Abstract
Introduction: Pts with AML often present with hyperleukocytosis, defined with a white blood cell count (WBC) of >50 or >100 × 109/L. Hyperleukocytosis is associated with higher rates of complications and death especially when associated with clinical leukostasis. There are no clear guidelines outlining the best cytoreductive strategy and the use of leukapheresis is based on institutional practice. Limited data is available regarding characteristics of AML pts with hyperleukocytosis, treatment patterns, short and long-term clinical outcomes.
Methods: Data were collected retrospectively from 12 centers in the United States and Europe. Eligible pts had newly diagnosed AML, WBC > 50 × 109/L, and had received intensive chemotherapy (IC). Pts with hyperleukocytosis who did not receive IC are described in a separate abstract. Kaplan-Meier methods estimated overall survival (OS) from time of presentation till death or end of follow-up. Clinical evidence of leukostasis was defined as new onset hypoxia, chest pain, headache, focal neurological symptoms, priapism, intestinal ischemia and acute renal failure attributed to hyperleukocytosis by the primary provider of the pt.
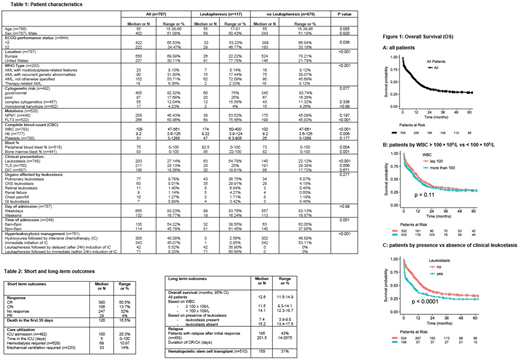
Results: Among 1050 pts with AML and hyperleukocytosis whose data were collected, 787 were reported to have received intensive chemotherapy and were included in this analysis. The median age was 55 years (range [R], 15-86), and 51% were male (Table 1). Median WBC at presentation was 109 × 109/L (R, 47-561) and 57% had WBC > 100 × 109/L. Median hemoglobin was 9.2 g/dL (R, 3.6-126) and median platelet count was 31 x 109/L (R, 3-1268). A good, intermediate and poor risk karyotype were present in 31%, 51% and 18% of pts, respectively. Clinical leukostasis, tumor lysis syndrome (TLS) and disseminated intravascular coagulation (DIC) were present in 27%, 28% and 18% of pts at time of presentation, respectively (Table 1). Organs affected by leukostasis were the lung, CNS, retina, heart, kidney and GI tract in 44%, 36%, 6%, 6%, 5% and 4%, respectively. Leukapheresis was administered in 117 pts (15%) (Figure 1). Four centers did not use leukapheresis. 31% of pts with clinical leukostasis underwent leukapheresis and 10% of pts without clinical leukostasis received leukapheresis (p<0.001). Pts, who underwent leukapheresis, had statistically significant higher WBC and higher % of FLT3 mutations compared to pts, who did not undergo leukapheresis (Table 1). In pts who did not receive leukapheresis, hyperleukocytosis was managed either by immediate initiation of intensive chemotherapy (n=342, 53%) or by the administration of hydroxyurea followed by intensive chemotherapy (n=302, 47%). Pts managed with leukapheresis, received either immediate (within 24h) induction of intense chemotherapy (n=71, 61%) or delayed (after 24h) induction of intensive chemotherapy (n=42, 36%) after completion of leukapheresis. The 30-day mortality was 16.6% (95%CI, 13.9-19.3%); 20%, 11% and 14% of pts were admitted to the ICU, underwent hemodialysis, or required mechanical ventilation, respectively (Table 2). After initiation of chemotherapy, 51% (95%CI, 46.9%-54.1%) had a complete remission (CR), 14% (95%CI, 11.4%- 16.4%) had a complete remission with incomplete count recovery (CRi) and 4% (95%CI, 2.5%-5.4%) achieved a partial remission (PR) whereas 32% (95%CI, 28.7%-35.4%) had no response to therapy (Table 2). Response to chemotherapy lasted a median of 202 days (R,14-3575) and 43% of pts experienced a relapse of their disease; 31% of pts underwent a hematopoietic stem cell transplant (HSCT). Median OS for all pts was 12.6 months (95%CI, 11.5-14.9) (Figure 1A). Median OS was 14.1 months (95%CI, 12.3-18.7) for pts with a WBC <100 × 109/L versus 11.5 months (95%CI, 9.3-14.1) for pts with a WBC >100 × 109/L (p=0.11) (Figure 1B). Median OS was 15.2 months (95%CI, 13.4-17.5) for pts without clinical leukostasis, significantly longer than the median OS of 7.4 months (95%CI, 3.9-9.8) for pts with symptoms of leukostasis (p<0.0001) (Figure 1C).
Conclusions: To our knowledge, this is largest reported cohort of pts with AML and hyperleukocytosis treated with intensive chemotherapy. Clinical leukostasis was present in about a quarter of pts and was associated with worse OS. Most pts were managed with chemotherapy alone, and leukapheresis was only used in a small subgroup of pts (15%). The impact of leukapheresis and other variables on outcomes are presented in a separate abstract.
Montesinos:Novartis: Research Funding, Speakers Bureau; Daiichi Sankyo: Consultancy, Speakers Bureau. Bhatt:Pfizer: Consultancy; CSL Behring: Consultancy; Incyte: Research Funding. Sekeres:Celgene: Membership on an entity's Board of Directors or advisory committees; Opsona: Membership on an entity's Board of Directors or advisory committees; Celgene: Membership on an entity's Board of Directors or advisory committees; Opsona: Membership on an entity's Board of Directors or advisory committees. Fathi:Boston Biomedical: Consultancy, Honoraria; Jazz: Honoraria; Celgene: Consultancy, Honoraria, Research Funding; Takeda: Consultancy, Honoraria; Astellas: Honoraria; Agios: Honoraria, Research Funding; Seattle Genetics: Consultancy, Honoraria. Khan:Teva: Speakers Bureau. Roboz:Janssen Pharmaceuticals: Consultancy; Aphivena Therapeutics: Consultancy; Roche/Genentech: Consultancy; Argenx: Consultancy; Aphivena Therapeutics: Consultancy; Jazz Pharmaceuticals: Consultancy; Sandoz: Consultancy; Pfizer: Consultancy; Roche/Genentech: Consultancy; Cellectis: Research Funding; Orsenix: Consultancy; Bayer: Consultancy; Bayer: Consultancy; Novartis: Consultancy; Pfizer: Consultancy; Cellectis: Research Funding; Sandoz: Consultancy; Otsuka: Consultancy; Astex Pharmaceuticals: Consultancy; Celgene Corporation: Consultancy; AbbVie: Consultancy; AbbVie: Consultancy; Astex Pharmaceuticals: Consultancy; Argenx: Consultancy; Janssen Pharmaceuticals: Consultancy; Eisai: Consultancy; Otsuka: Consultancy; Daiichi Sankyo: Consultancy; Orsenix: Consultancy; Celltrion: Consultancy; Novartis: Consultancy; Jazz Pharmaceuticals: Consultancy; Celgene Corporation: Consultancy; Eisai: Consultancy; Daiichi Sankyo: Consultancy; Celltrion: Consultancy. Cluzeau:AbbVie: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Celgene: Consultancy, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Sanofi: Speakers Bureau; Pfizer: Speakers Bureau; Amgen: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Menarini: Consultancy; Jazz Pharma: Consultancy, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau. Germing:Novartis: Honoraria, Research Funding; Celgene: Honoraria, Research Funding; Janssen: Honoraria. Mukherjee:Pfizer: Honoraria; Projects in Knowledge: Honoraria; BioPharm Communications: Consultancy; LEK Consulting: Consultancy, Honoraria; Takeda: Membership on an entity's Board of Directors or advisory committees; Bristol Myers Squib: Honoraria, Speakers Bureau; Novartis: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Takeda Pharmaceuticals: Membership on an entity's Board of Directors or advisory committees; Aplastic Anemia & MDS International Foundation in Joint Partnership with Cleveland Clinic Taussig Cancer Institute: Honoraria. Brunner:Celgene: Consultancy, Research Funding; Takeda: Research Funding; Novartis: Research Funding. Ritchie:Incyte: Consultancy, Speakers Bureau; NS Pharma: Research Funding; Celgene: Consultancy, Other: Travel, Accommodations, Expenses, Speakers Bureau; Pfizer: Consultancy, Research Funding; Novartis: Consultancy, Other: Travel, Accommodations, Expenses, Research Funding, Speakers Bureau; ARIAD Pharmaceuticals: Speakers Bureau; Astellas Pharma: Research Funding; Bristol-Myers Squibb: Research Funding. Podoltsev:Pfizer: Research Funding; Sunesis Pharmaceuticals: Research Funding; LAM Therapeutics: Research Funding; Genentech: Research Funding; Boehringer Ingelheim: Research Funding; Pfizer: Membership on an entity's Board of Directors or advisory committees; Astex Pharmaceuticals: Research Funding; Celgene: Research Funding; Astellas Pharma: Research Funding; Celator: Research Funding; Daiichi Snakyo: Research Funding. Gore:Celgene: Consultancy, Research Funding. Zeidan:Otsuka: Consultancy, Honoraria; AbbVie: Consultancy, Honoraria; Novartis: Consultancy, Honoraria; Celgene: Consultancy, Honoraria; Agios: Consultancy, Honoraria; Takeda: Honoraria, Speakers Bureau; Pfizer: Consultancy, Honoraria.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal