In this issue of Blood, Ho et al1 report that inhibition of mouse double minute 2 (MDM2) increases the potency of T-cell responses to acute myeloid leukemia (AML) and facilitates the graft-versus-leukemia (GVL) effect in murine models of allogeneic hematopoietic cell transplantation(allo-HCT).
HCT reduces the risk of leukemic relapse and is curative for many patients with AML, partly because of an immunologic GVL effect mediated by donor T cells that recognize minor and major histocompatibility complex (MHC) antigens in MHC-matched and MHC-mismatched HCT, respectively.2 Unfortunately, about one-third of HCT recipients with AML relapse after HCT, and most patients with relapse after HCT die of their disease. There is a clear need to improve our understanding of the mechanisms of AML resistance to immune responses and to develop novel countermeasures in the context of HCT and targeted T-cell therapies. The article by Ho et al addresses this area of significant unmet medical need.
Several mechanisms of AML escape from allogeneic T-cell responses have been defined, including downregulation of HLA class II molecules in leukemia, secretion of lactic acid, and resistance to apoptosis.3 Ho et al report a new strategy for countering AML immune evasion by inhibiting MDM2. The MDM2 protein functions as a ubiquitin ligase that recognizes the N-terminal transactivation domain of the proapoptotic tumor suppressor p53. MDM2 inhibits p53 transcriptional activation and facilitates proteasomal p53 degradation and export from the cell, which reduces intracellular p53. Because p53 upregulates the expression of some immune-related genes, including type I interferons,4 and increases MHC expression5 and peptide presentation on MHC class I molecules,6 the authors hypothesized that MDM2 inhibitors could also reverse AML immune evasion after HCT.
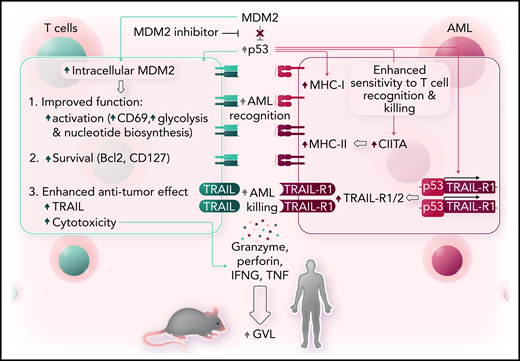
In a comprehensive series of experiments that used human leukemia cell lines, primary AML cells in vitro and in xenografts, and murine cell lines in vitro and in vivo, the authors showed that inhibition of MDM2 affects both AML and T cells to favor effective anti-leukemic immune responses (see figure). Specifically, MDM2 inhibition induced MHC class I and II expression in murine and human AML cells and led to increased tumor necrosis factor–related apoptosis-inducing ligand receptor 1 and receptor 2 (TRAIL-R1/2) expression on leukemia cells, making the leukemia cells easier for T cells to recognize and also making them more susceptible to T-cell–mediated cytotoxicity. The effects of MDM2 inhibition on MHC molecules and TRAIL-R expression were confirmed to be p53 dependent and could also be induced by inhibition of MDMX.
MDM2 inhibition enhances GVL effects. Left, inhibition of MDM2 increases intracellular MDM2 and p53 in T cells, which leads to phenotypic and metabolic changes consistent with more activation and less inhibition, along with improved survival and enhanced cytotoxicity. Right, in AML, increased p53 leads to increased expression of MHC class I and class II molecules along with increased expression of TRAIL-R1/2, which together render the leukemia cells more susceptible to recognition and killing by T cells. Professional illustration by Somersault18:24.
MDM2 inhibition enhances GVL effects. Left, inhibition of MDM2 increases intracellular MDM2 and p53 in T cells, which leads to phenotypic and metabolic changes consistent with more activation and less inhibition, along with improved survival and enhanced cytotoxicity. Right, in AML, increased p53 leads to increased expression of MHC class I and class II molecules along with increased expression of TRAIL-R1/2, which together render the leukemia cells more susceptible to recognition and killing by T cells. Professional illustration by Somersault18:24.
Inhibition of MDM2 demonstrated beneficial effects on T cells and leukemia cells. Inhibition of MDM2 led to increased frequencies of CD8+CD27lowPD-1lowTIM-3low T cells with features of cytotoxicity (perforin+CD107a+TRAIL+) and longevity (Bcl-2+IL-7R+) and enhanced glycolytic activity. The mechanism of action of MDM2 on T cells was not evaluated in this study, but previously published data indicate that inhibition of MDM2 can increase intracellular levels of MDM2 by a feedback mechanism which, together with p53, can stabilize STAT5 in T cells.7 The combined effects of MDM2 inhibition on AML and allogeneic T cells resulted in enhanced cytotoxicity against leukemia cells and improved survival in xenografts of human AML and mouse models of leukemia. Although lactic acid produced by AML has previously been shown to inhibit T-cell function,8 inhibition of MDM2 improved survival of mice with leukemia who received T cells that had been exposed to lactic acid.
MDM2 inhibitors are already in clinical trials for AML and solid tumors. The research by Ho et al provides a clear rationale for using clinical trials to evaluate MDM2 inhibitors in HCT recipients. In fact, there are plans to explore the treatment concept in a phase 1/2 proof-of-principle trial (siremadlin [HDM201] after allo-HCT to prevent AML relapse) in a Novartis-sponsored study (CHDM201K12201). One unknown that will be addressed by clinical trials using this class of drugs in the post-HCT setting is whether the cytopenias and gastrointestinal toxicity frequently observed in previous trials of MDM2 inhibitors in other contexts might hinder their use in HCT recipients. More broadly, the finding that MDM2 inhibition increases AML susceptibility to T-cell cytotoxicity could indicate that this strategy might enhance the efficacy of anti-leukemic T-cell responses outside of HCT and could be evaluated in the future in the context of targeted T-cell therapies, such as chimeric antigen receptor T cells.
Conflict-of-interest disclosure: The authors declare no competing financial interests.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal