Myelodysplastic syndromes (MDS) are incurable neoplasms that arise from early hematopoietic stem cells (HSCs). The standard of care for MDS is hypomethylating agent (HMA) therapy. However, the disease invariably fails these agents, resulting in fatal cytopenias or progression to acute myeloid leukemia (AML) with a dismal prognosis.
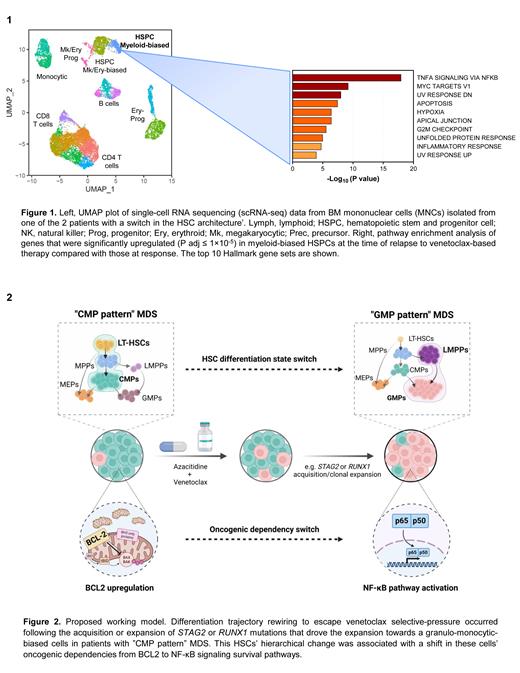
We previously showed that the cellular hierarchy of MDS HSCs predicts the mechanisms of progression after HMA failure and can guide the design or choice of specific therapeutic approaches targeting these cells (Ganan-Gomez et al., Nature Medicine 2022). MDS HSCs in one of two differentiation states, long-term HSCs or lymphoid-primed multipotent progenitors, have a “common myeloid progenitor (CMP) pattern” or “granulocytic-monocytic progenitor (GMP) pattern” of differentiation. HSCs in these 2 differentiation states persist throughout the disease course and, driven by the recurrent activation of BCL2- or NF-κB-mediated survival pathways, respectively, expand at disease progression. In our preclinical studies, pharmacologically inhibiting these pathways depleted MDS HSCs and reduced tumor burden, which suggests that only patients with “CMP pattern” MDS may benefit from venetoclax-based therapies. Accordingly, patients with “CMP pattern” MDS, whose disease progressed after frontline HMA failure, enrolled in clinical trials of venetoclax-based therapy (NCT04160052, NCT04550442, or NCT04655755; n=28: 12 “CMP pattern” MDS and 16 “GMP pattern” MDS) had a shorter cumulative time to complete remission (P=0.012) and a longer relapse-free survival duration (14.0 months vs 5.2 months) than those with “GMP pattern” MDS did. Consistent with the hypothesis that venetoclax-based therapy selectively targets different HSC populations after HMA therapy failure, bone marrow (BM) HSC frequencies were significantly decreased at the time of remission after venetoclax-based therapy in “CMP pattern” MDS patients, but not in those with “GMP pattern” MDS. These results explain why “GMP pattern” MDS are resistant to venetoclax therapy.
However, 8 of the 12 patients with “CMP pattern” MDS relapsed during venetoclax treatment and/or progressed to AML after an initial remission. In 2 patients with “CMP pattern” MDS, sequential flow cytometry and next generation sequencing analyses showed that the HSC hierarchy switched to “GMP pattern” MDS before venetoclax failure. This immunophenotypic hierarchical change was associated with the detection or expansion of STAG2- or RUNX1-mutant clones with isolated trisomy 8.
To evaluate whether the immunophenotypic switch resulted from the distinct differentiation potential of early HSCs or from the aberrant expression of cell surface markers, we performed sequential single-cell RNA sequencing analyses of BM mononuclear cells isolated from the 2 “CMP pattern” MDS patients whose disease changed hierarchy before venetoclax failure. Our analyses revealed that after undergoing significant depletion during disease remission, HSCs expanded at therapy failure. Differential expression analysis showed that the expansion of STAG2- or RUNX1-mutant clones in the HSC compartment not only rewired MDS HSCs' differentiation state towards a granulo-monocytic-biased trajectory but also changed HSCs' survival dependency from BCL2-mediated anti-apoptotic pathways to TNFa-induced pro-survival NF-κB signaling, thus enabling HSCs to evade the cytotoxic effects of venetoclax (Fig. 1). The anti-apoptotic gene MCL1, a direct transcriptional target of NF-κB, was also significantly upregulated in HSCs at the time of progression, which suggests that the combination of venetoclax with an MCL1 inhibitor can overcome secondary failure to venetoclax.
Together, these results reveal a novel mechanism of venetoclax resistance. HSCs under venetoclax therapy undergo survival pressure, which results in the expansion or acquisition of clones carrying specific mutations that change HSCs' survival dependencies (Fig. 2). Our study suggests that MDS patients receiving venetoclax-based therapy should be monitored at the molecular level and enrolled in clinical trials of other targeted agents before their disease undergoes clonal evolution and HSC hierarchical rewiring.
Disclosures
Ma:Amgen: Current Employment. Chien:Rigel Pharmaceuticals: Consultancy; AbbVie: Consultancy. Montalban-Bravo:Takeda: Research Funding; Rigel: Research Funding. Loghavi:Daiichi Sankyo: Consultancy; Recordati/ EUSA Pharma: Consultancy; Caris Diagnostics: Consultancy; Blueprint Medicine: Consultancy; Abbvie: Consultancy; Gerson Lehrman Group: Consultancy; QualWorld: Consultancy; Guidepoint: Consultancy; Astellas: Research Funding; Amgen: Research Funding; Abbvie: Current equity holder in publicly-traded company. Garcia-Manero:Genentech: Research Funding; Bristol Myers Squibb: Other: Medical writing support, Research Funding; AbbVie: Research Funding.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal