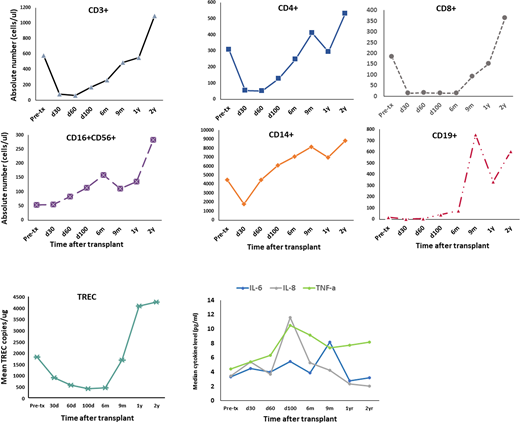
Umbilical cord blood transplantation (UCBT) is an alternative for patients who need hematopoietic stem cell transplant (HSCT), but lack HLA-matched adult donors. Rabbit anti-thymoglobulin (ATG) has been used in UCBT conditioning to achieve T cell depletion, but ATG-induced immunosuppression is associated with delayed immune reconstitution, increased infectious complications and higher non-relapse mortality. In a clinical trial of reduced intensity double-unit UCBT (dUCBT), we substituted low dose total body irradiation (TBI) for ATG to determine whether dUCBT without ATG would alter kinetics and quality of immune reconstitution. Thirty-one patients with hematopoietic malignancies and a median age of 58 yr were treated with Flu/Mel/TBI, followed by dUCBT and GVHD prophylaxis with tacrolimus and sirolimus. We examined reconstitution of immune cell populations, thymic regeneration by quantifying T cell receptor excision circles (TREC) and serum cytokines (IL-1β, IL-2, IL-4, IL-5, IL-6, IL-8, IL-10, IL-12, IFN-γ, TNF-α, IL-12p70, GM-CSF) using the LUNARISTM Human 11-Plex Cytokine Biochip384 from AYOXXA Biosystems. Assessments were done prior to and at 1, 2, 3, 6, 12 and 24 months after dUCBT. Results are based on 28 evaluable patients. The 2-yr overall survival and progression-free survival were 53% and 48%. Median time to neutrophil and platelet engraftment was 25 and 52 days, respectively. Before UCBT, the median values for most leukocyte subsets were below normal limits, except for monocytes. CD3+ cells remained depressed until 2 months post-transplant when gradually began to re-emerge. However, CD4+ and CD8+ subsets had distinct reconstitution kinetics (Figure 1). CD4+ T cells declined at 1 month but gradually increased and exceeded pre-transplant levels by 9 months after UCBT. In contrast, at 9 months, CD8+ lymphocytes remained depressed to 50% of pre-transplant levels, but increased thereafter and reached normal values by 2-yr. NK cells and monocytes reached normal values at 3 months post-UCBT. B cells were mostly undetectable for the first 6 months, followed by a 10-fold increase at 9 months and exceeded the upper normal limit by 2-yr. TREC, which were within normal range before transplant, decreased after UCBT but remained detectable between 1-6 months and recovered to normal levels by 9 months. Among cytokines, only IL-8, IL-6 and TNF-α displayed significant changes. IL-8 and IL-6 peaked at day 100 and 9 months, and subsequently declined. In contrast, TNF-α increased by day 100 and remained elevated thereafter. To evaluate functional immunity, we assessed correlations between viral reactivation and reconstitution of immune cell populations and thymic function. Nineteen patients experienced 24 clinically significant viral reactivations or infections, with 1-year cumulative incidence of 62%, which was comparable to 53% observed in dUCBT cohorts receiving ATG-containing conditioning. Although there was no difference in CMV, EBV, adenovirus and VZV reactivation, there was a significant increase in the incidence of HHV-6 reactivation. HHV-6 viremia was observed in 24 of 28 (86%) patients during the first month after dUCBT. Six of these 24 (25%) patients developed HHV-6-related encephalitis. There was a correlation between development of encephalitis and HHV-6 viral load ≥50.000 copies/ml (p=0.007). Pre-transplant TREC levels ≥1.200 copies/ml negatively correlated with subsequent HHV-6 reactivation (p=0.04), indicating that baseline reserve of thymic function has a significant role in post-transplant immune reconstitution. On days 30, 60 and 100 post-transplant, higher TREC levels correlated with lack of HHV-6 viremia (p<0.04). A tendency of higher CD4+ counts in the first 100 days was also observed in patients without HHV-6 reactivation. Neutrophil and platelet engraftment, reconstitution of CD4+ and CD8+ T effectors, NK cells and monocytes, IL-6, IL-8 and TNF-α levels, and development of GVHD did not correlate with HHV-6 reactivation or its absence. Our results indicate that substitution of low dose TBI for ATG in the conditioning regimen is characterized by superior recovery of thymopoiesis and reconstitution of CD4+ T cells, both of which have a protective role against HHV-6 reactivation and end organ disease. Further studies will identify why HHV-6 reactivation is selectively increased in UCBT recipients treated with TBI-containing conditioning.
Defilipp:Incyte: Research Funding. Politikos:Angiocrine Bioscience Inc: Research Funding. Armand:Otsuka: Research Funding; Roche: Research Funding; Affimed: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Adaptive: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Bristol-Myers Squibb: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Merck: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau; Infinity: Consultancy; Sigma Tau: Research Funding; ADC Therapeutics: Consultancy; Tensha: Research Funding; Genentech: Research Funding; Pfizer: Consultancy. Ho:Jazz Pharmaceuticals: Consultancy. Koreth:Amgen: Consultancy; Equillium: Consultancy; Cugene: Consultancy. Avigan:Celgene: Membership on an entity's Board of Directors or advisory committees, Research Funding; Pharmacyclics: Research Funding; Juno: Membership on an entity's Board of Directors or advisory committees; Partners Tx: Membership on an entity's Board of Directors or advisory committees; Partner Tx: Membership on an entity's Board of Directors or advisory committees; Karyopharm: Membership on an entity's Board of Directors or advisory committees; Bristol-Myers Squibb: Membership on an entity's Board of Directors or advisory committees; Janssen: Consultancy; Parexel: Consultancy; Takeda: Consultancy. Rosenblatt:Celgene: Research Funding; Amgen: Other: Advisory Board; Merck: Other: Advisory Board; BMS: Other: Advisory Board ; Parexel: Consultancy; Imaging Endpoint: Consultancy; Partner Tx: Other: Advisory Board; Dava Oncology: Other: Education; BMS: Research Funding. Chen:Abbvie: Consultancy; Incyte: Consultancy; Magenta: Consultancy; Takeda: Consultancy; Kiadis: Consultancy. Soiffer:Gilead, Mana therapeutic, Cugene, Jazz: Consultancy; Juno, kiadis: Membership on an entity's Board of Directors or advisory committees, Other: DSMB; Mana therapeutic: Consultancy; Kiadis: Other: supervisory board; Jazz: Consultancy; Cugene: Consultancy. Cutler:Kadmon: Consultancy; Incyte: Consultancy; Pharmacyclics: Consultancy; Fate Therapeutics: Consultancy. Ritz:Equillium: Research Funding; Merck: Research Funding; Kite Pharma: Research Funding; Aleta Biotherapeutics: Consultancy; Celgene: Consultancy; Avrobio: Consultancy; LifeVault Bio: Consultancy; Draper Labs: Consultancy; Talaris Therapeutics: Consultancy; TScan Therapeutics: Consultancy.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal