The coagulation factors V (FV) and VIII (FVIII) are important at sites of vascular injury for the amplification of the clotting cascade. Natural variants of these factors frequently lead to severe bleeding disorders. To understand the mechanisms of activation of FVIII by thrombin, we used a bank of mutant thrombins to define residues important for its activation. From the initial screening of 53 mutant thrombins for the activation of human recombinant FVIII, we mapped thrombin mutants with 50% or less activity to anion-binding exosite-I (Lys21Ala, His66Ala, Lys65Ala, Arg68Ala, Arg70Ala, and Tyr71Ala) and anion-binding exosite-II (Arg98Ala), the Na+-binding site (Glu229Ala, Arg233Ala, Asp234Ala, and Asp193Ala/Lys196Ala), and the 50-insertion loop (Trp50Ala), which were similar to our results for the activation of FV. The role of these residues for cleavage at Arg372 and Arg1689 was investigated using plasma FVIII. Anion-binding exosite-I appears to be important for cleavage at both sites, whereas the anion-binding exosite-II residue Arg98Ala is important for cleavage at Arg372 alone. The Glu229Ala mutant, which contributes to the Na+-binding site, and the 50-insertion loop mutant W50A have severely impaired cleavage at Arg372 and Arg1689. This suggests that the integrity of the active site and the Na+-bound form of thrombin are important for its procoagulant activity against FVIII. Detailed mutagenic analysis of thrombin can assist in understanding the pathogenesis of bleeding disorders and may lead to the rational design of selective thrombin inhibitors.
Introduction
The serine protease thrombin acts as a positive feedback amplifier of the coagulation cascade through the specific cleavage of factor V (FV) and factor VIII (FVIII). Factor Va (FVa) and factor VIIIa (FVIIIa) function as cofactors for prothrombinase and tenase complexes, respectively, on the surfaces of anionic phospholipids. This leads to the amplification of the coagulation pathway at sites of vascular injury.1 Both factors measure approximately 300 kDa and share a common domain organization, A1-A2-B-A3-C1-C2.2,3The A and C domains are 40% identical between FV and FVIII, but there is little sequence homology between the B domains. The A domains are similar to the A domains of ceruloplasmin,4 and the C domains are similar to phospholipid-binding proteins. The crystal structures of the FV and FVIII light-chain C2 domains have been determined and show hydrophobic and electrostatic interactions important for anchoring to the phospholipid membrane.5,6 Several studies have shown that the FVIII C2 domain also contains the von Willebrand factor (VWF)–,7,8 thrombin-,9 and factor Xa–binding sites.10
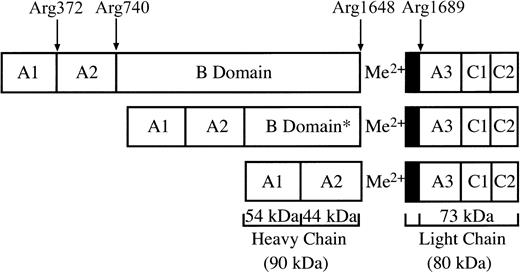
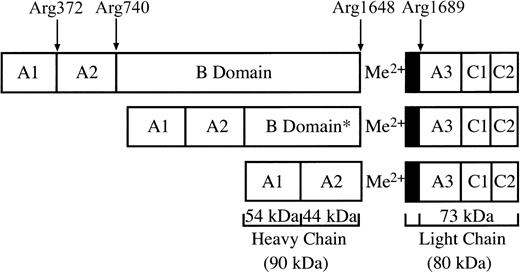
FV circulates as a full-length protein and is activated by thrombin releasing the B-domain activation products (E fragment and C1 fragment) by cleavage at the residues Arg709, Arg1018, and Arg1545. FVa is composed of the heavy (A1-A2 domain, 105 kDa) and light (A3-C1-C2 domain, 74 kDa) chains bound by a calcium ion.2,11,12 Specific cleavage of FV occurs preferentially at Arg709, then at Arg1018, followed by the rate-limiting cleavage at Arg1545. Cleavage at Arg709 and Arg1545 are important for full FVa cofactor activity, whereas cleavage of Arg1018 enhances the rate of Arg1545 cleavage.13 In contrast, FVIII circulates in plasma bound to VWF as a heterogeneous mixture of heterodimers (Figure 1) because of variable intracellular processing of the B domain. The variable heavy-chain fragments (A1-A2-B) range from 90 to 210 kDa, and they are bound to the 80-kDa light chain (A3-C1-C2). Thrombin activates FVIII by cleavage at Arg372 between the A1-A2domain and at Arg740 between the A2 and B domains to generate the 54-kDa A1 and the 44-kDa A2domains, and at Arg1689 to release a 40-amino acid acidic peptide from the 80-kDa light chain to generate the 73-kDa A3-C1-C2 fragment. FVIIIa is a metal-linked heterotrimer composed of the A1/A2/A3-C1-C2domains and lacking the B domain.14,15 Activation of plasma FVIII/VWF requires cleavages at Arg372 and Arg1689 for cofactor activity,16,17 whereas cleavage at Arg740 appears not to be rate limiting.16 Cleavage at Arg1689 releases an acidic 14-kDa fragment that leads to dissociation of VWF, allowing the association of FVIIIa to the phospholipid membrane.18 In the absence of VWF, cleavage at Arg372 is sufficient for full cofactor activity. These results are consistent with the naturally occurring mutations that give rise to hemophilia A.19
Domain organization of human FVIII.
FVIII circulates as a heterogenous mixture of heterodimers formed by variable intracellular processing of the B domain. The diagram shows the expected size fragments because of thrombin cleavage at Arg372, Arg740, and Arg1689. The asterisk denotes variable intracellular processing of the B domain.
Domain organization of human FVIII.
FVIII circulates as a heterogenous mixture of heterodimers formed by variable intracellular processing of the B domain. The diagram shows the expected size fragments because of thrombin cleavage at Arg372, Arg740, and Arg1689. The asterisk denotes variable intracellular processing of the B domain.
The specificity of thrombin is defined by 2 surface loops, the 50-insertion (Leu45-Asn57, thrombin-numbering system) and autolysis (Leu144-Gly155) loops, which occlude the active site, restricting access, and by 2 ligand-binding sites (exosites). Many thrombin substrates, receptors, and inhibitors overcome the occluded active site by binding to either of the 2 exosites, which are characterized by a high density of solvent-exposed basic residues. Anion-binding exosite-I (ABE-I, fibrinogen-binding exosite) binds fibrinogen,20-22PAR1,23-25 thrombomodulin,20,26-28 heparin cofactor II,29-31 and hirudin.32-34Anion-binding exosite II (ABE-II, heparin-binding exosite) binds glycosaminoglycan-bound serpins,29,35,36 platelet glycoprotein Ib,37,38 and hemadin.39 Studies using the inhibitor hirugen (ABE-I specific) and the ABE-II mutant thrombin RA (Arg89/Arg93/Arg98Ala) implicate ABE-I and ABE-II for thrombin cleavage and activation of FV and FVIII.40 Using a bank of 53 mutant thrombins with surface-exposed basic and polar residues substituted with alanine, we recently defined residues in both exosites, the Na+-binding site41 and the 50-insertion loop, important for FV activation and for cleavage at Arg709.42 In this study, we have used the same bank of mutant thrombins to define thrombin residues important in the activation of FVIII. We show that ABE-I and ABE-II are important for FVIII activation through cleavage of Arg372 but that ABE-II appears to be less important for cleavage at Arg1689. Recognition and cleavage at both sites requires the Na+-bound procoagulant form of thrombin41 for full cofactor activity. These studies are important for understanding the mechanisms of thrombin activation of FV and FVIII because no crystal structure data are available for these cofactors in complex with thrombin. This could lead to coherent strategies designed for the selective inhibition of thrombin to prevent the activation of platelets, cleavage of fibrinogen, and dampening of the coagulation cascade by impairing the formation of the prothrombinase and tenase complexes.
Materials and methods
Materials
Recombinant human FVIII (Kogenate) was purchased from Bayer (West Haven, CT), purified plasma human FVIII and D-CHG-Gly-Arg-pNA (Spectrozyme) are from American Diagnostica (Greenwich, CT). Purified human factor IX, X, and Xa were purchased from Hematologic Technologies (Essex Junction, VT). Unilamellar phosphatidylcholine/phosphatidylserine vesicles (PC-PS; 75%/25% wt/wt) were a gift from COR Therapeutics (South San Francisco, CA). The expression and purification of wild-type (WT) and alanine-substituted mutant thrombins are from CHO cells.43 The concentration of active thrombin molecules was determined by active site titration using D-Phe-Pro-Arg-chloromethyl ketone (PPACK; Sigma, St Louis, MO).42 Catalytic activity of the purified recombinant WT and mutant thrombins toward H-D-Phe-Pip-Arg-pNA (S-2238), fibrinogen, protein C, and thrombin-activatable fibrinolysis inhibitor (TAFI) has been described.43 Antibodies OBT0037A, FVIIIC monoclonal antibody, and ImmunoPure goat antimouse immunoglobulin G (IgG) were purchased from Accurate Chemical and Scientific (San Diego, CA), QED Bioscience (San Diego, CA), and Pierce (Rockfield, IL), respectively.
Activation of human FVIII by WT and mutant thrombins
Cleavage of human FVIII was assessed using a chromogenic assay. Forty-microliter volumes containing 100 nM human recombinant FVIII was incubated with WT or mutant thrombin (0.2 nM) in assay buffer (20 mM HEPES [N-2-hydroxyethylpiperazine-N′-2-ethanesulfonic acid], 150 mM NaCl, 5 mM CaCl2, and 0.1% poly(ethylene glycol) 6000, pH 7.4) for 10 minutes at 37°C. Thrombin concentration and incubation time were determined empirically such that depletion of FVIII was less than 20% for WT thrombin and diffusion of the A2 fragment was negligible. Reactions were stopped by adding PPACK and quenching on ice. The cleavage reaction was diluted 50-fold in a 200-μL reaction volume containing 20 μM PC-PS (75%/25% wt/wt), and 2 nM FIXa. FX was added to 100 nM, and the reaction was quenched after 30 seconds (time point determined empirically) by adding 20 μL of 0.5 M EDTA (ethylenediaminetetraacetic acid). The amount of FXa generated was determined by adding 80 μL of 250 mM spectrozyme to 20 μL FVIIIa cofactor Xa activation reaction and measuring the rate of spectrozyme hydrolysis. Initial rates of P-nitroanilide release were determined in a Softmax plate reader at 405 nm at room temperature. Thrombin mutants with markedly reduced activity compared to WT thrombin were used in dose-response curves over a range of thrombin concentrations such that they could be compared to WT thrombin within the linear range, where substrate depletion and A2 fragment diffusion are negligible.
SDS-PAGE and Western blot analysis of cleavage reactions
Cleavage reactions containing 50 nM human plasma–derived FVIII and 1 nM thrombin in assay buffer were incubated from 1 to 120 minutes before they were terminated by the addition of sodium dodecyl sulfate (SDS) loading buffer and boiling for 5 minutes. Cleavage products were resolved by SDS–polyacrylamide gel electrophoresis (SDS-PAGE) on 4% to 20% gradient SDS polyacrylamide gels (Bio-Rad, Hercules, CA) and then were stained with Biosafe Coomassie blue (Bio-Rad). Gels were Western blotted and probed with the monoclonal antibodies OBT0037A (specific for the heavy chain) and FVIIIC (specific for the light chain). Detection was achieved using goat antimouse IgG–horseradish peroxidase (HRP) and enhanced chemiluminescence (Amersham Pharmacia Biotech, Piscataway, NJ). The intensity of developed bands was determined by direct scanning of autoradiographs using the UMAX Astra 4000U scanner and Umax VistaScan 3.5.2 software. Scanned images were saved as tagged image file format (TIFF) files at a resolution 600 dots per inch. Pixel densities were calculated for each band from TIFF files imported into Scion Image 1.62c (http://rsb.info.nih.gov/nih-image/).
Results
Identification of thrombin mutants defective in human FVIII activation
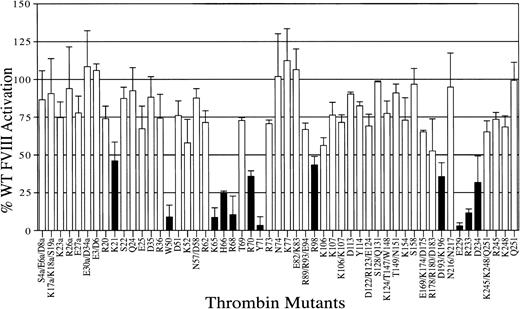
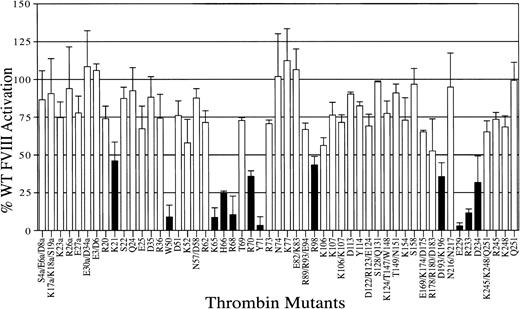
We used a site-directed mutagenesis strategy to determine thrombin residues important for human FVIII activation. A bank of 53 mutant thrombins in which solvent-exposed polar and charged residues were substituted with alanine43 was used in a 2-stage chromogenic screening assay with recombinant human FVIII. Recombinant FVIII is devoid of VWF, which usually binds plasma FVIII at a 1:1 molar ratio. For the activation of recombinant FVIII, cleavage at residue Arg372 appears to be sufficient for full cofactor activity of the tenase complex.16-18 40 Hence, decreases in activity from the mutations should reflect impaired cleavage at Arg372. Twelve mutants were identified with 50% or less FVIII activation compared with WT thrombin (Figure 2; Table1). These residues mapped mainly to ABE-I, ABE-II, the 50-insertion loop, and the Na+-binding site. ABE-I had 3 residues (Lys21, His66, and Arg70) with less than 50% FVIII activation and 3 residues (Lys65, Arg68, Tyr71) with less than 10% activity. Dose-response curves were performed for these mutants with severely diminished activation to ensure that they were within the linear range for cleavage and were found to be comparable to the initial screening (Figure3; Table 1). Compared to the screening with FV activation, alanine substitution of residues Lys21, His66, Arg68, Arg70, and Tyr71 appears to have similar decreases in FVIII cofactor activation. Among these, Arg68 and Tyr71 are the most important in that alanine substitution leads to a greater than 90% decrease in activation. However, some differences are observed. Thrombin residue Lys65 appears to be more important for FVIII activation than FV (8.5% vs 46.2% of WT activation), whereas the thrombin residues Arg73 and Lys77 appear to be less important for FVIII activation (70.7% and 112.4% vs 37.8% and 36% WT activation, respectively; Table 1).
Activation of recombinant human FVIII by thrombin mutants.
A 2-step chromogenic assay was used to screen the ability of WT and mutant thrombins to activate recombinant human FVIII, as described in “Materials and methods.” The effect of alanine substitution on FVIII activation is shown relative to 100% WT activity, with error bars representing the standard deviation for at least 2 individual experiments performed in duplicate. Single-letter amino acid codes have been used in this figure for spatial considerations.
Activation of recombinant human FVIII by thrombin mutants.
A 2-step chromogenic assay was used to screen the ability of WT and mutant thrombins to activate recombinant human FVIII, as described in “Materials and methods.” The effect of alanine substitution on FVIII activation is shown relative to 100% WT activity, with error bars representing the standard deviation for at least 2 individual experiments performed in duplicate. Single-letter amino acid codes have been used in this figure for spatial considerations.
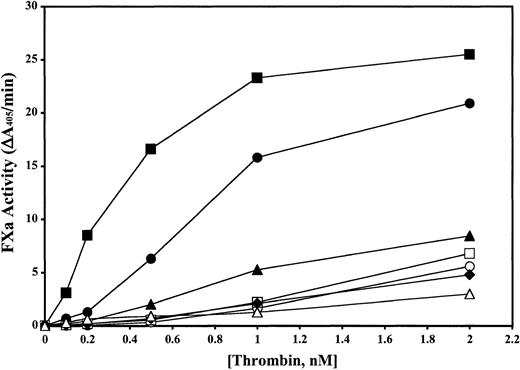
Dose dependence of thrombin cleavage of recombinant human FVIII.
The dose dependence of WT and mutant thrombin (0.1-2.0 nM) cleavage of human factor VIII (100 nM) was investigated using the 2-step chromogenic assay, as described in “Material and methods.” The diagram shows the dose dependence for WT thrombin (▪), the ABE-I mutants Lys65Ala (▴), Arg68Ala (■), and Tyr71Ala (♦), the ABE-II mutant Arg98Ala (●), the 50-insertion loop mutant Trp50Ala (○), and the Na+ loop mutant Glu229Ala (▵). All points had standard deviations of 15% or less from at least 2 separate experiments performed in duplicate.
Dose dependence of thrombin cleavage of recombinant human FVIII.
The dose dependence of WT and mutant thrombin (0.1-2.0 nM) cleavage of human factor VIII (100 nM) was investigated using the 2-step chromogenic assay, as described in “Material and methods.” The diagram shows the dose dependence for WT thrombin (▪), the ABE-I mutants Lys65Ala (▴), Arg68Ala (■), and Tyr71Ala (♦), the ABE-II mutant Arg98Ala (●), the 50-insertion loop mutant Trp50Ala (○), and the Na+ loop mutant Glu229Ala (▵). All points had standard deviations of 15% or less from at least 2 separate experiments performed in duplicate.
ABE-II also plays a role in the activation of FVIII, but its importance appears to be less than that for FV activation. The mutant Arg98Ala has 43.3% of WT activation in the initial screen; however, dose-response studies for this mutant show 32.4% WT FVIII activation, which is similar for its activation of FV (Figure 3; Table 1). Two triple mutants, Arg89Ala/Arg93Ala/Glu94Ala and Arg245Ala/Lys248Ala/Gln251Ala, have decreased FV activation but not FVIII activation. The only common ABE-II residue for FV and FVIII activation appears to be Arg98. More than 13 basic residues comprise ABE-II, some of which are not represented in the current collection of alanine-thrombin mutants. Therefore, it is possible that not all electrostatic interactions are represented in this study. All mutant thrombins in ABE-I and ABE-II with impaired FVIII activation have normal catalysis for the chromogenic substrate, S-2238,43suggesting that impaired binding within the exosites rather than an effect on catalysis is responsible for decreased cofactor activation.
The residues Glu229, Arg233, and Asp234, which are important for the integrity of the Na+-binding site,41 have drastically reduced FVIII activation when substituted with alanine (2.9%, 11.5%, and 35.6% of WT FVIII activation, respectively). Dose-response studies for Glu229Ala show a similar decrease in FVIII activation, consistent with the screening (Figure 3; Table 1). These are similar to results observed for FV activation. The double mutant Asp193Ala/Lys196Ala showed 35.6% of WT FVIII activation. Residues lie on an extended insertion loop (residues Tyr190 to Gly198), where both side chains point into the solvent.44 The residue Arg197 on this loop makes a bidentate ion pair with the Na+-binding site residues Asp221 and Asp22241,45; hence, it is possible that either of these 2 mutations could disrupt interactions between the loop and the Na+-binding site, compromising the Na+-bound procoagulant form of thrombin. Alternatively, the residues could be involved in making direct interactions. A large decrease in FVIII activation (8.9% WT activation) was noted for the residue Trp50Ala, which forms part of the hydrophobic 50-insertion loop that occludes the active site restricting the specificity of thrombin. Trp50Ala has diminished activity toward the chromogenic substrate S-2238,43 as reflected by an increase inKm, implying the mutation affects substrate binding by altering the topology of the S subsites affecting specific contacts. FVIII activation is more impaired than FV activation, which suggests that Trp50 has a greater contribution to the specificity of cleavage at one or more of the FVIII cleavage sites.
Analysis of human FVIII activation by SDS-PAGE
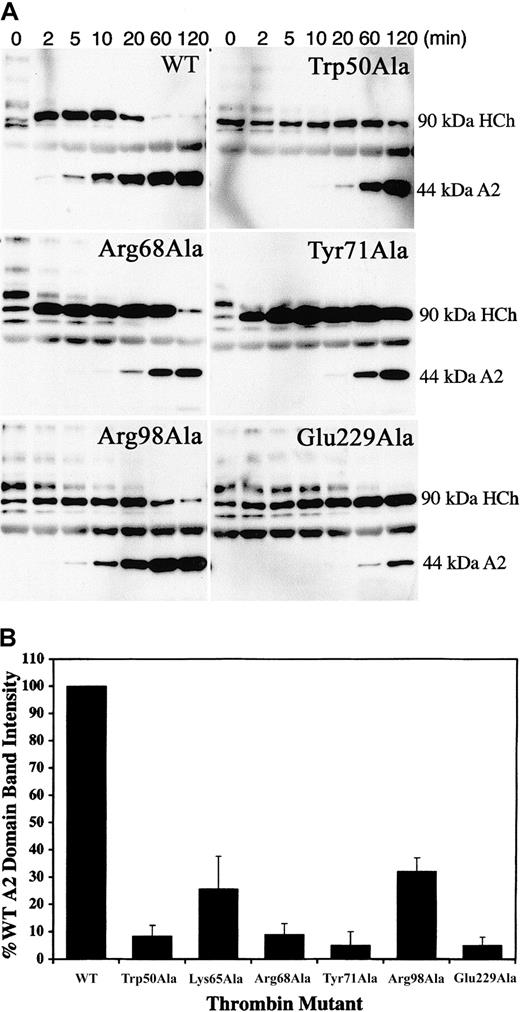
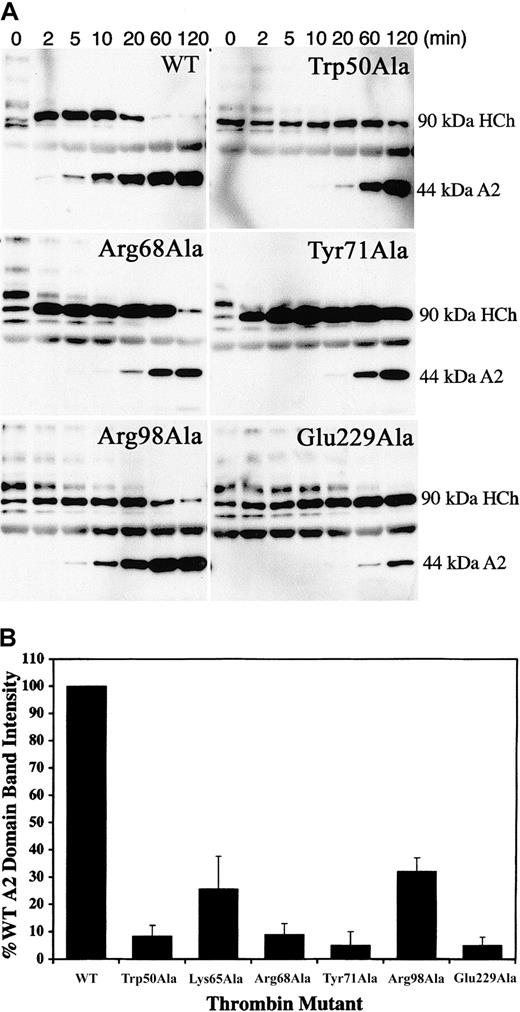
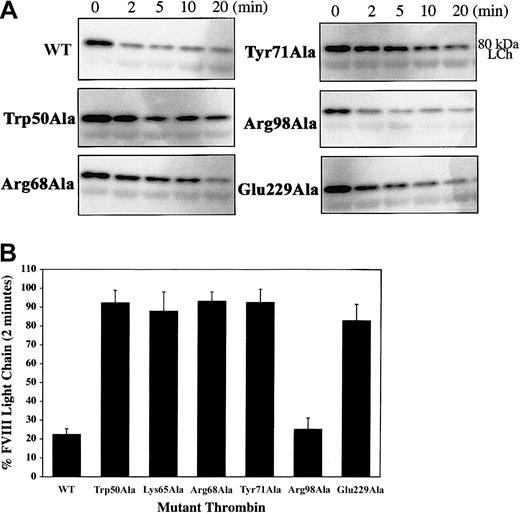
To determine the structural requirements of thrombin necessary for the activation of FVIII by specific cleavage at Arg372 and Arg1689, we used Western blot analysis of cleavage reactions containing purified plasma FVIII using monoclonal antibodies specific for the heavy-chain A2 domain and the light-chain acidic N terminus. Cleavage at Arg1689 releases the 14-kDa acidic fragment, leading to a dissociation of VWF that allows FVIIIa to act as a cofactor in the tenase complex. Using the monoclonal antibody OBT0037A (specific for the heavy-chain A2 domain), we monitored the cleavage of the heavy-chain heterodimers by the specific appearance of the 44-kDa A2 domain. Compared with WT, the thrombin mutants Trp50Ala, Arg68Ala, Try71Ala, Arg98Ala, and Glu229Ala had slightly delayed appearance of the 90-kDa A1-A2 heavy chain from the cleavage of the various-sized heavy-chain heterodimers (ranging from 220 to 90 kDa), suggesting cleavages at Arg740 and Arg1313 (located in the B domain) are not severely affected by the mutations. For most mutants, however, the 90-kDa A1-A2band persisted, and the appearance of the 44-kDa A2 domain was significantly delayed (Figure 4A). Quantitation of the A2 domain generated by the cleavage of FVIII by WT and mutant thrombins at 10 minutes (Figure 4B) showed dramatically reduced band intensities, suggesting that ABE-I, ABE-II residue Arg98, 50-insertion loop residue Trp50, and Na+-binding site are all necessary for cleavage at Arg372.
Thrombin cleavage at the Arg372-Ser373 scissile bond.
(A) Cleavage reactions containing 50 nM human plasma FVIII and 1 nM thrombin were performed at 37°C over several time points, ranging from 2 to 120 minutes. Cleavage products were resolved by SDS-PAGE and were Western blotted using the monoclonal antibody OBT0037A, which is specific for the heavy-chain A2 domain. Cleavage at Arg372-Ser373 gives rise to the 44-kDa A2 fragment, detected by the antibody. (B) To directly compare the effects of the alanine-substituted thrombins on cleavage at Arg372, we performed cleavage reactions for FVIII by WT and mutant thrombins for one time point (10 minutes) in duplicate and loaded onto the same gel. The reactions were resolved by SDS-PAGE and were Western blotted as outlined in panel A. The intensity of the A2 fragment generated by cleavage at Arg372 was determined by direct scanning of the Western blot (data not shown). A2 band intensity generated by each mutant (the mutant Lys65Ala was included in these studies) is presented in a bar graph as a percentage of A2band intensity with respect to WT thrombin (100%).
Thrombin cleavage at the Arg372-Ser373 scissile bond.
(A) Cleavage reactions containing 50 nM human plasma FVIII and 1 nM thrombin were performed at 37°C over several time points, ranging from 2 to 120 minutes. Cleavage products were resolved by SDS-PAGE and were Western blotted using the monoclonal antibody OBT0037A, which is specific for the heavy-chain A2 domain. Cleavage at Arg372-Ser373 gives rise to the 44-kDa A2 fragment, detected by the antibody. (B) To directly compare the effects of the alanine-substituted thrombins on cleavage at Arg372, we performed cleavage reactions for FVIII by WT and mutant thrombins for one time point (10 minutes) in duplicate and loaded onto the same gel. The reactions were resolved by SDS-PAGE and were Western blotted as outlined in panel A. The intensity of the A2 fragment generated by cleavage at Arg372 was determined by direct scanning of the Western blot (data not shown). A2 band intensity generated by each mutant (the mutant Lys65Ala was included in these studies) is presented in a bar graph as a percentage of A2band intensity with respect to WT thrombin (100%).
Cleavage at Arg1689 was monitored by Western blot analysis using the monoclonal antibody FVIIIC, which appears to recognize an epitope on the 14-kDa acidic fragment. With the exception of Arg98Ala, all mutant thrombins showed a delay in cleavage at Arg1689, indicated by the continued presence of the 80-kDa uncleaved light chain (Figure5A), consistent with reduced cofactor activation. Arg98Ala appears to efficiently cleave at Arg1689 with a band intensity of 75% that of WT (Figure 5B), suggesting that ABE-II may not play an important role in cleavage of Arg1689 at this site in the presence of VWF. The Arg98Ala mutant has only 32.4% FVIII activation using recombinant FVIII (Table 1), suggesting that cleavage at Arg372 is sufficient for full cofactor activity in the absence of VWF.
Thrombin cleavage at the Arg1689-Ser1690 scissile bond.
Cleavage reactions and Western blotting conditions were similar to those in Figure 4, except the primary antibody FVIIIC, which recognizes the 80-kDa light chain, was used. (A) Western blots depicting the disappearance over time for the 80-kDa light chain caused by the specific cleavage at Arg1689-Ser1690 and the loss of the N-terminal fragment bearing the antibody epitope. (B) Percentage of FVIII light chain remaining after 2-minute incubation with WT and mutant thrombin (including Lys65Ala), as determined by measuring the band intensity, and presented on the bar graph as a percentage of total light chain (time 0).
Thrombin cleavage at the Arg1689-Ser1690 scissile bond.
Cleavage reactions and Western blotting conditions were similar to those in Figure 4, except the primary antibody FVIIIC, which recognizes the 80-kDa light chain, was used. (A) Western blots depicting the disappearance over time for the 80-kDa light chain caused by the specific cleavage at Arg1689-Ser1690 and the loss of the N-terminal fragment bearing the antibody epitope. (B) Percentage of FVIII light chain remaining after 2-minute incubation with WT and mutant thrombin (including Lys65Ala), as determined by measuring the band intensity, and presented on the bar graph as a percentage of total light chain (time 0).
Discussion
The activation of human FV and FVIII by thrombin plays an essential role in the amplification of the coagulation cascade at sites of vascular injury. In this report, we have investigated the role of specific thrombin residues important in the activation of FVIII and have compared these findings with our recent studies for the activation of the structurally related FV using a collection of alanine-scanning, mutagenesis-generated thrombin mutants.43 The preliminary screen and dose dependence for human recombinant FVIII activation primarily reflects cleavage at Arg372-Ser373 because, in the absence of VWF, cleavage at this site is sufficient for full cofactor activity. The results implicate the role of ABE-I, ABE-II, the 50-insertion loop residue Trp50, and the Na+-binding site for recombinant FVIII cofactor activation. This shows a similar pattern for FV activation though there are subtle differences (Table 1; Figure 6). Thrombin uses ABE-I residues Lys21, Lys65, His66, Arg68, Arg70, and Tyr71 for the activation of recombinant FVIII (presumably at Arg372-Ser373). By comparison, more residues in ABE-I are required for the activation of FV (Lys21, Lys65, His66, Arg68, Arg70, Tyr71, Arg73, and Lys77). The role of these residues could be direct interaction with factor FVIII, or, alternatively, substitution of these residues may cause a conformational change affecting other contacts. ABE-I extends away from the thrombin active site and is characterized by a high density of surface-exposed basic and hydrophobic amino acids. Crystal structures of thrombin bound to PAR1,24thrombomodulin,46 and the inhibitors hirudin,33 triabin,47ornithodorin,48 and rhodniin49 have shown the importance of one or more of the basic residues (Lys21, Arg68, Arg70, Arg73, and Lys77) in ABE-I for direct ion pair interactions. They have also shown the role of Tyr71 on the surface-exposed patch formed with Phe34, Ile82, Met84, and Leu65 for hydrophobic interactions. Mutant thrombins Lys65Ala, Arg68Ala, and Tyr71Ala—all of which had less than 10% WT FVIII activation, were chosen to explore the role of ABE-I for the recognition and cleavage of plasma-derived FVIII bound with VWF. Activation of FVIII/VWF occurs through cleavage at Arg372 and Arg1689 for full cofactor activity. Cleavage at Arg1689 releases an acidic 14-kDa N-terminal fragment that allows the dissociation of VWF. The binding sites on FVIII for VWF50 are distinct from the thrombin-binding site,9 and it is thought that VWF does not compromise thrombin-catalyzed activation of FVIII. All 3 mutants were impaired for cleavage at both sites, suggesting the importance of ionic and hydrophobic interactions for ABE-I binding. The ABE-I residues Arg73 and Lys77 have little effect on the preliminary screen with recombinant FVIII but may have an effect at Arg1689-Ser1690 of FVIII bound with VWF. Potential hirudinlike sites on FVIII, capable of binding to ABE-I, are located N- and C-terminally to the Arg372-Ser373 bond. The thrombin-binding site, however, is thought to be in the A2 domain.9 A potential ABE-I binding site for cleavage at the Arg1689-Ser1690 site is located N-terminally in the 14-kDa acidic domain. Studies by Nogami et al9 using a monoclonal antibody against the C2 domain suggest this is where the major thrombin-binding site is located. Two possibilities exist. One is that the antibody could be sterically hindering the access of thrombin. The other is that the binding of thrombin to the C2 domain juxtaposes the thrombin-active site adjacent to the thrombin-cleavage site on FVIII. Interestingly, there are no hirudinlike sequences in the C2 domain.
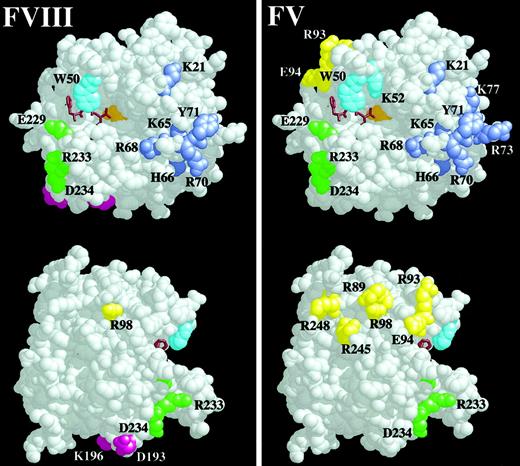
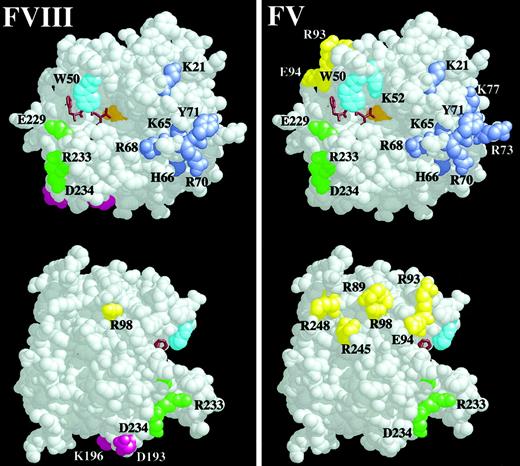
Space-filling model of thrombin residues important for recombinant human FVIII activation.
The RasMol V2.5 software package was used to depict thrombin (E.C.3.4.21.5, Brookhaven entry 1PPB) as a space-filling model with solvent removed. Residues substituted with alanine with less than 50% WT human FVIII activation are colored and shown in the left panel, and they are compared with thrombin residues important for the activation of FV in the right panel. The upper thrombin molecule is the classic front view of thrombin,51 with the active-site cleft running horizontally left to right and the 50-insertion loop on the top of the cleft occluding the active site Ser205 (orange). The active site is shown with the bound active-site inhibitor, PPACK, as a brown stick model. The 50-insertion loop residues are cyan, and the Na+-binding loop residues are green. ABE-I runs to the right of the active site cleft, and the residues are lavender. The bottom thrombin molecule is rotated 90° to the right and shows ABE-II residues in yellow and the residues Lys196 and Asp193 in magenta. Residues of interest are labeled using the single-letter amino acid code.
Space-filling model of thrombin residues important for recombinant human FVIII activation.
The RasMol V2.5 software package was used to depict thrombin (E.C.3.4.21.5, Brookhaven entry 1PPB) as a space-filling model with solvent removed. Residues substituted with alanine with less than 50% WT human FVIII activation are colored and shown in the left panel, and they are compared with thrombin residues important for the activation of FV in the right panel. The upper thrombin molecule is the classic front view of thrombin,51 with the active-site cleft running horizontally left to right and the 50-insertion loop on the top of the cleft occluding the active site Ser205 (orange). The active site is shown with the bound active-site inhibitor, PPACK, as a brown stick model. The 50-insertion loop residues are cyan, and the Na+-binding loop residues are green. ABE-I runs to the right of the active site cleft, and the residues are lavender. The bottom thrombin molecule is rotated 90° to the right and shows ABE-II residues in yellow and the residues Lys196 and Asp193 in magenta. Residues of interest are labeled using the single-letter amino acid code.
ABE-II does not appear to be as important for the activation of FVIII as for the activation of FV using our collection of mutant thrombins. Only residue Arg98 appears to have an effect on the activation of FVIII at the Arg372-Ser373 cleavage site. Esmon and Lollar40suggest ABE-II is more important for the activation of FVIII using the triple mutant, RA (Arg89Ala/Arg93Ala/Arg98Ala). The thrombin mutant collection does not encompass all the possible residues within ABE-II; therefore, it is possible that ABE-II may still have an important role in the activation of FVIII at both cleavage sites. The loss of activity caused by the Arg98Ala substitution could implicate this residue in direct interactions with FVIII. For example, Arg98 has been shown to be involved in ion-pair interactions from the crystal structures of thrombin bound to the leech inhibitor, heamadin,39 and the prothrombin F2 domain.51For cleavage at Arg372, both ABE-I and ABE-II are required for hydrolysis, suggesting binding to distinct FVIII domains. The A2 and C2 domains have been implicated in binding thrombin,9 and a hirudinlike sequence N-terminal to the Arg372-Ser373 bond (residues 330-363) in the A1domain could also play a role.
Consistent with the mapping studies of FV is the importance of Glu229, Arg233, and Asp234 at the Na+-binding site.41,45 The Na+-binding site is defined by a cavity formed by 3 antiparallel B-chain β-strands (Met185-Gly189, Val225-Cys231, and Tyr237-Tyr240) and the loops Tyr190-Gly198 and Cys231-Tyr237.41,45 The site is adjacent to the autolysis loop and forms, in part, the southern rim of the thrombin-active site cleft.45 The Cys231-Tyr237 loop appears to be stabilized by a bidentate ion pair between Arg197 and the residues Asp232 and Asp234, between Glu229-Lys235 and between Glu146-Arg233. The carbonyl oxygens of Arg233 and Lys224 are involved in coordinating an Na+ atom.41,45 Mutation of Glu229, Arg233, and Asp234 leads to greatly diminished FVIII cofactor activity. Each substitution could have the effect of destabilizing the Na+-binding loop, leading to the unbound “slow form” of thrombin. Further, either of the Asp193Ala/Lys196Ala substitutions could indirectly affect the Cys231-Tyr237 Na+-binding loop by impairing the ion-pair interactions between Asp232 and Asp234 to Arg197. Previous alanine-scanning studies have shown the double mutant, D193/K196, has mild decreases in fibrinogen clotting,28 FV activation,42 and thrombomodulin-dependent protein C activation but near normal activity toward thrombomodulin-dependent TAFI activation43 and inhibition by antithrombin III.52 These studies do not exclude the possibility that either residue may be involved in direct interactions. The mutant Glu229Ala was used to assess the role of the Na+-bound procoagulant form of thrombin for recognition and cleavage at FVIII residues Arg372 and Arg1689. Cleavage at both sites was severely diminished. The Glu229Ala, Arg233Ala, and Asp234Ala mutants have been shown by previous mapping studies to have an effect on fibrinogen clotting, activation of protein C, and ATIII inhibition, whereas Glu229Ala has a major effect on platelet activation.53,54These mutations do not affect thrombin aptamer55 or thrombomodulin binding, and they only mildly affect TAFI activation.43 The crystal structures of thrombin-fibrinopeptide A56 and thrombin-hemadin39 complexes have shown direct contacts made by Glu229 and Arg233, respectively, but whether these residues form direct contacts for the recognition and cleavage of FV and FVIII has yet to be determined. Our studies suggest the Na+-bound form of thrombin is important for efficient cleavage at both FVIII cleavage sites.
The thrombin residue Trp50 forms part of the 50-insertion loop that occludes the active site, restricting the specificity of thrombin. Trp50 plays an important role in defining the apolar S2subsite and has been shown to make substantial contacts in the thrombin complex with hirudin,33 hemadin,39rhodniin,49 fibrinopeptide A,56 and PPACK44 complexes. Consistent with these crystal structures is the dramatic effect of the W50A mutation for cleavage at FV residue Arg709 and FVIII residues Arg372 and Arg1689, which can be explained as the loss of important contacts or disruption of the S2 subsite. Evidence exists that the mutation can have long-range effects on the Na+-binding site.57
To date, no crystal structures exist to define the precise structural requirements of thrombin in the recognition and cleavage of FV and FVIII. Saturated alanine-scanning mutagenesis of thrombin has proved valuable in defining thrombin residues important in the recognition and cleavage of FV and FVIII that should aid in the rational design of thrombin inhibitors.
Prepublished online as Blood First Edition Paper, May 31, 2002; DOI 10.1182/blood-2002-03-0843.
Supported by National Institutes of Health grant R01 HL57530 and the Cheong Har Family Foundation.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.
References
Author notes
Timothy Myles, Lawrence L. K. Leung, Division of Hematology, CCSR Rm 1155, Stanford University School of Medicine, Stanford, CA 94305-5156; e-mail: tmyles@stanford.edu,lawrence.leung@stanford.edu.