In the bustle of everyday life, taking a break allows one to regain composure and focus, improving the chances of successfully completing a challenging task. Apparently, this is not so different in modern cancer immunotherapies. In this issue of Blood, Philipp et al1 show for the first time that continuous engagement of T cells by bispecific antibodies (BsAbs) eventually leads to their exhaustion, whereas interjecting some treatment-free intervals (TFIs) in the therapeutic schedule helps preserve the fitness of immune effectors, thus improving long-term results (see figure).
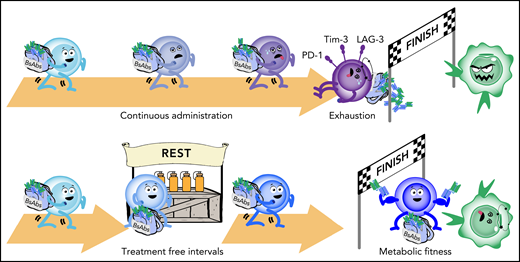
TFIs improve the therapeutic efficacy of T-cell engagers. The sketch summarizes the main findings from the Zieger et al study. (Top) Continuous exposure of T cells (represented by the blue character) to BsAbs (in the backpack) leads to progressive impairment of their function, exhaustion, and ultimately, to their inability to kill tumor cells. (Bottom) Implementation of TFIs in the therapeutic program allows preservation of function, improves metabolic fitness, and overall favors long-term disease control. Professional illustration by Somersault18:24.
TFIs improve the therapeutic efficacy of T-cell engagers. The sketch summarizes the main findings from the Zieger et al study. (Top) Continuous exposure of T cells (represented by the blue character) to BsAbs (in the backpack) leads to progressive impairment of their function, exhaustion, and ultimately, to their inability to kill tumor cells. (Bottom) Implementation of TFIs in the therapeutic program allows preservation of function, improves metabolic fitness, and overall favors long-term disease control. Professional illustration by Somersault18:24.
The authors observed that in patients with B-cell acute lymphoblastic leukemia (B-ALL) who were treated with the CD19×CD3 BsAb blinatumomab (commonly administered by 28-day continuous intravenous infusion cycles), T-cell function progressively declined during the course of treatment and recovered to some extent upon its cessation.
The authors then developed an in vitro system to reproduce and model their observations, showing that regardless of which BsAb and target cell they used, continuous engagement of T cells through a BsAb leads to upregulation of the exhaustion markers programmed cell death protein 1 (PD-1), T-cell immunoglobulin and mucin domain 3 (Tim-3), and lymphocyte activation gene 3 (LAG-3), together with their lead transcriptional regulator TOX, which ultimately results in loss of function. Then they showed that including some TFIs in their model resulted in better preservation of the T-cell cytokine profile and cytotoxicity because of the induction of beneficial rewiring of the T-cell metabolism (increasing glycolysis and oxidative phosphorylation [OXPHOS]) and transcriptional profile (promoting a TCF7-driven program). Finally, the authors confirmed the relevance of their findings in vivo by showing that T cells exposed to BsAbs with a TFI-based regimen outperform their standard continuously engaged counterparts, thereby conveying superior T-cell persistence, phenotype, and killing of B-ALL xenografts.
The findings from Philipp et al parallel and corroborate recent observations made by Mestermann et al2 and Weber et al3,4 in the setting of chimeric antigen receptor (CAR) T-cell therapy. The researchers showed that exposure of the gene-modified cells to the tyrosine kinase inhibitor dasatinib (also tested in the Philipp et al study) can temporarily and reversibly switch off their functionality. This transient rest period promotes the acquisition of a memory-like phenotype, promotes global transcriptional and epigenetic reprogramming, and also restores the antitumor functionality of the CAR T cells.
Over the last few years, incorporating BsAbs and genetically engineered T cells into our therapeutic armamentarium has revolutionized cancer therapy and has persuaded the medical community that immunotherapies have tremendous potential based on unequivocal efficacy data.5,6 Still, the exceedingly rapid transition to practice of these conceptually new “living drugs” has led the community to underappreciate that these drugs also obey the general principles of pharmacology, including biodistribution, pharmacokinetics, and pharmacodynamics, but with an exponentially higher complexity than any chemical compound. The intrinsic ability of cells to react to the external environment and modify their features over time is unique and offers tremendous potential but also needs to be studied with sufficient depth to be fully exploited.
Studies like the one from Philipp et al provide a remarkable example of a roadmap for future improvement in the field of BsAbs; the roadmap will help researchers make relevant observations from clinical follow-up of patients and develop new cutting-edge systems to model and dissect the underlying biology of their treatments. This latter point is a critical need for predicting the toxicities and efficacy of BsAbs, and it will help balance the need for reductionism with the necessity of recapitulating the complex dynamics and cellular networks that are characteristic of immune responses.7,8
Overall, the clinical implications of the study by Philipp et al are straightforward, and they promote the notion of conducting clinical trials to compare the current schedule of administration of BsAbs to a schedule that alternates infusions of the drug with TFIs. Such studies should test the effect of this change of protocol in term of disease control, and should also include biological monitoring to corroborate the findings of the Zeiger et al study and promote further optimization of BsAbs.
Conflict-of-interest disclosure: L.V. is a consultant for Moderna Therapeutics, Novartis, Chroma Medicine, and Gen-Dx, has received research funding from Moderna Therapeutics and Gen-Dx, and receives royalties from Gen-Dx. C.T. declares no competing financial interests.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal