In this issue of Blood, Hurwitz et al report that the sphingolipid enzyme neutral sphingomyelinase-2 (nSMase2) regulates hematopoietic stem cell (HSC) fitness.1 Sphingolipids are amphipathic lipids that include both structural species, like sphingomyelin (SM), as well as bioactive lipids, like sphingosine-1-phosphate (S1P), that all contain a sphingoid base backbone with fatty acid chains of various length.2 The interest and increased understanding of these enigmatic lipids, so named because of their “sphinx”-like complexity in structure, in normal hematopoiesis has recently gained traction. Mechanistically, this study shows that transient inhibition of hematopoietic stem and progenitor cells (HSPCs) with the nSMase2 inhibitor GW4869 during ex vivo culture activates an activating transcription factor 4-mediated integrated stress response3 (ISR) to improve repopulation in competitive transplantation assays1 (see figure).
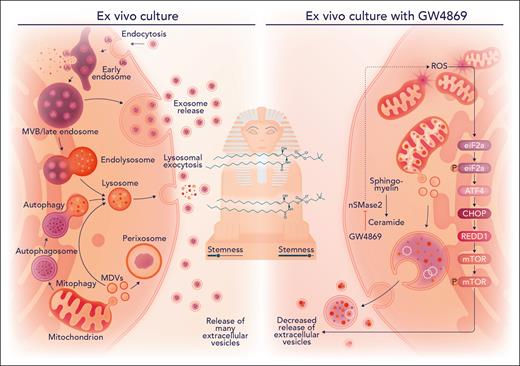
Transient inhibition of the sphingolipid enzyme nSMase2 with GW4869 safeguards hematopoietic stem cell fitness in ex vivo culture. Mitogenic activation from ex vivo culture conditions promotes release of various extracellular vesicles and a decrease in stemness. On nSMase2 blockade with GW4869 in hematopoietic stem and progenitor cells, mitochondrial stress occurs and an activating transcription factor 4-mediated integrated stress response is activated to decrease release of extracellular vesicles that results in enhanced stemness, as measured in competitive transplantation assays. CHOP, CCAAT/enhancer-binding protein homologous protein; eiF2a, eukaryotic translation initiation factor 2A; mTOR, mammalian target of rapamycin; MVB, multivesicular bodies; REDD1, regulated in development and DNA damage response 1, also known as DNA-damage-inducible transcript 4 (DDIT4); ROS, reactive oxygen species. Professional illustration by Somersault18:24.
Transient inhibition of the sphingolipid enzyme nSMase2 with GW4869 safeguards hematopoietic stem cell fitness in ex vivo culture. Mitogenic activation from ex vivo culture conditions promotes release of various extracellular vesicles and a decrease in stemness. On nSMase2 blockade with GW4869 in hematopoietic stem and progenitor cells, mitochondrial stress occurs and an activating transcription factor 4-mediated integrated stress response is activated to decrease release of extracellular vesicles that results in enhanced stemness, as measured in competitive transplantation assays. CHOP, CCAAT/enhancer-binding protein homologous protein; eiF2a, eukaryotic translation initiation factor 2A; mTOR, mammalian target of rapamycin; MVB, multivesicular bodies; REDD1, regulated in development and DNA damage response 1, also known as DNA-damage-inducible transcript 4 (DDIT4); ROS, reactive oxygen species. Professional illustration by Somersault18:24.
HSCs are responsible for replenishing every blood cell lineage, and their use in HSC transplantation has proven to be a highly successful regenerative medicine (>50 000 allotransplants/year globally).4 The linkage between modulation of sphingolipid metabolism and proteostatic quality control programs to preserve HSC function during ex vivo culture is concordant with findings following pharmacologic inhibition of the de novo sphingolipid enzyme dihydroceramide desaturase 1 in human cord blood (CB).4 GW4869 blocks the generation and secretion of exosomes, a subpopulation of extracellular vesicles (EVs).5 Impairment of endolysomal trafficking6 and EV release with activation of the prosurvival ISR pathway altogether promotes metabolic quiescence in both mouse and human HSPCs, thereby safeguarding self-renewal.1 Thus, the growing body of evidence for sphingolipid metabolism as an HSPC regulatory axis that can be exploited for regenerative medicine points to both cell-autonomous and non–cell-autonomous mechanisms.
Metabolic reprogramming is vital to HSC self-renewal and differentiation fate decisions, especially in the transition from quiescence to cellular activation.4,6 Modulating energy and lipid metabolism directly or indirectly during ex vivo culture (eg, glycolysis, mitochondrial function, and eicosanoid and sphingolipid metabolism) improves CB engraftment and/or self-renewal in vivo.4,7 In addition to intracellular metabolism, the signaling lipid S1P also regulates HSC function.2 A sphingolipid signaling gene set was shown to be enriched in a molecular signature of ex vivo expanded murine HSCs,8 and loss of sphingosine kinase 2 was found to promote the metabolic fitness of mouse HSCs through an Sphk2-hypoxia inducible factor 1 subunit alpha-pyruvate dehydrogenase kinase 3 axis.9 GW4869 treatment led to elevation of mitochondria-associated reactive oxygen species and increases in mitochondrial mass in both human and murine HSPCs.1 The supposition is that such metabolic stress activated the ISR to alter EV release. A more formal proof to ask if inhibition of the ISR with integrated stress response inhibitor would be sufficient to restore EV release and block the competitive repopulation advantage of GW4869-treated HSPCs in vivo would be of interest in future studies. Altogether, rewiring sphingolipid metabolism and signaling may engage metabolic and proteostatic stress response programs to impact cell-cell communication via EVs that safeguard HSC fitness (see figure).
Human HSCs and committed progenitor populations have differential expression of lipid metabolism genes, especially those in the sphingolipid pathway, including SMPD3.4SMPD3, which encodes nSMase2, is transcriptionally enriched in human committed progenitors relative to HSCs,4 suggesting a possible regulatory role in HSC self-renewal/lineage fate decisions. Hurwitz et al showed SMPD3-knockout (KO) murine HSPCs had competitive repopulation advantage relative to control-KO HSPCs, and GW4869-treated human HSPCs engrafted better than control-treated HPSCs in xenotransplantation assays.1 Notably, quantitative mass spectrometry on EVs isolated from human SMPD3-KO HSPCs compared with control HSPCs showed a depletion of key proteins associated with myeloid lineage transcription and signaling factors, such as PU.1 and colony-stimulating factor 1, as well as mitochondrial protein and DNA cargo.1 These data point directly to the importance of nSMase2 in HSPC fate decisions, but also raise the question of whether it is directly attributed to its sphingomyelinase activity and blocking of hydrolyzation of SM to ceramide (a precursor to sphingosine) and phosphocholine to activate a “lipid biostat.”4 Interestingly, the authors did not find overall global depletion of ceramide nor SM accumulation with GW4869 treatment.1 However, lipid quantitation by mass spectrometry was performed on a bulk murine HSPC subpopulation expanded for 14 days. The authors noted that ceramide/SM ratios for some specific long-chain acyl species were perturbed. Previous lipidomic studies have shown that some sphingolipid species are differentially enriched in HSC-enriched cells relative to committed progenitors in both the human and mouse settings at steady state.2 Thus, future low-input lipidomic studies on highly refined stem and progenitor subsets should further shed light on the complexity of sphingolipid metabolism and its functional role in HSC vs committed progenitor subsets.
Finally, drug resistance in leukemia stem cells is a perplexing issue that has been linked to proteostatic stress response programs and mitochondrial metabolism/metabolic reprogramming, including of sphingolipids.2 Thus, future studies integrating the findings from this study on HSC fitness to cancer could have major translational implications for proteostasis programs, EV secretion, and metabolic reprogramming to disrupt drug resistance in hematological malignancies and solid tumors.2,10
Conflict-of-interest disclosure: The author declares no competing financial interests.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal