Key Points
MAA868 is a novel, fully human, potent factor XI antibody that uniquely binds with high affinity both active enzyme and zymogen
MAA868 exhibits strong and long-lasting anticoagulant activity in humans with a promising safety profile
Abstract
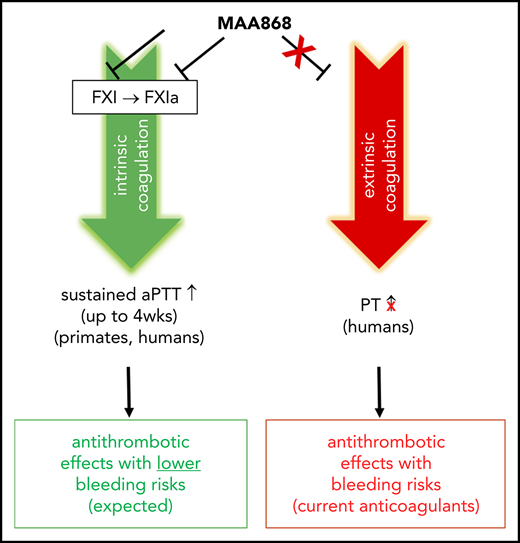
A large unmet medical need exists for safer antithrombotic drugs because all currently approved anticoagulant agents interfere with hemostasis, leading to an increased risk of bleeding. Genetic and pharmacologic evidence in humans and animals suggests that reducing factor XI (FXI) levels has the potential to effectively prevent and treat thrombosis with a minimal risk of bleeding. We generated a fully human antibody (MAA868) that binds the catalytic domain of both FXI (zymogen) and activated FXI. Our structural studies show that MAA868 traps FXI and activated FXI in an inactive, zymogen-like conformation, explaining its equally high binding affinity for both forms of the enzyme. This binding mode allows the enzyme to be neutralized before entering the coagulation process, revealing a particularly attractive anticoagulant profile of the antibody. MAA868 exhibited favorable anticoagulant activity in mice with a dose-dependent protection from carotid occlusion in a ferric chloride–induced thrombosis model. MAA868 also caused robust and sustained anticoagulant activity in cynomolgus monkeys as assessed by activated partial thromboplastin time without any evidence of bleeding. Based on these preclinical findings, we conducted a first-in-human study in healthy subjects and showed that single subcutaneous doses of MAA868 were safe and well tolerated. MAA868 resulted in dose- and time-dependent robust and sustained prolongation of activated partial thromboplastin time and FXI suppression for up to 4 weeks or longer, supporting further clinical investigation as a potential once-monthly subcutaneous anticoagulant therapy.
Introduction
Thromboembolic diseases, including deep vein thrombosis, pulmonary embolism, acute coronary syndrome, and ischemic stroke, are a leading cause of death and disability accounting for 1 in 4 deaths worldwide.1 Currently available anticoagulant agents, including warfarin and the newer direct anticoagulant drugs, that are prescribed to treat and prevent thromboembolic diseases interfere with components of the common coagulation pathway, which maintains blood hemostasis; this interference increases the risk of potentially lethal bleeding events.2-4 To reduce the risk of severe bleeding, current anticoagulant agents must be dosed in a therapeutic window. With rather short half-lives requiring at least daily dosing, missed doses may lead to insufficient anticoagulant activity.5,6 Furthermore, appropriate dosing is often challenging in an older population because plasma exposures can be affected by age, renal/hepatic function, or drug–drug interactions.7 Consequently, better treatment options are needed. Inhibition of factor XI (FXI), a crucial component of the intrinsic (contact) coagulation pathway, has been proposed as a new and promising anticoagulation therapy with minimal bleeding risks.3,8,9
FXI is the zymogen of a S1 serine-type plasma protease, and activated FXI (FXIa) contributes to fibrin formation and stability by activating factor IX.10,11 Factor XIIa (FXIIa) and thrombin can activate FXI, resulting in sustained thrombin generation and inhibition of fibrinolysis.9 Overall, FXI seems to play a key role in thrombosis and only a minor role in hemostasis, particularly after vessel injury in a high tissue factor environment.11
Indeed, human genetics as well as epidemiology and clinical pharmacology studies suggest that reducing FXI levels may efficiently prevent thrombosis with minimal bleeding risk, thus making FXI a very attractive target for treatment and prevention of thromboembolic diseases. For example, severe FXI deficiency has been associated with a lower incidence of ischemic stroke and venous thromboembolism events,12-14 whereas increased FXI levels are associated with increased thrombosis risk.15,16 Moreover, a study in patients undergoing elective total knee arthroplasty who received an FXI–antisense oligonucleotide (FXI-ASO) provided pharmacologic evidence that thrombosis can be safely prevented by reducing FXI levels.17 Importantly, bleeding manifestations in humans with severe FXI deficiency are infrequent, often mild, and injury induced, and they preferentially affect tissues with increased fibrinolytic activity, such as oral mucosa, nasal mucosa, and urinary tract. Spontaneous bleeding events in vital organs are extremely rare.18,19
The present article describes the generation and the preclinical and early-clinical evaluation of an inhibitory FXI antibody (MAA868) that binds both FXI and FXIa with high affinity and displays favorable pharmacokinetic (PK) and pharmacodynamic (PD) properties.
Materials and methods
Extended Materials and methods are provided in the supplemental Methods (available on the Blood Web site).
Phage display
A commercially available phage display library (HuCAL PLATINUM library)20 was used to conduct liquid phase phage panning with full-length biotinylated FXIa and FXI and with the biotinylated catalytic/protease domain (CD) of FXIa. Variable domain fragments of heavy (VH) and light chains of specific binders were used to generate full-length immunoglobulin G1 (IgG1). Six light chain framework mutations were introduced to make the MAA868 amino acid sequence as similar as possible to the human germline Vλ1_1c sequence (“germlining”). The VH framework (VH3-23) did not require any changes.
Binding measurements
MAA868 affinity to human and cynomolgus monkey FXI and FXIa was determined by using solution equilibrium titration measurements. Serial dilutions of antigens were coincubated with a fixed concentration of MAA868 until equilibrium was reached. Free antibody was captured on Meso Scale Discovery array plates and detected with an electrochemiluminescence-labeled detection antibody, and the signals were plotted against the antigen concentration. The dissociation constant (KD) was determined by nonlinear curve fitting.
X-ray structure solution
Equimolar mixtures of recombinantly produced FXIa CD and the antigen-binding fragment (Fab) portion of MAA868 generated by papain cleavage of the IgG were used to grow crystals. The crystal used for data collection was obtained at 277 K employing sitting drop vapor diffusion and diffracted to high resolution (2.04 Å) with unit cell dimensions of a = 191.27, b = 53.22, c = 65.164 Å, α = 90.0, β = 94.56, γ = 90.0 (Space group C2), and with one copy of the complex per asymmetric unit. The complex structure was solved by molecular replacement using structures of the FXI CD and a truncated Fab previously solved in-house as search models using PHASER.21 The data collection and refinement statistics are summarized in supplemental Table 2.
Ferric chloride mouse thrombosis model
All animal procedures complied with the policies of the Novartis Animal Care and Use Committee. Mice deficient in FXI (FXI−/− mice) on a C57BL/6 background were reconstituted IV with 0.6 mg/kg human FXI (hFXI) followed by IV administration of antibodies. Thrombosis was induced by applying 5% ferric chloride (FeCl3) solution to the surface of the exposed carotid artery, and blood flow was measured.
Cynomolgus monkey ascending dose study
The study was conducted in accordance with the US Department of Agriculture Animal Welfare Regulations and approved by the Novartis Animal Care and Use Committee. MAA868 was administered subcutaneously (SC) and IV to female cynomolgus monkeys as solution in phosphate-buffered saline (vehicle). Blood samples (for total MAA868 plasma concentration, free FXI [FXIf] plasma concentration, and coagulation assessment by activated partial thromboplastin time [aPTT]) were collected at specified time points after each dose.
First-in-human study
The first-in-human (FiH) study was a randomized, subject and investigator blinded, placebo-controlled, single ascending dose study, conducted at a single site (Medpace Clinical Pharmacology Unit, Cincinnati, OH) in accordance with the ICH Harmonised Tripartite Guidelines for Good Clinical Practice and with the ethical principles laid down in the Declaration of Helsinki. All study procedures were reviewed and approved by the site’s institutional review board (Schulman IRB, Cincinnati, OH; reference #201603918). All subjects provided written informed consent before any study-related procedures were performed.
The study comprised a screening period (∼28 days), a baseline period (∼3 days), and a 7-day inpatient observation period (days 1-8), followed by an outpatient observation period (days 9-106). Male and postmenopausal or surgically sterile female subjects between 18 and 60 years of age were eligible if they were in good health. aPTT and FXI coagulation activity (FXI:C) were measured in citrated platelet-poor plasma using Actin FS (Siemens Healthcare Diagnostics, Tarrytown, NY) as a reagent at Esoterix Coagulation, Inc. (Englewood, CO). Total MAA868 was quantified by immune–liquid chromatography/tandem mass spectrometry and measured in a central laboratory (LGC, Fordham, Cambridgeshire, United Kingdom).
Results
Generation of a human IgG1 that binds FXIa and FXI with high affinity
To generate a potent FXI inhibitory antibody, we sought to develop an antibody that binds to the CD of both, the activated enzyme and the zymogen. Phage display technology was used to generate fully human antibodies. One unique phage panning–derived antibody clone, NOV1090, exhibited high binding affinity to FXI and FXIa when expressed as a Fab (Table 1) and was selected for functional testing. NOV1090 Fab inhibited FXIa with a 50% inhibitory concentration of 2.8 nM in an enzyme activity assay with a fluorescent peptide substrate but did not inhibit other human serine protease–type coagulation factors, including factor VIIa, factor IXa, factor Xa, FXIIa, thrombin, and its closest homolog plasma kallikrein (supplemental Table 1), indicating that NOV1090 Fab is a highly specific FXIa inhibitor. MAA868 was generated by converting NOV1090 Fab into a full-length human IgG1 and by rendering its amino acid sequence as close as possible to a human germline sequence. Binding measurements revealed that MAA868 binds with high affinity to the CD of human FXI and FXIa (KD of 1.3 ± 0.3 pM and 4.7 ± 2.1 pM, respectively), as well as to cynomolgus monkey FXI and FXIa (Table 1) but not to mouse or rat FXI and FXIa, consistent with sequence differences within MAA868-binding epitope regions (supplemental Figure 1).
Affinity (KD) of FXI antibodies determined by using solution equilibrium titration measurements
| Antibody . | Antigen . | KD . | n . |
|---|---|---|---|
| NOV1090-Fab | Human FXIa | 244 ± 96 pM | 3 |
| MAA868-IgG | 4.7 ± 2.1 pM | 3 | |
| NOV1090-Fab | Human FXI | 163 ± 24 pM | 3 |
| MAA868-IgG | 1.3 ± 0.3 pM | 3 | |
| MAA868-IgG | cyno FXIa | 6-19 pM | 2 |
| MAA868-IgG | cyno FXI | 4-6 pM | 2 |
| Antibody . | Antigen . | KD . | n . |
|---|---|---|---|
| NOV1090-Fab | Human FXIa | 244 ± 96 pM | 3 |
| MAA868-IgG | 4.7 ± 2.1 pM | 3 | |
| NOV1090-Fab | Human FXI | 163 ± 24 pM | 3 |
| MAA868-IgG | 1.3 ± 0.3 pM | 3 | |
| MAA868-IgG | cyno FXIa | 6-19 pM | 2 |
| MAA868-IgG | cyno FXI | 4-6 pM | 2 |
cyno, cynomolgus monkey.
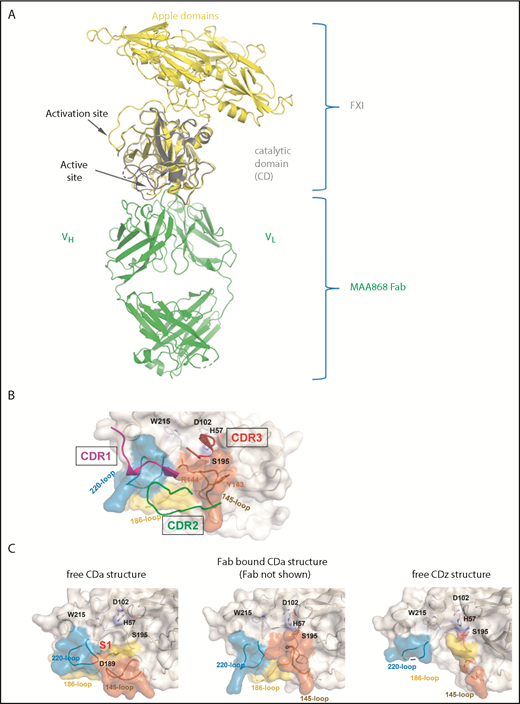
High-resolution X-ray structure of MAA868 (Fab) in complex with the catalytic domain of FXIa reveals a unique binding mechanism
To better understand how MAA868 is able to bind both FXI and FXIa, with equally high affinity, we determined the X-ray structure of the Fab portion of MAA868 in complex with the FXIa CD at a resolution of 2.04 Å. The structure revealed that MAA868 binds to the protease active site region (Figure 1A) with the complementarity-determining region (CDR) 3 of the heavy chain covering portions of the S3, S2, S1-β, and S1′ subsites of the protease (Figure 1B). Binding of the heavy chain CDR1 and CDR2 loops in concert induces conformational changes in the protease 145- and 220-loops (chymotrypsin numbering), leading to auto-occlusion of the S1 pocket and the S2′ subsite residues Arg144 and Tyr143, respectively. In addition, the 4 N-terminal FXI protease residues as well as residues surrounding and including Asp189 become disordered. These conformational changes affect portions of the enzyme that are critical for its catalytic activity. Thus, MAA868 traps and stabilizes an inactive conformation of the protease. The portions of the FXI CD that have changed conformation or have become disordered as a result of antibody binding (Figure 1C) are disordered in the zymogen as well (Protein Data Bank accession number: 2F83).22 Hence, the conformation of the Fab-bound CD of FXIa resembles the inactive CD of the zymogen and provides a plausible explanation for the strong binding of MAA868 to both FXI and FXIa. The fact that FXIIa can still cleave FXI bound to MAA868 (supplemental Figure 2) further supports the model that MAA868 locks FXIa in a zymogen-like state.
X-ray structure of MAA868 Fab in complex with FXI CD. (A) Ribbon representation of the MAA868 Fab–FXI CD complex structure (green and yellow with gray) in superposition with a published FXI zymogen structure (PDB 2F83)22 is shown. The apple domains of FXI, which were not part of the MAA868 Fab–FXI CD complex, are shown in yellow. The activation cleavage site and active site location are indicated by arrows, showing that MAA868 binds close to the active site but away from the activation cleavage site (see also supplemental Figure 2). (B) MAA868 Fab binding site on FXI is shown. Parts of the heavy and light chains that make close contacts with FXI are shown as ribbons. The catalytic domain is shown as gray surface, and the side chains of selected amino acid residues are shown including residues of the catalytic triad. In addition, the heavy chain loops CDR1 (magenta), CDR2 (green), and CDR3 (red) are shown. Y145 and R144 are shown with their side chains (see text). (C) Fab-bound CD conformation shares structural similarities with the inactive CD in the zymogen structure (PDB 2F83). Compared with the free CDa structure (left panel), S1 pocket is not formed in either the Fab bound CDa structure (middle panel) or the free CDz structure (right panel). Three crucial loops near the active site that are all disordered in the free CDz structure (right panel) are either shifted (220 and 145 loop) or also disordered (186 loop) in the Fab complex (middle panel).
X-ray structure of MAA868 Fab in complex with FXI CD. (A) Ribbon representation of the MAA868 Fab–FXI CD complex structure (green and yellow with gray) in superposition with a published FXI zymogen structure (PDB 2F83)22 is shown. The apple domains of FXI, which were not part of the MAA868 Fab–FXI CD complex, are shown in yellow. The activation cleavage site and active site location are indicated by arrows, showing that MAA868 binds close to the active site but away from the activation cleavage site (see also supplemental Figure 2). (B) MAA868 Fab binding site on FXI is shown. Parts of the heavy and light chains that make close contacts with FXI are shown as ribbons. The catalytic domain is shown as gray surface, and the side chains of selected amino acid residues are shown including residues of the catalytic triad. In addition, the heavy chain loops CDR1 (magenta), CDR2 (green), and CDR3 (red) are shown. Y145 and R144 are shown with their side chains (see text). (C) Fab-bound CD conformation shares structural similarities with the inactive CD in the zymogen structure (PDB 2F83). Compared with the free CDa structure (left panel), S1 pocket is not formed in either the Fab bound CDa structure (middle panel) or the free CDz structure (right panel). Three crucial loops near the active site that are all disordered in the free CDz structure (right panel) are either shifted (220 and 145 loop) or also disordered (186 loop) in the Fab complex (middle panel).
MAA868 specifically inhibits FXIa function within the intrinsic coagulation pathway
Coagulation assays in human plasma were used to assess the in vitro anticoagulation activity of MAA868. We found that MAA868 prolonged the clotting time of human plasma in a concentration-dependent manner as determined by using the aPTT assay (Figure 2A), which measures the impact of FXIa inhibition on the intrinsic pathway of the coagulation cascade. Similarly, MAA868 reduced the amount of thrombin in human plasma in a concentration-dependent manner in the thrombin generation assay (TGA), which uses very low tissue factor concentrations as a trigger, indicating an inhibitory effect of MAA868 on the thrombin→FXIa feed-forward loop (Figure 2B).
MAA868 concentration dependently prolongs aPTT and inhibits thrombin generation in human plasma. (A) The concentration-dependent effect of MAA868 on the clotting time of human plasma in the aPTT assay is shown. In the aPTT assay, trigger reagents specific for triggering the intrinsic pathway are used (phospholipid, silicate, and calcium chloride). MAA868 prolongs aPTT clotting times in a concentration-dependent manner (50% inhibitory concentration [IC50] = 13 nM). Doubling of aPTT clotting time was observed at 15 nM. (B) The dose-dependent effect of MAA868 on inhibiting thrombin generation in the TGA is shown. In the TGA, the thrombin→FXIa feedback loop is triggered by low concentrations of tissue factor, and thrombin activity is measured by adding an artificial, fluorogenic substrate to plasma. An IC50 of 24 nM and a residual thrombin concentration of 159 nM, generated by low tissue factor concentration, were calculated for MAA868. The residual thrombin concentration in this assay is likely due to thrombin generation induced by small amounts of tissue factor.
MAA868 concentration dependently prolongs aPTT and inhibits thrombin generation in human plasma. (A) The concentration-dependent effect of MAA868 on the clotting time of human plasma in the aPTT assay is shown. In the aPTT assay, trigger reagents specific for triggering the intrinsic pathway are used (phospholipid, silicate, and calcium chloride). MAA868 prolongs aPTT clotting times in a concentration-dependent manner (50% inhibitory concentration [IC50] = 13 nM). Doubling of aPTT clotting time was observed at 15 nM. (B) The dose-dependent effect of MAA868 on inhibiting thrombin generation in the TGA is shown. In the TGA, the thrombin→FXIa feedback loop is triggered by low concentrations of tissue factor, and thrombin activity is measured by adding an artificial, fluorogenic substrate to plasma. An IC50 of 24 nM and a residual thrombin concentration of 159 nM, generated by low tissue factor concentration, were calculated for MAA868. The residual thrombin concentration in this assay is likely due to thrombin generation induced by small amounts of tissue factor.
MAA868 had no effect on clotting time up to an antibody concentration of 1.3 µM in the prothrombin assay, which measures changes in the extrinsic coagulation pathway (supplemental Figure 3). Thus, MAA868 inhibited FXIa, activated by FXIIa in the intrinsic pathway, as well as by thrombin in the thrombin→FXIa feed-forward loop, but did not interfere with the initial thrombin burst triggered by the extrinsic pathway, which is crucial for normal blood hemostasis.
MAA868 exhibits antithrombotic and anticoagulant activity in FXI−/− mice reconstituted with hFXI
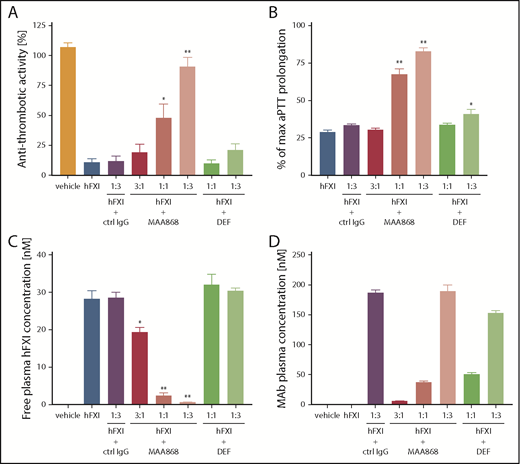
Because MAA868 does not bind mouse FXI, we used FXI−/− mice reconstituted with hFXI to evaluate MAA868 in vivo. FXI−/− mice are protected from thrombosis in the FeCl3 carotid artery thrombosis model23 but develop occlusive thrombi if reconstituted with hFXI (supplemental Figure 4A). In this model, MAA868 prevented formation of occlusive thrombi when present at molar excess over hFXI in plasma (Figure 3A; supplemental Figure 4B). MAA868 prolonged aPTT in these “humanized” FXI−/− mice to levels similar to those in FXI−/− animals that were not reconstituted with hFXI (Figure 3B; supplemental Figure 4C). MAA868 dosed equimolar to hFXI led to comparable plasma exposures for MAA868 and hFXI (compare Figure 3C and 3D) and reduced free hFXI already by ∼90% (Figure 3C) due to the high affinity of MAA868. An increased aPTT and some thromboprotection was observed at this dose; however, >95% reduction of free hFXI seem to be required for full thromboprotection in this model (Figure 3A,C).
Antithrombotic and anticoagulant activity of MAA868 in FXI−/− mice reconstituted with hFXI. A total of 0.6 mg/kg hFXI was administered IV to FXI−/− mice followed by IV administration of vehicle, an isotype control IgG, MAA868, or DEF. Thrombosis was induced 20 minutes later by applying 5% FeCl3 to the surface of the carotid artery, and blood flow was monitored for 30 minutes thereafter. Blood was collected from the vena cava, and plasma was prepared for aPTT analysis as well as for FXIf and antibody measurements. (A) Antithrombotic activity is shown for antibodies injected at the indicated molar ratios of hFXI and was assessed by measuring carotid blood flow (inversely related to extent of vessel occlusion) over 30 minutes. Antithrombotic activity (%) = (mean blood flow during the thrombogenic period)/(baseline blood flow) × 100. (B) Anticoagulant activity of antibodies as assessed by aPTT measurement and is shown as % of maximum aPTT observed in FXI−/− not reconstituted with hFXI. (C) Free total hFXI plasma levels (including free hFXIa) are shown. (D) Total monoclonal antibody (mAb) plasma levels measured during the study. Values for each group are shown as means ± standard error of the mean. *P < .05 vs hFXI, **P < .01 vs hFXI.
Antithrombotic and anticoagulant activity of MAA868 in FXI−/− mice reconstituted with hFXI. A total of 0.6 mg/kg hFXI was administered IV to FXI−/− mice followed by IV administration of vehicle, an isotype control IgG, MAA868, or DEF. Thrombosis was induced 20 minutes later by applying 5% FeCl3 to the surface of the carotid artery, and blood flow was monitored for 30 minutes thereafter. Blood was collected from the vena cava, and plasma was prepared for aPTT analysis as well as for FXIf and antibody measurements. (A) Antithrombotic activity is shown for antibodies injected at the indicated molar ratios of hFXI and was assessed by measuring carotid blood flow (inversely related to extent of vessel occlusion) over 30 minutes. Antithrombotic activity (%) = (mean blood flow during the thrombogenic period)/(baseline blood flow) × 100. (B) Anticoagulant activity of antibodies as assessed by aPTT measurement and is shown as % of maximum aPTT observed in FXI−/− not reconstituted with hFXI. (C) Free total hFXI plasma levels (including free hFXIa) are shown. (D) Total monoclonal antibody (mAb) plasma levels measured during the study. Values for each group are shown as means ± standard error of the mean. *P < .05 vs hFXI, **P < .01 vs hFXI.
MAA868 exhibits strong and sustained anticoagulant effects in cynomolgus monkeys
MAA868 was administered IV and SC to cynomolgus monkeys in a dose-escalation study to show target engagement by measuring FXIf levels and to evaluate its anticoagulant activity (aPTT prolongation) in vivo.
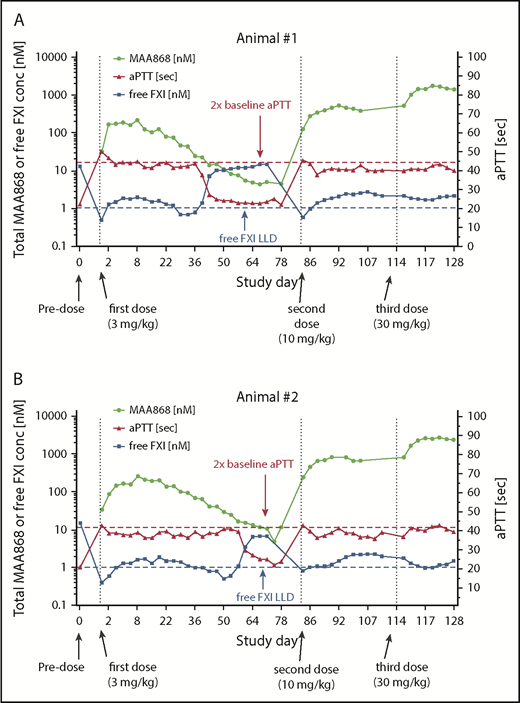
A single 3 mg/kg MAA868 SC dose significantly prolonged aPTT (Figure 4A-B), as long as MAA868 plasma exposure exceeded ∼20 nM (up to 5 and 8 weeks, respectively). Additional higher doses increased the plasma exposures of MAA868 proportionally but did not result in any significant additional aPTT prolongation. Accordingly, FXIf levels were sharply reduced after the first MAA868 dose and remained low until total MAA868 plasma levels declined to ∼20 nM. Similar effects were observed when MAA868 was dosed IV, with aPTT prolongation coinciding with total MAA868 plasma levels >15 to 25 nM and reduction of FXIf levels by ∼90% from pretreatment levels (supplemental Figure 6). PK parameters for both routes of administration are described in Table 2 and concentration-time profiles are shown in supplemental Figure 7.
MAA868 prolongs aPTT in cynomolgus monkeys and inversely lowers FXIf when dosed SC. MAA868 was administered subcutaneously to 2 female cynomolgus monkeys at 3, 10, and 30 mg/kg on days 1, 85, and 114, respectively. Two control animals received vehicle on days 1, 85, and 114. MAA868 plasma exposure, FXIf, and aPTT as a function of time are shown for the 2 individual animals (A) and (B) starting at 6 h postdose. Doubling of aPTT (2× baseline aPTT) and free FXI lower level of detection (LLD) are indicated by dashed lines. Total plasma MAA868 and FXIf were quantified by using custom sandwich immunoassays.
MAA868 prolongs aPTT in cynomolgus monkeys and inversely lowers FXIf when dosed SC. MAA868 was administered subcutaneously to 2 female cynomolgus monkeys at 3, 10, and 30 mg/kg on days 1, 85, and 114, respectively. Two control animals received vehicle on days 1, 85, and 114. MAA868 plasma exposure, FXIf, and aPTT as a function of time are shown for the 2 individual animals (A) and (B) starting at 6 h postdose. Doubling of aPTT (2× baseline aPTT) and free FXI lower level of detection (LLD) are indicated by dashed lines. Total plasma MAA868 and FXIf were quantified by using custom sandwich immunoassays.
Mean PK parameters after single (rising) dose administrations of MAA868 in female cynomolgus monkeys
| Dose (mg/kg) . | Route . | Tmax (h) . | Cmax (µg/mL) . | AUC0-14d (µg ∙ d/mL) . |
|---|---|---|---|---|
| 3 | IV | 0.25 | 96.0 | 544 |
| SC | 168 | 36.0 | 360 | |
| 10 | IV | 0.25 | 325 | 1810 |
| SC | 132 | 101 | 1160 | |
| 30 | IV | 1.08 | 1170 | 6770 |
| SC | 132 | 344 | 4140 |
| Dose (mg/kg) . | Route . | Tmax (h) . | Cmax (µg/mL) . | AUC0-14d (µg ∙ d/mL) . |
|---|---|---|---|---|
| 3 | IV | 0.25 | 96.0 | 544 |
| SC | 168 | 36.0 | 360 | |
| 10 | IV | 0.25 | 325 | 1810 |
| SC | 132 | 101 | 1160 | |
| 30 | IV | 1.08 | 1170 | 6770 |
| SC | 132 | 344 | 4140 |
Tmax, median time-to-peak MAA868 plasma concentrations.
No safety concerns arose from the study, and, importantly, no evidence of bleeding was observed (Table 3). In addition, no significant toxicity findings or signs of bleeding were observed in a repeat dose general toxicity Good Laboratory Practice study in cynomolgus monkeys up to 13 weeks (supplemental Table 3); this study included the highest tested dose of 100 mg/kg per week, which led to an exposure exceeding the anticipated human therapeutic exposure by ∼400-fold.
Safety evaluation after single (rising) dose administrations of MAA868 in female cynomolgus monkeys
• No mortality occurred during the study |
• No drug-related effects on clinical signs, body weight, food consumption, ophthalmologic and electrocardiographic parameters, hematology, clinical chemistry, urinalysis |
• No macroscopic or microscopic indications of bleeding (including no bruising, no gum bleeds, and no extended bleeding at either SC or IV drug injection sites or blood sampling sites) |
• No occult blood detected (in feces) |
• No target organs of toxicity identified |
• No mortality occurred during the study |
• No drug-related effects on clinical signs, body weight, food consumption, ophthalmologic and electrocardiographic parameters, hematology, clinical chemistry, urinalysis |
• No macroscopic or microscopic indications of bleeding (including no bruising, no gum bleeds, and no extended bleeding at either SC or IV drug injection sites or blood sampling sites) |
• No occult blood detected (in feces) |
• No target organs of toxicity identified |
MAA868 was safe, well tolerated, and showed robust anticoagulation activity in an FiH study with healthy volunteers
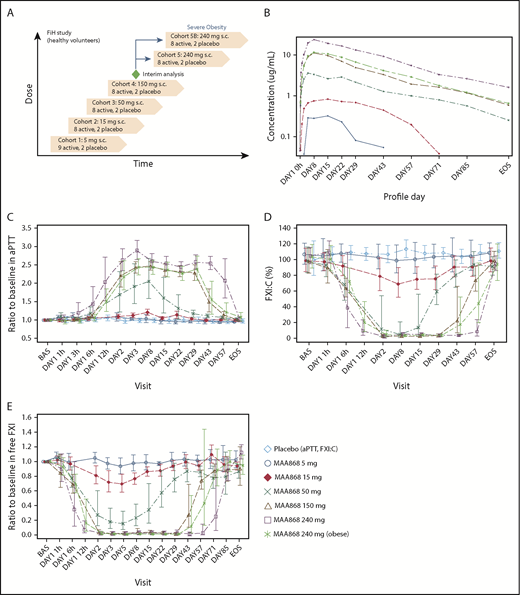
To assess safety, tolerability, and the PK and PD parameters of MAA868 in humans, we administered single, ascending doses of MAA868 (5 mg up to 240 mg administered SC) to sequentially enrolled cohorts. Cohorts 1 to 5 enrolled healthy volunteers with normal body weight, and cohort 5B enrolled severely obese subjects with body mass index >35 kg/m2 who were otherwise healthy. The study design is depicted in Figure 5A. A total of 61 subjects were enrolled and received MAA868 or placebo at an 8:2 ratio for each dose cohort. The demographic and baseline characteristics were similar across the treatment groups, with the exception of higher body weight and body mass index of subjects in cohort 5B. Supplemental Table 4 provides a descriptive analysis of demographic characteristics according to treatment group.
Single SC administration of MAA868 induced a sustained, dose-dependent anticoagulant effect in humans. (A) Depicts the design of the FiH study, a single ascending dose, randomized, subject- and investigator-blinded, placebo-controlled study. Healthy volunteers were recruited into consecutive cohorts and administered SC injections of placebo or increasing single doses of MAA868. In addition, a cohort (cohort 5B) of severely obese but otherwise healthy subjects received a single SC dose of 240 mg. (B) Plasma total MAA868 concentration–time profile of each cohort in a semi-logarithmic view; data are expressed as geometric mean ± standard deviation. Change of aPTT (C), FXI:C (D), and FXIf levels (E) are depicted over time for each cohort; data are expressed as ratio to baseline and displayed as geometric mean ± 95% CI. BAS, baseline; EoS, end of study (day 106).
Single SC administration of MAA868 induced a sustained, dose-dependent anticoagulant effect in humans. (A) Depicts the design of the FiH study, a single ascending dose, randomized, subject- and investigator-blinded, placebo-controlled study. Healthy volunteers were recruited into consecutive cohorts and administered SC injections of placebo or increasing single doses of MAA868. In addition, a cohort (cohort 5B) of severely obese but otherwise healthy subjects received a single SC dose of 240 mg. (B) Plasma total MAA868 concentration–time profile of each cohort in a semi-logarithmic view; data are expressed as geometric mean ± standard deviation. Change of aPTT (C), FXI:C (D), and FXIf levels (E) are depicted over time for each cohort; data are expressed as ratio to baseline and displayed as geometric mean ± 95% CI. BAS, baseline; EoS, end of study (day 106).
Safety
The 240-mg SC dose was chosen as the maximal dose because PK/PD modeling based on an interim analysis of preliminary data from cohort 4 suggested that doses >240 mg may result in aPTT prolongations longer than intended (more than twofold for >42 days, which was prospectively considered the maximal ethically acceptable duration of PD effects in healthy volunteers). Physical examination, vital signs, hematology, blood biochemistry, and electrocardiogram revealed no changes in the MAA868 treatment groups or in the placebo group. There were no reports of injection site reactions or hypersensitivity reactions in subjects administered MAA868. One (8.3%) of 12 placebo subjects (at day 29) and 4 (8.2%) of 49 active subjects (1 in cohort 2, day 8; 1 in cohort 3, day 29; and 2 in cohort 5B, day 8) tested positive for fecal occult blood at one time point each. However, none of these findings was confirmed during follow-up visits, and none of these subjects had a drop in hemoglobin levels or other signs of bleeding. The incidence of adverse events (AEs) was well balanced between the MAA868 treatment groups and the placebo group. There was a total of 9 AEs (MAA868, n = 7 [14%]; placebo, n = 2 [17%]) considered by the investigator to be potentially drug related, and all were of mild intensity with headache (4 events) being the only AE reported in >1 subject. AEs reported in ≥2 subjects are listed in supplemental Table 5. There was 1 fatal, nonrelated, serious AE (SAE) of a cardiac arrest in a subject with severe obesity (MAA868, 240-mg dose) undergoing general anesthesia for an elective surgery on day 89; FXIf was 98% of baseline at day 84 in this subject, suggesting that MAA868 anticoagulation effects were reversed at the time of the SAE. Two additional SAEs, also not related to MAA868, were reported in 1 subject (MAA868, 50-mg dose) who required popliteal artery bypass surgery and bone reconstruction due to a gunshot wound on day 11 and later developed a wound infection. Interestingly, despite an aPTT prolongation of ∼2.5 fold, there were no reports of excessive bleeding in this patient.
PK parameters
MAA868 exposures were dose proportional within the studied dose range (maximum serum concentration [Cmax] and the area under the curve [AUC] from zero to infinity, with 90% confidence intervals [CIs] for slope β of 0.97 to 1.10 and 0.93 to 1.17, respectively). Plasma total MAA868 concentration–time profiles are presented in Figure 5B (semi-logarithmic view) and supplemental Figure 8 (linear view). Intersubject variability of systemic MAA868 exposures was low to moderate (coefficient of variation, 9.4% to 53.5% for Cmax and 15.4% to 42.7% for AUC from zero to infinity). The median time-to-peak MAA868 plasma concentrations ranged from 7 to 21 days and were dose independent. However, levels adequate to prolong aPTT more than twofold were achieved by 24 hours (discussed in the following section). Severe obesity was associated with moderately decreased MAA868 Cmax and AUC. The terminal elimination half-life (T1/2) ranged from 20.1 to 28.6 days. A summary of PK parameters for each dose cohort is given in supplemental Table 6.
PD parameters
Dose- and time-dependent prolongations in aPTT and reductions in FXI:C and FXIf levels were observed. Specifically, statistically significant aPTT prolongations started with the 50-mg dose but were maintained more than twofold only at higher doses (from 24 hours’ postdose up to day 29 at the 150-mg dose in cohort 4 and at the 240-mg dose in cohort 5B, and from 12 hours’ postdose up to day 57 at the 240-mg dose in cohort 5). Figure 5C displays the aPTT ratio to baseline over time according to treatment. Exposure–response modeling using a maximum effect model revealed that maximal aPTT prolongations of ∼2.5 fold and 70 seconds were achieved with MAA868 plasma levels >30 nM. Conversely, robust and sustained reductions of FXIf and FXI:C ≥80% were observed from day 2 up to day 29 with the 150-mg dose and with the 240-mg dose from 12 hours’ postdose up to day 57 (normal body weight) and up to day 43 (severe obesity), respectively. The maximal inhibition was observed on day 2 with a mean FXI:C of 3% of baseline (95% CI, 2-4) in the 150-mg and 240-mg dose group (normal body weight); of note, no complete suppression (<1% FXI:C) was observed at any time point. The time courses for changes in FXIf and FXI:C ratio to baseline are displayed according to treatment in Figure 5D and E. Likely due to a T1/2 extension of FXI bound to MAA868 and possibly increased FXI biosynthesis, a slight increase in total FXI levels was observed (up to ∼50% with the 150-mg dose and ∼70% with the 240-mg dose), however, without affecting aPTT. No changes were observed in prothrombin or thrombin time (supplemental Figure 9A-B).
Discussion
With the goal of developing an effective and safer anticoagulant, we chose to develop an FXI antibody that binds both the FXI zymogen and active FXIa. Human genetic data suggest that deficiency of the FXI zymogen can safely prevent thrombosis.12-14 Human pharmacologic data with FXI-ASO–mediated reduction of FXI zymogen levels provides further validation.17 In addition, recent descriptions of an antibody specifically directed against FXIa suggest that FXIa inhibition alone can have sufficient antithrombotic activity, at least in a preclinical model.24 However, the coagulation cascade features rapid activation and multiple amplification mechanisms, and active site–specific inhibitors of coagulation proteases face the kinetic challenge of binding with sufficient speed and completeness and in competition with natural substrates to block this process. A prebound FXI zymogen inhibitor would not face this challenge. Whether there are clinically relevant advantages of zymogen-focused or active site–focused mechanisms is unknown. Hence, we sought an antibody with features of both mechanisms, and biochemical studies and X-ray structural analysis revealed that MAA868 binds to the active site region of the CD of FXIa and induces conformational changes that inactivate the protease and prevent substrates from binding to the active site of FXIa. By inhibiting the active enzyme and by trapping circulating zymogen, MAA868 prevents FXI from entering the coagulation process and is capable of inhibiting any FXIa that has already been formed.
Our in vitro and in vivo studies show that MAA868 potently, selectively, and sustainably inhibits FXI-mediated coagulation. Specifically, MAA868 prolonged clotting time in a human plasma aPTT assay and reduced thrombin formation in the TGA, 2 assays sensitive to changes in the intrinsic pathway and thrombin-induced FXI activation via a positive feedback loop, respectively. Conversely, MAA868 did not affect prothrombin time, which is sensitive to changes in the extrinsic pathway.
In a mouse thrombosis model, substantial MAA868-dependent FXI reduction led to strong thromboprotection accompanied by anticoagulant effects. The sensitivity of the FeCl3-induced thrombosis model to anticoagulant and antiplatelet agents varies with the intensity of FeCl3 injury, and the model used here was additionally dependent on reconstitution with hFXI. The relatively high levels of FXI reduction required for full thromboprotection in this model may or may not translate to human thrombosis.
This thrombosis model proved very useful to select a lead antibody and to compare hFXI-specific antibodies with different mechanism of actions. Interestingly, an FXIa-specific antibody (DEF) that has comparable (if not higher) affinity for FXIa than MAA868 was not as efficacious in this model as MAA868 at the antibody and hFXI doses tested, supporting our hypothesis that developing a dual zymogen/active form binder might be advantageous. Although supported by preclinical data, the FXI hypothesis (anticoagulation without excessive bleeding) must ultimately be tested in humans. We believe that MAA868 is a promising antibody to conduct such studies.
Our FiH study showed that single SC administrations of up to 240 mg MAA868 were safe and well tolerated and dose dependently resulted in robust and sustained FXI inhibition beyond 4 weeks, which is anticipated to result in strong anticoagulation efficacy as well as thromboprotection. For example, an FXI-ASO dose of 300 mg, which resulted in an ∼80% reduction of FXI, was superior to enoxaparin in preventing venous thromboembolism in patients undergoing knee replacement surgery but did not increase bleeding risk (in fact, a numerically smaller number of bleeding episodes were observed).17 It is currently unclear whether higher reductions of FXI levels, as achievable with MAA868, may be even more efficacious in thromboembolism protection. To this end, it was suggested that severe FXI deficiency with residual FXI:C <15% confers protection from stroke and venous thromboembolism,13,14 whereas others found that even moderate FXI deficiency (residual FXI activity from 30% to 50%) already provides some protection from venous thrombosis and cardiovascular events.12
Anticipating that a large percentage of patients in need of a safer anticoagulation therapy will be overweight, we also investigated MAA868 in a cohort of severely obese subjects. Exposure to MAA868 and duration of FXI inhibition were moderately lower in this cohort, possibly due to the observed larger volume of distribution and slightly faster clearance of MAA868. Further studies are needed to determine whether dose adjustment in patients with severe obesity is required.
Besides the mild effect of body weight on PK parameters, it is expected that MAA868 will have a very predictable in vivo profile, due to the absence of renal clearance and a low risk for drug–drug interactions. Hence, MAA868 should also be suitable for an elderly population with multiple comorbidities and/or underlying chronic kidney disease, a patient population that often does not receive sufficient antithrombotic therapy due to the limitations of the currently available treatment options.
The ease of use associated with a once-monthly SC administration of MAA868 may result in enhanced adherence and improved efficacy compared with small-molecule–based anticoagulation therapies that may lose efficacy when trough concentrations are not maintained. The dose-dependent and relatively quick onset of action with MAA868 seems advantageous over the previously described FXI-ASO, which required multiple administrations over weeks to reduce FXI to therapeutic levels.17 A recently described small-molecule FXI inhibitor administered IV has a short T1/2 and is only suitable for short-term therapy (ranging from hours to days).25
MAA868 is expected to have a greatly reduced bleeding risk compared with current anticoagulant agents, and, in contrast to FXa or thrombin inhibition, a reversal agent might not be needed. However, it may be desirable to have a reversal agent available during early clinical trials to mitigate any residual bleeding risk or should urgent surgery be required in tissues with high fibrinolytic activity, such as the dental, oral and nasal mucosa, and urinary tract. To this end, we conducted in vitro thromboelastometry-monitoring experiments with MAA868 and recombinant factor VIIa (rFVIIa, NovoSeven), a commercially available, nonplasma-derived therapeutic product that has been used in FXI deficiency.26 These data showed that rFVIIa can bypass MAA868-dependent inhibition of FXIa in human blood (supplemental Figure 10), suggesting that NovoSeven may be used to reverse MAA868 anticoagulation effects.
In summary, MAA868, a fully human, inhibitory FXI antibody, is expected to effectively inhibit FXI activity and warrants further investigation in phase 2/3 clinical studies to determine safety and efficacy compared with current anticoagulant agents. Given the assumed safety profile associated with FXI inhibition, MAA868 may serve as the first long-term anticoagulant with once-monthly SC dosing, including patients with high bleeding risk. MAA868 therefore has the potential to introduce a paradigm shift in anticoagulation therapy, away from short-acting agents that are subject to peak–trough excursions and need to balance efficacy with bleeding risk, and toward safe, long-acting FXI inhibitors able to target maximal efficacy.
The online version of this article contains a data supplement.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Acknowledgments
The authors are grateful to the subjects who participated in the FiH study as well as to Lukasz Biernat and the other staff at the clinical study site, the Medpace Clinical Pharmacology Unit in Cincinnati, who performed the FiH study. Furthermore, the authors thank David Conen, (Hamilton, ON, Canada) and Bernd Jilma (Vienna, Austria) for helpful scientific discussions regarding the design of the clinical study, as well as Markus Hinder (Novartis, Basel, Switzerland) and Shaun Coughlin (Novartis, Cambridge, MA) for critically reviewing the manuscript and for helpful scientific discussions. They are grateful to George Broze (Washington University, St. Louis, MO) for providing the FXI−/− mice and to Nancy Yao and Jacqueline Lengyel from the Novartis Preclinical Safety Group for overseeing the cynomolgus monkey studies. The authors also thank Daniela Stoellner for valuable assistance with assay development, Christian Wiesmann for providing guidance in X-ray crystallography, and Gordon Turner, Mary Russell, and Carla Klattenhoff for many helpful discussions.
This work was funded by Novartis.
Authorship
Contribution: A.W.K designed and supervised preclinical studies, analyzed and interpreted data, and cowrote the manuscript; N.S. performed X-ray structure solution, and analyzed and interpreted data; S.M. designed and supervised preclinical studies, and analyzed and interpreted data; S.E. designed and supervised the phage panning experiments, and analyzed and interpreted data; J.S. supervised the clinical study and contributed to its design; Y.Z. performed statistical analysis; P.M. analyzed and interpreted clinical data; Z.C. designed the mouse model study and collected and analyzed data; J.Y. analyzed samples from mouse and monkey studies, performed binding assays, and collected data; X.H. performed coagulation experiments; Y.-H.C. designed and supervised coagulation experiments; S.S. developed assays for preclinical studies and collected data; G.H. performed phage panning and collected data; K.D. supervised the monkey study and interpreted data; L.M., A.H., and F.V. generated crucial reagents; M.S. developed the solution equilibrium titration assay and supervised experiments; P.R. designed reagents and experiments; U.H. developed coagulation assays and supervised experiments; J.D. and C.K. developed assays for clinical studies; J.E. designed experiments; B.S. performed PK analysis for preclinical studies; K.K. performed PK analysis for clinical studies; D.L.F. developed the mouse model and contributed to the design of preclinical studies; P.H. designed and supervised toxicology studies; S.C. performed the thromboelastometry experiments; C.T.B. contributed to the design and interpretation of the clinical study; R.J.A.F. cosupervised the clinical study, analyzed and interpreted data, and cowrote the manuscript; and Y.K. contributed to the design of preclinical experiments and the clinical study, cosupervised the clinical study, analyzed and interpreted data, and cowrote the manuscript.
Conflict-of-interest disclosure: All authors are current or former employees of Novartis while engaged in the research project and own stock and/or stock options of Novartis. The following authors are listed as inventors on a patent application covering FXI antibodies: N.S., S.E., S.M, U.H, J.E., and Y.K.
The current affiliation for N.S. is Novartis Global Drug Development, Basel, Switzerland.
The current affiliation for S.M. is Molecular Partners AG, Zürich, Switzerland.
The current affiliation for B.S. is AbbVie Biotherapeutics Research, Redwood City, CA.
The current affiliation for K.K. is Ironwood Pharmaceuticals, Cambridge, MA.
The current affiliation for D.L.F. is National Kidney Foundation, New York, NY.
The current affiliation for Y.K. is Novartis Global Drug Development, Basel, Switzerland.
Correspondence: Alexander W. Koch, Cardiovascular and Metabolic Diseases, Novartis Institutes for BioMedical Research, Inc., 22 Windsor St, Cambridge, MA 02139; e-mail: alexander.koch@novartis.com; and Robert J. A. Frost, Translational Medicine, Novartis Pharma AG, Novartis Institutes for BioMedical Research, WSJ-386.12.48.44, CH-4002 Basel, Switzerland; e-mail: robert.frost@novartis.com.
REFERENCES
Author notes
R.J.A.F. and Y.K. are joint senior authors.

![Figure 2. MAA868 concentration dependently prolongs aPTT and inhibits thrombin generation in human plasma. (A) The concentration-dependent effect of MAA868 on the clotting time of human plasma in the aPTT assay is shown. In the aPTT assay, trigger reagents specific for triggering the intrinsic pathway are used (phospholipid, silicate, and calcium chloride). MAA868 prolongs aPTT clotting times in a concentration-dependent manner (50% inhibitory concentration [IC50] = 13 nM). Doubling of aPTT clotting time was observed at 15 nM. (B) The dose-dependent effect of MAA868 on inhibiting thrombin generation in the TGA is shown. In the TGA, the thrombin→FXIa feedback loop is triggered by low concentrations of tissue factor, and thrombin activity is measured by adding an artificial, fluorogenic substrate to plasma. An IC50 of 24 nM and a residual thrombin concentration of 159 nM, generated by low tissue factor concentration, were calculated for MAA868. The residual thrombin concentration in this assay is likely due to thrombin generation induced by small amounts of tissue factor.](https://ash.silverchair-cdn.com/ash/content_public/journal/blood/133/13/10.1182_blood-2018-10-880849/3/m_blood880849f2.png?Expires=1768071379&Signature=g6X5PFMUAPJIa73UL3nlvXeSLn29npW4t0ZHKjJYX5Qy4YOVMHwnsuY7Jgu3n2fQK8H~gAHGCz3XgC-nmFYlkpcvvohqiitGwR~nFT9S23tGFXiV~JB8URilUbK2weqoITPb0BVRnOK56iFY5zQZtdoEdVmsTj52sEbXw8yYfAdArEhh6nkXa~vSdz-oyGhQaU7Es0WYGi76vhaqi0UkkD3B38HfG54lSBiMnoAdwEkwOsltGxZxRtrtVaX5bZKkP6l9QnZ6O2fRCkfszGE7x-jTRIkAiEqZi78lMEL2RUk52Aj-WXqlDOS81yqijrOr6W7pzgMs8GIzbCy5vgSy~Q__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)
This feature is available to Subscribers Only
Sign In or Create an Account Close Modal