent mean ± standard error of the mean. FMO, fluorescence minus one; MFI, median fluorescence intensity.
Loss of TLR2 accelerates leukemogenesis in the NHD13 mice. (A) Representative flow plots of TLR2 surface expression on the bone marrow lineage− c-Kit+ Sca-1+ (KSL) cells of WT and NHD13 mice. A TLR2 FMO control was included as a negative control. These data are quantified in panel B, which shows the MFI values for each of the samples analyzed (n = 6 mice/group, age 6-8 weeks). (C) Kaplan-Meier survival curve of NHD13;Tlr2+/+ (n = 16), NHD13;Tlr2−/− (n = 23), Tlr2−/− (n = 16), and WT mice (n = 7). *P = .03 by the Gehan-Breslow-Wilcoxon test and .07 by log-rank (Mantel-Cox) test comparing NHD13;Tlr2−/− with NHD13;Tlr2+/+. (D) TLR2 surface expression was assessed by flow cytometry on bone marrow KSL cells of 6- to 8-week-old (preleukemic) WT vs NHD13 mice (left) and the bone marrow blast cells of leukemic NHD13 mice compared with the c-Kit+ cells of healthy WT controls (right). A TLR2 FMO was included as a negative control. Data for each mouse analyzed are plotted in panel E, with each data point representing the MFI of the NHD13 cells (KSL cells or blasts) normalized to the MFI of WT cells (KSL cells or c-Kit+ cells for preleukemic HSPCs and blasts, respectively) run at the same time. n = 6-7 mice/group. Error bars represent mean ± standard error of the mean. FMO, fluorescence minus one; MFI, median fluorescence intensity.
To begin, the NHD13 mice were bred to Tlr2−/− mice (NHD13; Tlr2+/− × Tlr2+/−) to generate 4 groups: NHD13;Tlr2−/−, NHD13;Tlr2+/+, Tlr2−/−, and WT (NHD13-;Tlr2+/+). Surprisingly, loss of TLR2 was associated with worse survival (P = .03 comparing NHD13; Tlr2−/− to NHD13;Tlr2+/+; Figure 1C; supplemental Figure 2A; supplemental Table 1), and did not confer an improvement in peripheral cytopenias (supplemental Figure 2B-D). In addition, we more recently crossed the NHD13 mice to mice lacking MyD88, an adapter required for most TLR signaling, including TLR2, and found an even more significant acceleration of death (P < .0001 comparing NHD13; MyD88−/− to NHD13 and P < .01 comparing NHD13; MyD88−/− to NHD13; Tlr2−/−; supplemental Figure 2A,E). As shown in supplemental Figure 2A and supplemental Table 1, the most common causes of death were leukemia and myeloproliferative disorder–like leukemia. Of note, although we detected high levels of surface TLR2 on premalignant NHD13;Tlr2+/+ HSPCs, this expression was often markedly reduced on the blasts of leukemic mice (Figure 1D-E), suggesting that TLR2 is frequently downregulated at some point during disease progression. Thus, TLR signaling may play a protective role against leukemic transformation in the NHD13 mice.
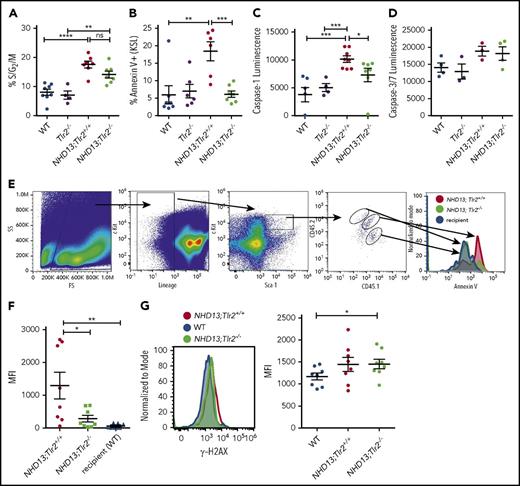
To investigate the potential mechanism by which loss of TLR signaling accelerates death in NHD13 mice, the KSL cells of preleukemic young adult NHD13;Tlr2+/+ and NHD13;Tlr2−/− mice were evaluated for cell-cycle status, cell death, and DNA damage. Previous studies have shown that the KSL population in NHD13 mice contains the transplantable disease-initiating cells,14,16 and displays both enhanced cycling and apoptosis compared with WT controls.13,17 Although loss of TLR2 in the NHD13 mice did not confer a significant difference in KSL cycling (Figure 2A; supplemental Figure 3), it did lead to a loss of Annexin V+ KSL cells in the bone marrow (Figure 2B; supplemental Figure 4) and an accumulation of c-Kit+ lineage− cells in the liver (supplemental Figure 5). An assessment of Caspase-1 and Caspase-3/7 activities showed a significant reduction in activated Caspase-1 in the KSL cells of NHD13;Tlr2−/− mice compared with NHD13;Tlr2+/+ mice (Figure 2C-D), suggesting that TLR2 specifically promotes inflammasome-dependent cell death in the NHD13 mice. Further supporting a role for TLR2 signaling in the death of premalignant NHD13 HSPCs, RNA sequencing of sorted KSL cells demonstrated an enrichment of apoptosis-related gene pathways in the NHD13 compared with the NHD13;Tlr2−/− cells (supplemental Table 2). To determine whether the TLR2-associated cell death is cell autonomous or cell-nonautonomous, chimeric animals were generated by transplanting a mixture of NHD13;Tlr2+/+ and NHD13;Tlr2−/− bone marrow cells into lethally irradiated WT recipients (supplemental Figure 6). Analysis of these chimeras revealed that the enhanced apoptosis of NHD13 HSPCs requires cell-autonomous TLR2 signaling (ie, only cells expressing TLR2 have elevated Annexin V staining; Figure 2E-F). Finally, the accumulated HSPCs in the NHD13;Tlr2−/− mice show elevated levels of γ-H2AX staining compared with WT controls (Figure 2G), indicating that TLR2 loss promotes the survival of damaged premalignant HSPCs.
Cell-autonomous TLR2 signaling promotes apoptosis of preleukemic HSPCs. The bone marrow KSL cells of 6- to 8-week-old WT, Tlr2−/−, NHD13;Tlr2+/+, and NHD13;Tlr2−/− mice were analyzed by flow cytometry. (A) Percent of KSL cells in the S/G2/M phase of the cell cycle as determined by Ki-67 and 4′,6-diamidino-2-phenylindole staining (n = 4-8 mice/group). (B) Bone marrow KSL cells from the same groups of mice were analyzed for Annexin V staining by flow cytometry. Shown are the percentage of Annexin V+ KSL cells (n = 6-7 mice/group). (C) Caspase-1 and (D) Caspase-3/7 activities were assessed from sorted KSL cells as described in the supplemental Methods (n = 3-8 mice/group). (E) Chimeras (containing a mixture of NHD13;Tlr2+/+ and NHD13;Tlr2−/− bone marrow cells) were generated as described in supplemental Figure 6, and Annexin V flow cytometry staining was performed on bone marrow. Shown is a representative flow analysis of the KSL cells of 1 of the chimeric animals. These data are quantified in panel F, which shows the MFI of Annexin V staining on the KSL cells from each of the genotypes in 8 total chimeras analyzed. (G). γ-H2AX staining was performed on the bone marrow KSL cells. Left, a representative flow plot comparing γ-H2AX levels between young adult (6-8 weeks old) WT (NHD13-;Tlr2+/+), NHD13, and NHD13+;Tlr2−/− animals. Right, each data point represents the MFI for KSL cells from individual mice from each of the WT, NHD13, and NHD13+;Tlr2−/− cohorts. n = 8 mice/group performed over 4 independent experiments. *P < .05; **P < .01, ***P < .001, ****P < .0001 by an unpaired Student t test or 1-way analysis of variance. Error bars represent mean ± standard error of the mean.
Cell-autonomous TLR2 signaling promotes apoptosis of preleukemic HSPCs. The bone marrow KSL cells of 6- to 8-week-old WT, Tlr2−/−, NHD13;Tlr2+/+, and NHD13;Tlr2−/− mice were analyzed by flow cytometry. (A) Percent of KSL cells in the S/G2/M phase of the cell cycle as determined by Ki-67 and 4′,6-diamidino-2-phenylindole staining (n = 4-8 mice/group). (B) Bone marrow KSL cells from the same groups of mice were analyzed for Annexin V staining by flow cytometry. Shown are the percentage of Annexin V+ KSL cells (n = 6-7 mice/group). (C) Caspase-1 and (D) Caspase-3/7 activities were assessed from sorted KSL cells as described in the supplemental Methods (n = 3-8 mice/group). (E) Chimeras (containing a mixture of NHD13;Tlr2+/+ and NHD13;Tlr2−/− bone marrow cells) were generated as described in supplemental Figure 6, and Annexin V flow cytometry staining was performed on bone marrow. Shown is a representative flow analysis of the KSL cells of 1 of the chimeric animals. These data are quantified in panel F, which shows the MFI of Annexin V staining on the KSL cells from each of the genotypes in 8 total chimeras analyzed. (G). γ-H2AX staining was performed on the bone marrow KSL cells. Left, a representative flow plot comparing γ-H2AX levels between young adult (6-8 weeks old) WT (NHD13-;Tlr2+/+), NHD13, and NHD13+;Tlr2−/− animals. Right, each data point represents the MFI for KSL cells from individual mice from each of the WT, NHD13, and NHD13+;Tlr2−/− cohorts. n = 8 mice/group performed over 4 independent experiments. *P < .05; **P < .01, ***P < .001, ****P < .0001 by an unpaired Student t test or 1-way analysis of variance. Error bars represent mean ± standard error of the mean.
Together, these data suggest that cell-autonomous TLR2 signaling promotes the death of premalignant HSPCs in NHD13 mice, and that complete loss of TLR2 leads to worse survival (with the most common cause of death being leukemia or myeloproliferative disorder–like leukemia). These findings are consistent with patient data demonstrating that higher TLR2 expression is associated with increased HSPC apoptosis, lower risk disease, and longer survival.3,11 Elevated HSPC apoptosis is characteristic of low-risk MDS, and is lost as the disease progresses to leukemia and cells overcome pro-death signals.18 The lack of surface TLR2 on transformed cells in NHD13 mice further supports the idea that TLR2 signaling contributes to this elevated death of premalignant MDS cells. Thus, although recent studies have demonstrated an association between high TLR2 expression and signaling and MDS, prompting enthusiasm for TLR2 as a therapeutic target in this disease, the data presented here caution that complete inhibition of TLR2 signaling may expedite leukemogenesis. Although these data are based on a single MDS model, it nonetheless suggests that this pathway may in fact serve a protective role against transformation, stimulating the death of damaged premalignant HSPC. Studies are under way to determine whether TLR2 signaling contributes more broadly to premalignant HSPC death and protection against transformation in other genetic backgrounds. In addition, future studies are needed to define the specific downstream effectors of TLR2 signaling contributing to HSPC death in MDS, as well as to identify factors that regulate the expression of TLR2 in premalignant and transformed HSPCs and determine the timing of TLR2 downregulation during disease progression. Finally, while this data supports a role for cell-autonomous TLR2 signaling in regulating premalignant HSPC death, it does not rule out a potential contribution of cell-nonautonomous TLR2 signaling to the regulation of premalignant HSPCs. Studies using conditional deletion of TLR2 from different hematopoietic and stromal bone marrow populations are under way to address the contribution of TLR2 signaling from the microenvironment to the pathogenesis of MDS.
The online version of this article contains a data supplement.
Acknowledgments
The authors thank the Alvin J. Siteman Cancer Center at Washington University School of Medicine and Barnes-Jewish Hospital in St. Louis, MO, for the use of the Siteman Flow Cytometry Core. The authors also thank Jackie Tucker-Davis for animal care and the Genome Technology Access Center at Washington University School of Medicine for sequencing services.
This work was supported by grants from the Children’s Discovery Institute of Washington University and St. Louis Children’s Hospital (L.G.S.), Washington University Specialized Program of Research Excellence in Leukemia’s Career Enhancement Program (L.G.S.), American Cancer Society Institutional Research Grant (L.G.S.), Hyundai Hope on Wheels (S.T.B.), and the National Institutes of Health, National Heart, Lung, and Blood Institute (1R01 HL134896-01) (L.G.S.).
Authorship
Contribution: D.A.M., S.T.B., Z.J.G., J.L.K., M.P.R., and L.G.S. performed experiments and analyzed data; W.Y. analyzed data; D.A.M., S.T.B., and M.J.W. designed and oversaw experiments; E.J.D. reviewed all pathology; P.D.A. provided mice; L.G.S. directed the study; and D.A.M., S.T.B., and L.G.S. wrote and edited the manuscript.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Laura G. Schuettpelz, Department of Pediatrics, Washington University School of Medicine, 660 S Euclid Ave, St. Louis, MO 63110; e-mail: schuettpelz_l@wustl.edu.
References
Author notes
D.A.M. and S.T.B. contributed equally to this study.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal