Key Points
In persons with NSHA, inhibitor development occurred at an earlier age than previously reported.
The development of inhibitors in persons with NSHA was not associated with increased mortality.
Abstract
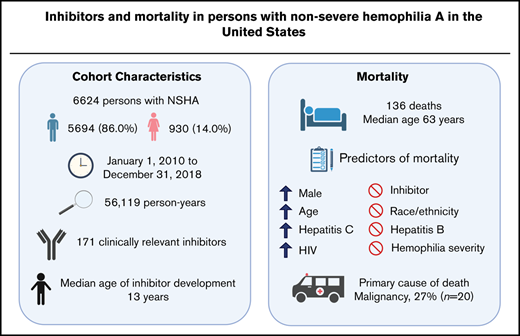
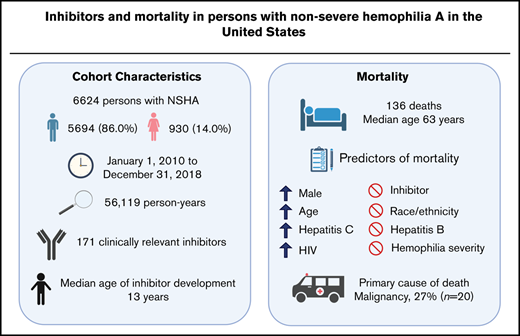
Although persons with nonsevere hemophilia A (NSHA) account for about one-half of the hemophilia A population, epidemiological data in this subset of individuals are scarce. We set out to describe the clinical characteristics of persons with NSHA with inhibitors, and to determine mortality rates, predictors of mortality, and primary causes of death in persons with NSHA in the United States over a 9-year period (2010-2018). We queried the American Thrombosis and Hemostasis Network dataset (ATHNdataset) for information on demographics, inhibitor status, and date and cause of death. A total of 6624 persons with NSHA (86.0% men; 14.0% women) were observed for an average of 8.5 years; total 56 119 person-years . The prevalence of inhibitors was 2.6% (n = 171), occurring at a median age of 13 years. At the end of follow-up, 136 persons died at a median age of 63 years; an age-adjusted mortality rate of 3.3 deaths per 1000 person-years. Three deaths occurred in inhibitor participants. Presence of inhibitors was not associated with increased mortality risk (hazard ratio [HR], 0.7, 95% confidence interval [CI], 0.2-2.3). Factors independently associated with increased risk of death (HR, 95% CI) were the following: age (10-year increase) (2.1, 2.0-2.4); male (2.6, 1.0-6.4); hepatitis C (2.2, 1.5-3.1); and HIV (3.6, 2.2-6.0). The most common primary cause of death was malignancy (n = 27, 20.0%). In persons with NSHA, the development of inhibitors occurred at an early age and was not associated with increased mortality.
Introduction
Hemophilia A is an X-linked congenital bleeding disorder with an estimated prevalence at birth of 23.2 per 100 000 males.1 Since the establishment of US federally funded hemophilia treatment centers (HTCs) in 1975 to provide integrated, specialized hemophilia care, substantial improvements related to mortality have been made within this patient population.2 Despite greatly improved hemophilia care, the most significant treatment-related complication is the development of neutralizing antibodies (inhibitors) to factor VIII (FVIII), which has been associated with increased mortality.3-5 The majority of published studies looking at mortality trends and inhibitor development have focused mainly on patients with severe hemophilia A.6-8 Even though persons with nonsevere hemophilia A (NSHA) account for about one-half of the overall hemophilia A population,9 epidemiological data addressing this subset of individuals are limited. Similarly, data on inhibitors in persons with NSHA, specifically mortality, are scarce and include a single European and Australian cohort: the International Study on Etiology of Inhibitors in Patients with a Moderate or Mild Form of Hemophilia A, Influences of Immuno-Genetic & Hemophilia Treatment Factor (INSIGHT) study.3,4,10,11 This is particularly relevant because the incidence of inhibitors in persons with NSHA is increasing.10,12
The INSIGHT study consisted of a predominantly White population (94.3%), with only 1.3% Black participants.10 In contrast, the US hemophilia population is more diverse, with 80.1% White, 11.6% Black, and 2.7% Asian.13 Given that the prevalence of FVIII inhibitors in Blacks is about twice that in Whites in the severe hemophilia A population,14 it is unclear if the results of the INSIGHT study are generalizable to the US hemophilia population. Thus, the aims of this study were twofold. The first was to describe the clinical characteristics of persons with NSHA with inhibitors, and the second was to determine mortality rates, predictors of mortality, and primary causes of death in persons with NSHA in the United States over a 9-year period (1 January 2010 to 31 December 2018) using the American Thrombosis and Hemostasis Network (ATHN) dataset (ATHNdataset).
Methods
Subjects and study design
ATHN maintains the ATHNdataset, a database consisting of a “limited dataset” as defined under the Health Insurance Portability and Accountability Act to be free of protected health information, with data collection by clinical teams and data managers from 135 HTCs within the 8 Human Resources and Services Administration-supported regional hemophilia networks across the United States.15 A limited dataset allows the following health information to remain in the ATHNdataset: dates such as admission, discharge, service, birth, death; city, state, 5 digit or more ZIP code; and age in years, months, days, or hours.
The ATHNdataset includes persons with congenital bleeding disorders in the United States who have authorized the sharing of their demographic and clinical information for research. The ATHNdataset started enrolling alive persons with congenital bleeding disorders on 1 January 2010. As the data obtained from the ATHNdataset for this study were deidentified with no means to identify individual participants, the definition of a human subject was not met. This study was thus considered nonhuman subject research by the University of Utah institutional review board.
Data collection and definitions
Persons with NSHA were defined as those with a documented diagnosis of mild (FVIII activity [FVIII:C] >5%-50%) or moderate hemophilia A (FVIII:C 1%-5%) in the ATHNdataset, based on the definition by the Centers for Disease Control and Prevention.16 The ATHNdataset was queried on 31 December 2018, to extract the following information on persons with NSHA: demographics, hemophilia severity, inhibitor status, date of death, and primary cause of death. The primary cause of death was entered into the ATHNdataset by clinical teams at the time of death or during annual mortality reporting. In addition, the following clinical information was extracted: exposure to FVIII concentrate, current or past infection with hepatitis B and/or hepatitis C virus, and presence of HIV infection. For participants with documented exposure to FVIII concentrates, data on the number of exposure days (ED) to FVIII concentrates were not available in the ATHNdataset. For hepatitis B and C status, cases with a diagnosis status in the ATHNdataset of “active” or “history of” were classified as having the condition.
Inhibitor testing was performed at the discretion of each HTC and entered into the ATHNdataset with no central laboratory confirmation. There is no mandatory requirement for inhibitor testing in the ATHNdataset. Data on the number of inhibitor tests performed on each participant were not available. Only participants with a clinically relevant inhibitor were classified to be positive for an inhibitor. A clinically relevant inhibitor was defined as having at least 2 positive Bethesda inhibitor assay titers of >1.0 Bethesda inhibitor assay unit per milliliter (BU/mL) (criterion 1). Participants with at least 2 inhibitor titers >0.6 BU/mL who did not qualify under criterion 1 had to fulfill 1 of the following 3 criteria to be classified as having a clinically relevant inhibitor: a decrease in FVIII plasma level to at least 50% of the baseline level (criterion 2), spontaneous bleeding symptoms (criterion 3), or an annual bleed rate >1 (criterion 4). The annual bleed rate was the number of bleeds a person experienced over the course of 1 year. The definition for a clinically relevant inhibitor was adapted from the INSIGHT study.10 Because data on reduced half-life after FVIII administration of less than 6 hours were not available, we did not use this as a criterion. Instead, we added bleeding presentation as an additional criterion for subjects with inhibitor titers >0.6 BU/mL who did not fulfill criterion 1 because it has been demonstrated that persons with NSHA with an inhibitor have a median annual bleed rate of 1.1.17
Similar to the INSIGHT study,4 all participants who were ever documented to have a clinically relevant inhibitor were classified as “inhibitors” because it was not possible to determine the end of an inhibitor period either through eradication or spontaneous clearance in the ATHNdataset. Participants who did not meet the definition of a clinically relevant inhibitor, had a negative inhibitor history, or had never been tested for FVIII inhibitors were classified as “noninhibitors.”
Statistical analyses
Bivariate associations between demographic and clinical characteristics and inhibitor status were examined using χ2 test or Fisher’s exact test, as appropriate.
The overall mortality rate was calculated as the ratio of the number of deaths to the number of person-years at risk, presented as rates per 1000 person-years. Person-years at risk was calculated for each participant as the time between the start of the observation period (1 January 2010, or date of birth for participants who were born later) and the end of the observation period (death date, termination date from ATHNdataset, or 31 December 2018). Inhibitor person-years at risk for participants with inhibitors was calculated as the time interval from 1 January 2010, if the first positive inhibitor test occurred before 1 January 2010, or from the date of the first positive inhibitor test that occurred during the observation period until the end of the observation period. Inhibitor-related death was attributed to all participants who had an inhibitor history.
A crude mortality rate was calculated for each demographic and clinical characteristic, except for exposure to FVIII because of the high percentage of missing data. The crude mortality rate was expressed as the number of deaths among participants in a category of the characteristic divided by the number of years of follow-up of such participants, presented as rates per 1000 person-years. The mortality risk ratio between participants in different categories of the characteristic was assessed by using the ratio of these mortality rates. The age-adjusted mortality rate was calculated based on direct standardization to the 2000 US Standard Population.18 Methods for calculating the 95% confidence interval (CI) for the age-adjusted mortality rate was based on Curtin and Klein.19
To determine characteristics independently associated with mortality, we used Cox proportional hazards regression. We included the following predictors in the model: age, sex, race, ethnicity, disease severity, hepatitis B, hepatitis C, HIV, and inhibitor status. Because a complete-case analysis was used, participants with unknown status for ethnicity and/or race were excluded from the regression analysis. Right censoring was assumed where NSHA patients who dropped out before 2018 or who survived to the end of the observation period would be considered as censored subjects.
Standardized mortality ratios (SMRs) for selected causes of death were calculated as the ratio between the observed number of deaths in the NSHA cohort and the expected number of deaths based on the age-weights in the 2000 US Standard Population. CIs for the SMRs were calculated based on the Poisson distribution.20
All analyses were performed using SAS version 9.3 (SAS Institute, Cary, NC) software and P < .05 was considered statistically significant.
Results
All participant characteristics
Between 1 January 2010 and 31 December 2018, the ATHNdataset included 6624 persons with NSHA (5694 [86.0%] male and 930 [14.0%] female) who were born between 1920 and 2018 from 135 HTCs. The median number of persons with NSHA per HTC was 35. Between 2010 and 2018, 33 (0.5%) participants withdrew their consent and thus their participation in the ATHNdataset was terminated before completion of the observation period. Persons with NSHA were observed for a total of 56 119 person-years. The median observation period was 9 years and the average was 8.5 years (standard deviation [SD] 1.5).
The demographics and clinical characteristics of the NSHA cohort are shown in Table 1. Overall, the age distribution in the US NSHA cohort skewed toward a younger population with 38.9% between the ages of 0 to 19, as compared with the US general population at 24%.21 The proportion of Black participants in the ATHNdataset was lower at 8.2%, as compared with 11.6% in US hemophilia population.13 Of the 6624 persons with NSHA, 77.3% (n = 5122) had documented exposure to FVIII concentrates, 8.4% (n = 555) had no documented exposure, and information was unknown for the remaining 14.3% (n = 947).
Inhibitor participant characteristics
One hundred and seventy-one participants (2.6%; 95% CI, 2.2-3.0) met the definition of having a clinically relevant inhibitor. Ninety-four (55.0%) of the 171 participants with a clinically relevant inhibitor were identified through criterion 1, 27 (15.8%) using criterion 2, 35 (20.5%) using criterion 3 and 15 (8.8%) using criterion 4. There was no overlap between each criterion. Of the 94 inhibitor participants identified through criterion 1, 87 (93%) were positive on consecutive tests, whereas 7 were classified based on any 2 tests in the 9-year period. There were 114 participants with at least 2 inhibitor titers >0.6 BU/mL who did not qualify under criterion 1 and who did not meet the definition of a clinically relevant inhibitor. The median age for inhibitor development was 13 years (interquartile range, 6-36 years).
The demographic and clinical characteristics of the population by inhibitor status are summarized in Table 2. Four participants with no documented FVIII exposure were classified as having a clinically relevant inhibitor. With the exception of infection with hepatitis B and HIV, all of the studied demographic and clinical characteristics were associated with inhibitors. Inhibitors were more likely among participants who were male, Black, non-Hispanic, and had moderate hemophilia A.
Mortality
At the end of the observation period, there was a total of 136 deaths (131 [96.3%] male; 5 [3.7%] female) in the NSHA population, occurring at a median age of 63 years (interquartile range, 51-75 years). The youngest deceased participant was 11 years and the oldest was 97 years. The overall all-cause mortality rate was 2.4 per 1000 person-years (95% CI, 2.0-2.8). The age-adjusted mortality rate was 3.3 per 1000 person-years (95% CI, 2.7-3.9). Three deaths occurred in inhibitor participants, with the primary causes of death being malignancy, cardiovascular disease, and other. The age-adjusted mortality rates for inhibitor and noninhibitor participants were 2.6 (95% CI, 1.0-4.1) and 3.3 (95% CI, 2.7-3.9) per 1000 person-years, respectively. The age-adjusted mortality ratio between inhibitors and noninhibitors was 0.8 (95% CI, 0.2-2.4).
In the bivariate analysis, crude mortality rates increased considerably after 40 years of age (Table 1). Compared with female participants, male participants had a fourfold risk of death. Additional analysis stratifying crude mortality rate by sex and age strata where at least 1 death occurred was performed (supplemental Table 1). The mortality rate ratio was highest at 6.6 in the 60 to 69 years of age group, but this was not statistically significant because of the small number of deaths in female participants.
Crude mortality rates were similar among racial groups and hemophilia severity. Non-Hispanic participants were twice as likely to die than Hispanic participants. Similar to the age-adjusted morality rate, there was no excess crude mortality risk observed in the inhibitor cohort. The crude mortality rate was 7 times higher for NSHA participants with hepatitis C or HIV infection.
After adjusting for the effects of all of the studied characteristics in a multivariable analysis, NSHA men had 2.6 times the risk of death (Table 1). Mortality risk increased twofold with each additional decade of age. Persons with hepatitis C had 2 times the risk of death and persons with HIV had almost 4 times the risk compared with persons without these conditions.
Causes of death
The most common primary cause of death was malignancy (n = 27, 20.0%) (Table 3). The observed numbers of death from liver disease among the NSHA cohort was almost fivefold the expected death rate. The observed number of deaths caused by malignancy, cardiovascular disease, or accident/trauma was statistically lower than expected (Table 3).
Discussion
In this US cohort of 6624 persons with NSHA, we found that inhibitors occurred at an early age of 13 years with a prevalence of 2.6% and was not associated with an increased risk of mortality. Instead, mortality rates in the US NSHA cohort were influenced by age, male sex, and hepatitis C and HIV infections.
The age at inhibitor development in our cohort is much earlier than the commonly reported median age of about 30 years for inhibitor development in NSHA.8 In addition, the INSIGHT study, an international cohort of 2709 persons with NSHA, reported that inhibitors developed in 107 patients (4%) at a median age of 38 years, 25 years older than our cohort.4 The reason for this age difference is unclear. The earlier age of inhibitor development may be due to the observed time period as the availability of FVIII concentrates have evolved over the years, and may have been used more often from 2010 to 2018 as compared with the INSIGHT cohort (1980-2011). Future longitudinal study with a longer observation period will be required to explore reasons for the age difference in inhibitor development that we observed.
The prevalence of inhibitors in the INSIGHT cohort was higher at 4% vs 2.6% in our cohort.4 We report on a shorter duration of follow up (9 years), whereas the INSIGHT study included participants over a 31-year period (1980-2011). In persons with NSHA, the risk of inhibitor development increases with each ED to FVIII concentrates.10 As such, the short duration of follow-up could explain the lower prevalence of inhibitors in our cohort. Setting aside the differences in duration and time periods, and the lower prevalence and earlier age of inhibitor development in our cohort, the overall all-cause mortality for the INSIGHT cohort was almost identical to our study, 2.3 vs 2.4 per 1000 person-years, with a similar median age of death, 64 years vs 63 years.
We found that the development of inhibitors was not associated with an increased risk of mortality, even when age-adjusted. This is in contrast to the previously reported results of Eckhardt et al, who found that, as part of the INSIGHT study, inhibitor development was associated with a more than fivefold increased risk for all-cause mortality, compared with those without inhibitors.4 The higher mortality rate observed in their cohort could be explained by the older age of 38 years at time of inhibitor development compared with 13 years in our study. Older people are more susceptible to age-related comorbidities and bleeding complications, thus contributing to the increased mortality in the INSIGHT study.
Inhibitor status at time of death was not available in the ATHNdataset. We attributed inhibitor-related death to all participants who had an inhibitor history, irrespective of whether the inhibitor was present at time of death, eradicated, or spontaneously cleared. Given that the 3 deaths seen in our inhibitor cohort were due to malignancy, cardiovascular disease, and other, all non–hemophilia-related causes of death, these comorbidities likely affected our inhibitor-related mortality. Our finding of no excess mortality risk associated with an inhibitor is similar to a US study of 2950 males with severe hemophilia observed over a 3-year period (1993-1995).2 In that study, having an inhibitor was defined as a peak inhibitor titer of >1.0 BU/mL during the first year of follow-up.2 In contrast, a more recent US study of 7386 males with severe hemophilia A over a 13-year period (1998-2011) found that the odds of death were 70% higher among patients with a current inhibitor compared with those without (P < .01).5 Similarly, when the INSIGHT study only included patients with a known inhibitor at time of death, a fourfold higher risk of inhibitor-related mortality was observed.4 Given the lack of data on inhibitor status at time of death in our study, there is a possibility that the participants in the inhibitor group actually did not have an inhibitor at the time of death. This, along with the small number of inhibitor-related deaths, could have biased our results toward the finding of no association.
In our cohort, the age-adjusted mortality rate was 3.3 per 1000 person-years (95% CI, 2.7-3.9). This rate is roughly 10-fold lower than the reported results of Soucie et al, who described an age-adjusted mortality rate for severe hemophilia of 40.4 per 1000 person-years over a 3-year period (1993-1995).2 An analysis of US males with severe and mild hemophilia from 1998 to 2011 found that the mortality prevalence was substantially lower in the mild cohort as compared with the severe cohort, irrespective of age.7 The large difference in mortality rates between our cohort and severe hemophilia is not surprising. During the early 1980s, more than one-half of the US hemophilia population had become infected with HIV from contaminated factor concentrates.22 This led to an increase in mortality in all persons with hemophilia, but those with severe hemophilia were disproportionately affected.23,24 Notably, AIDS was the immediate cause of death in more than one-half of the severe hemophilia cohort that died in the study by Soucie et al.2 Also, our study reported a more recent time period from 2010 to 2018 with >70% of the cohort below the age of 40. Studies of birth cohorts have demonstrated that no persons with hemophilia, born in the United States in 1985 and later, were infected with HIV.25 This is evident in our study, where only 2.6% (n = 170) were infected with HIV, with 3 HIV-related deaths, thus partly explaining the low mortality rate.
Despite the relatively low mortality rate in persons with NSHA, we found an increased likelihood of death among persons with NSHA with hepatitis C or HIV, similar to prior mortality studies in the hemophilia population.2,5 Specifically, the observed death rate from liver disease in our cohort was significantly higher than the standard US population, after adjusting for age. In the past decade, treatment of hepatitis C has evolved rapidly with the introduction of direct-acting antiviral agents that results in a sustained virologic response of almost 100% within 12 weeks.26 Specifically in persons with inherited bleeding disorders, a phase 3 study found that 89.4% (42 of 47) of persons with hemophilia A/B or von Willebrand disease achieved sustained virologic response at 12 weeks, with only 1 serious adverse event in a hemophilia A patient with rectal bleeding.27 These results indicate a high level of efficacy, tolerability, and safety of interferon-free regimens in persons with inherited bleeding disorders with hepatitis C infection. As such, persons with hemophilia with chronic hepatitis C infection should be referred for eradication therapy, with the goal to reduce all-cause mortality and liver-related morbidity, including end-stage liver disease and hepatocellular carcinoma.
In our study, the most common primary cause of death was malignancy (n = 27, 20%). Similarly, the INSIGHT study reported that of their 148 deaths, the most common primary cause was malignancy (n = 38, 26%).4 Advances in hemophilia care have led to an increase in the overall life expectancy of persons with NSHA such that it is approaching that of the general population.12,28-30 As such, they are susceptible to similar age-related comorbidities such as malignancy and cardiovascular disease. Yet, it is reassuring that in our cohort, the observed number of deaths caused by malignancy was statistically lower than the standard US population. Other than hepatocellular carcinoma and lymphoma, which are highly prevalent in persons with hemophilia with hepatitis C and HIV, respectively, a review of the literature found no evidence of an increased incidence of other types of malignancy in persons with hemophilia as compared with the general population.31
With increasing longevity, the NSHA population is exposed to the same cardiovascular risk factors (hypertension, smoking, diabetes mellitus, hyperlipidemia, obesity) as the general population, which increases their risk for cardiovascular complications. A US study of 200 men with moderate and severe hemophilia aged between 54 and 73 years found that there was significantly less cardiovascular disease in the hemophilia cohort compared with age-comparable men (15% vs 25.8%; P < .001).32 In that study, men with mild hemophilia were specifically excluded.32 In our NSHA cohort, we found that the observed number of deaths caused by cardiovascular disease was statistically lower than the standard US population, after adjusting for age. This finding adds to the body of literature reporting that persons with NSHA appear to have some protection from fatal cardiovascular disease events as compared with the general population.
In our cohort, hemophilia-related cause of death occurred in 8 persons with NSHA. Because of the retrospective nature of this analysis, it is unclear if hemophilia-related deaths were due to bleeding complications or not. It would not be surprising if these were bleeding complications as fatal intracranial bleeding occurred in 12% of deaths in NSHA patients in the INSIGHT study (spontaneous, n = 13; traumatic, n = 4), a rate that is 3.5‐fold (95% CI, 2.0-5.8) higher than fatal intracranial bleeding in the general population.33 Again, this highlights the morbidity and mortality burden in NSHA patients and the need for active surveillance and monitoring by HTCs annually.
There are 2 major strengths to this study. First, this is the largest population of NSHA participants studied to date using a single, uniform data collection tool; the ATHNdataset is a national database where >32 000 persons with bleeding disorders have opted in. This unique resource provided us the ability to obtain information on persons with NSHA and on those with inhibitors that would otherwise be challenging given the small numbers involved. Second, this US study examines mortality rates for both males and females, whereas prior US studies were focused on males only.2,5,7
There are several limitations in this study that should be considered when interpreting the results. First, data entry into the ATHNdataset is voluntary and subject to missing or inaccurate data. This is evident by the findings that data on exposure to FVIII concentrate was missing in 14% of the NSHA cohort, and that 4 participants who were documented as having no exposure to FVIII concentrates were found to be inhibitor positive. Additionally, for participants with a documented exposure to FVIII concentrates, the number of EDs was not available in the ATHNdataset. This limited our ability to perform any inhibitor risk analysis. Second, the ATHNdataset is limited to persons cared for at HTCs that have authorized the sharing of their demographic and clinical information for research; thus, it may not be representative of the US hemophilia population. Because mortality data are highly influenced by age distribution, we compared our NSHA cohort with Community Counts, a public health monitoring program led by the Division of Blood Disorders at the Centers for Disease Control and Prevention. As of 29 September 2019, Community Counts included 10 608 persons with NSHA and had a similar age distribution as the ATHNdataset NSHA cohort, suggesting a representative population for the general US NSHA population.34 However, our NSHA cohort was not representative of the US general population. Because of unintentional nonrepresentativeness, there is a potential for bias because the outcome of interest, mortality, is affected by age. To account for this, we calculated age-adjusted mortality rates to allow for comparison with the general US population. Third, because the ATHNdataset was only established in 2010, this is a survival cohort in which participants had to survive to 2010 to be included, which likely led to underreported mortality rate for both inhibitor and noninhibitor participants. Last, because persons with inhibitors are more likely to be referred to HTCs for management, this could potentially bias our inhibitor cohort resulting in overestimation.
Our findings have important implications for future management and care of persons with NSHA. Continued monitoring of persons with NSHA by comprehensive care visits at HTC should occur annually to address hemophilia-related issues and other age-related comorbidities, in collaboration with the primary care physician and other subspecialists. Importantly, we found that in the NSHA cohort, the development of inhibitors occurred at an earlier age than previously reported. This highlights the importance of routine monitoring for inhibitors in the NSHA population, regardless of age, especially if they have recently received intense factor replacement therapy. Even though our study did not demonstrate any increased mortality risk with inhibitors, the management of inhibitors in NSHA remains a challenge with significant morbidity. In view of the lifelong risk for inhibitor development in persons with NSHA, future studies on risk factors for inhibitor development are much needed to gain a better understanding and to develop strategies to minimize the exposure, and thus prevent inhibitors.
Partially presented in oral form at the 61st annual meeting of the American Society of Hematology, Orlando, FL, 9 December 2019.
For original data, please contact support@athn.org.
Acknowledgments
The authors acknowledge the 135 ATHN-affiliated Hemophilia Treatment Centers and their patients for contributing to the ATHNdataset.
M.Y.L. received a 2015 HTRS/ATHN DREAM Award from the Hemostasis and Thrombosis Research Society and the American Thrombosis and Hemostasis Network, which was supported by an independent medical educational grant from Shire.
Authorship
Contribution: M.Y.L. designed the study, analyzed the data, and wrote the manuscript; D.C. analyzed the data and wrote the manuscript; M.R. reviewed and edited the manuscript; C.L.K. and N.S.K. supervised the study and reviewed and edited the manuscript; and all authors approved the final version of the manuscript and agree to be accountable for all aspects of the work, thereby ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Conflict-of-interest disclosure: M.R. has acted as a paid consultant to Bioverativ/Sanofi, CSL Behring, Genentech, Kedrion, NovoNordisk, Pfizer, Shire/Takeda, and uniQure; in addition, his organization has received research support from Bioverativ/Sanofi, BioMarin, Genentech, NovoNordisk, Shire/Takeda, Spark Therapeutics, and uniQure; is on the Board of Directors of Foundation for Women and Girls with Blood Disorders and Partners in Bleeding Disorders; and is employed by the American Thrombosis and Hemostasis Network. C.L.K. received honoraria for participation in advisory boards with Spark Therapeutics, Pfizer, and Genentech and research support from Novo Nordisk. N.S.K. has received research funding from Takeda, Grifols, and Pfizer; in addition, he is on a steering committee for clinical trials for uniQure and grants review committee for NovoNordisk. The remaining authors declare no competing financial interests.
Correspondence: Ming Y. Lim, Division of Hematology and Hematologic Malignancies, University of Utah, 2000 Circle of Hope, Room 4126, Salt Lake City, UT 84112; e-mail: ming.lim@hsc.utah.edu.
References
Author notes
The full-text version of this article contains a data supplement.