Abstract
Background:BCR::ABL1 mutations are key contributing factors to tyrosine kinase inhibitor (TKI) resistance in patients with chronic myeloid leukemia (CP-CML). Ponatinib is a third-generation BCR::ABL1 tyrosine kinase inhibitor (TKI) designed to potently inhibit BCR::ABL1 with or without any single resistance mutation, including T315I. The Phase 2 OPTIC trial evaluated benefit/risk outcomes of ponatinib using a novel response-based dosing strategy in patients with heavily pretreated resistant CP-CML. In the OPTIC trial, all 3 dosing regimens demonstrated benefit in resistant patients, with optimal benefit/risk outcomes observed in patients who received the response-based dosing of 45 → 15 mg/day. Here we present a post hoc analysis of long-term outcomes (progression-free survival [PFS] and overall survival [OS]) of patients in the OPTIC trial who reached landmark clinical responses (≤1%, >1%-≤10%, and >10% BCR::ABL1IS by Months 3, 6, and 12).
Methods: The OPTIC trial design has been previously described (Cortes J, et al. Blood. 2021;138:2042-50). Overall, 99% of patients had treatment-resistant disease; 61% of patients had a best response to their last prior therapy of complete hematologic response or worse; and 84% had a best response of >10% BCR::ABL1IS to prior therapy. This landmark analysis describes PFS and OS in patients with ≤1%, >1%-≤10%, and >10% BCR::ABLIS by Months 3, 6, and 12. Disease progression was defined as the loss of complete hematologic response or major cytogenetic response, development of acute- or blast-phase CML, death, or 2 instances of white blood cell count increases (>20,000 cells/μL, ≥4 weeks apart) after the first 4 weeks of treatment. Patients with ≤1% BCR::ABL1IS at baseline were excluded from this analysis. The probabilities of PFS and OS at 24 months were calculated using the Kaplan-Meier method. The proportion of patients attaining ≤1% BCR::ABLIS by 36 months was also assessed.
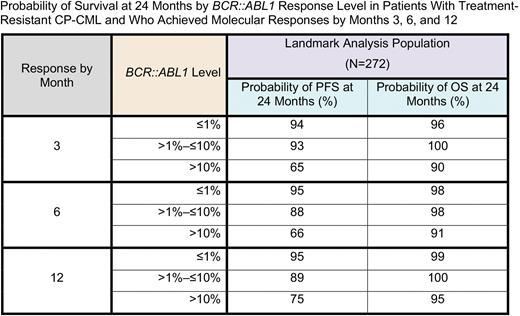
Results: A total of 276 patients with resistant CP-CML were included in the intention-to-treat (ITT) population: 45-mg (n=93), 30-mg (n=93), and 15-mg (n=90) starting dose cohorts. At the cutoff date of May 9, 2022, 56 (20%), 78 (28%), and 95 patients (34%) had reached ≤1% BCR::ABL1IS by Months 3, 6, and 12, respectively. Among patients remaining within each dosing cohort at 36 months, most had attained ≤1% BCR::ABLIS (57%-65%). Among all ITT patients within each dosing cohort (45 mg, n=93; 30 mg, n=93; 15 mg, n=90), the highest response rate by 36 months was observed in the 45-mg cohort (35%). Four patients with ≤1% BCR::ABL1IS at baseline were excluded from the landmark analysis. Based on this landmark analysis, regardless of starting dose, patients who attained landmark response levels of ≤1% and >1-≤10% BCR::ABL1IS in 3-12 months had substantially improved PFS outcomes at 24 months compared with patients with >10% BCR::ABL1IS (Table). In the landmark analysis population, patients who did not reach ≤10% BCR::ABL1IS within 12 months had unfavorable PFS outcomes (Table). Long-term OS outcomes in the landmark analysis population were similar across all BCR::ABL1IS levels, regardless of whether response was reached by 3, 6, or 12 months (Table).
Conclusion: Ponatinib provided robust responses in the OPTIC trial population comprising patients with heavily pretreated, TKI-resistant CP-CML. In this landmark analysis, attainment of ≤10% BCR::ABL1IS with ponatinib within 12 months was associated with improved long-term PFS outcomes compared with PFS outcomes associated with remaining at >10% BCR::ABL1IS. These findings underpin the clinical benefit of achieving molecular responses with ponatinib, especially achieving ≤10% BCR::ABL IS within 12 months.
Disclosures
Apperley:Novartis: Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Incyte: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau; Pfizer: Honoraria, Research Funding, Speakers Bureau. Cortes:Sun Pharma: Consultancy, Research Funding; Takeda: Consultancy, Honoraria; Gilead: Consultancy; Forma Therapeutic: Consultancy; Biopath Holdings Inc: Consultancy, Current equity holder in private company; Abbvie: Consultancy, Research Funding; Pfizer: Consultancy, Honoraria, Research Funding; Novartis: Consultancy, Honoraria, Research Funding. Jabbour:Amgen: Other: Advisory Role, Research Funding; Adaptive Biotechnologies: Other: Advisory Role, Research Funding; Pfizer: Other: Advisory Role, Research Funding; Takeda: Other: Advisory Role, Research Funding; AbbVie: Other: Advisory Role, Research Funding; Genentech: Other: Advisory Role, Research Funding; Spectrum: Research Funding; Bristol Myers Squibb: Other: Advisory Role, Research Funding. Hochhaus:Pfizer: Research Funding; Novartis: Research Funding; Bristol Myers Squibb: Research Funding; Incyte: Research Funding. Hughes:BMS: Consultancy, Research Funding; Novartis: Consultancy, Research Funding; Enliven: Consultancy, Research Funding. Chuah:Novartis: Honoraria; Pfizer: Other: Travel, Research Funding; Bristol Myers Squibb: Honoraria, Research Funding; Steward Cross: Korea Otsuka International Asia Arab: Honoraria; Otsuka [Philippines] Pharmaceutical: Honoraria; Korea Otsuka Pharmaceutical: Honoraria. de Lavallade:Novartis: Honoraria; Pfizer: Honoraria; Incyte: Honoraria, Research Funding; Bristol Myers Squibb: Honoraria, Research Funding. Deininger:Galena Biopharma: Consultancy, Honoraria; Pfizer Inc: Consultancy, Honoraria, Research Funding; Bristol Myers Squibb: Consultancy, Honoraria, Research Funding; Incyte: Consultancy, Honoraria, Research Funding; Novartis: Consultancy, Honoraria, Research Funding; Blueprint Medicines Corporation: Consultancy, Honoraria, Research Funding. Lipton:Bristol Myers Squibb: Consultancy, Research Funding; ARIAD Pharmaceuticals: Consultancy, Research Funding; Pfizer: Consultancy, Research Funding; Novartis: Consultancy, Research Funding. Lomaia:Fusion Pharma: Speakers Bureau; Bristol Myers Squibb: Other: Travel, Accommodation, Expenses ; Pfizer: Other: Travel, Accommodation, Expenses , Speakers Bureau; Novartis: Other: Travel, Accommodation, Expenses , Speakers Bureau. Mauro:Sun Pharma/SPARC: Research Funding; AbbVie, Bristol Myers Squibb, Novartis, Pfizer, Takeda: Consultancy, Honoraria, Other: Travel, accommodation, expenses , Research Funding. McCloskey:AbbVie, Amgen, Bristol Myers Squibb, Incyte, Jazz Pharmaceuticals, Stemline, and Takeda: Speakers Bureau; AbbVie, CTI BioPharma, and Novartis: Consultancy, Membership on an entity's Board of Directors or advisory committees. Moiraghi:Takeda: Speakers Bureau; Pfizer: Speakers Bureau; Novartis: Speakers Bureau. Pavlovsky:Novartis, Pfizer: Membership on an entity's Board of Directors or advisory committees; Novartis, Pfizer, BMS, Pint Pharma: Speakers Bureau. Rojas:Janssen: Other: Personal fees; Novartis: Other: Personal fees; Roche: Other: Personal fees; AstraZeneca: Other: Personal fees. Rousselot:Incyte: Consultancy, Research Funding; Pfizer: Consultancy, Research Funding; Bristol Myers Squibb: Consultancy; Novartis: Consultancy; Takeda: Consultancy. Sacha:Incyte: Consultancy, Honoraria, Speakers Bureau; Pfizer: Consultancy, Honoraria, Speakers Bureau; Novartis: Consultancy, Honoraria, Speakers Bureau; Bristol Myers Squibb: Consultancy, Honoraria, Speakers Bureau; AOP Orphan: Honoraria, Speakers Bureau; Adamed: Consultancy, Honoraria; Angelini: Honoraria, Membership on an entity's Board of Directors or advisory committees; BMs-Celgene: Honoraria, Speakers Bureau. Talpaz:Novartis: Consultancy, Other: Grant/research support ; BMS: Consultancy; IMAGO: Consultancy; Kirin: Membership on an entity's Board of Directors or advisory committees; SDp: Membership on an entity's Board of Directors or advisory committees; Takeda: Other: Grant/research support . Turkina:Novartis: Other: Travel, Accommodation, Expenses , Speakers Bureau; Pfizer: Other: Travel, Accommodation, Expenses , Speakers Bureau; Fusion Pharma: Speakers Bureau. Sutton:Pfizer: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Roche: Membership on an entity's Board of Directors or advisory committees; Novartis: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; AbbVie: Membership on an entity's Board of Directors or advisory committees; Janssen: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau. Ren:Takeda: Current Employment. Vorog:Takeda: Current Employment. Rosti:Novartis: Membership on an entity's Board of Directors or advisory committees, Other: Steering Committee , Speakers Bureau; Pfizer: Membership on an entity's Board of Directors or advisory committees, Other: Steering Committee , Speakers Bureau; Incyte: Membership on an entity's Board of Directors or advisory committees, Other: Steering Committee , Speakers Bureau; BMD: Speakers Bureau.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal