Abstract
The clinical and cytogenetic features associated with T-cell acute lymphoblastic leukemia (T-ALL) are not predictive of early treatment failure. Based on the hypothesis that microarrays might identify patients who fail therapy, we used the Affymetrix U133 Plus 2.0 chip and prediction analysis of microarrays (PAM) to profile 50 newly diagnosed patients who were treated in the Children's Oncology Group (COG) T-ALL Study 9404. We identified a 116-member genomic classifier that could accurately distinguish all 6 induction failure (IF) cases from 44 patients who achieved remission; network analyses suggest a prominent role for genes mediating cellular quiescence. Seven genes were similarly upregulated in both the genomic classifier for IF patients and T-ALL cell lines having acquired resistance to neoplastic agents, identifying potential target genes for further study in drug resistance. We tested whether our classifier could predict IF within 42 patient samples obtained from COG 8704 and, using PAM to define a smaller classifier for the U133A chip, correctly identified the single IF case and patients with persistently circulating blasts. Genetic profiling may identify T-ALL patients who are likely to fail induction and for whom alternate treatment strategies might be beneficial.
Introduction
Acute lymphoblastic leukemia (ALL) is the most common form of cancer among children and young adults. Approximately 15% of patients express cellular and molecular features that are unique to T-lineage acute lymphoblastic leukemia (T-ALL).1-4 Through the use of increasingly dose-intensive therapy, combined with an improved understanding of leukemic pathogenesis, disease-free survival for children with ALL has improved over the past 3 decades.5 However, when matched for NCI-designated clinical risk features of age, initial white blood cell count, and evidence of extramedullary disease, patients with T-ALL are at an increased risk of relapse compared with children treated for precursor B-lineage acute lymphoblastic leukemia (B-ALL).6 In addition, unlike many of the genetic biomarkers observed in patients with precursor B-ALL, the recurring karyotypic aberrations identified in T-ALL do not consistently correlate with outcome on modern treatment schemas.2,7,8 For these reasons, the identification of prognostically relevant karyotypic and clinicopathologic abnormalities in T-ALL has been difficult to elucidate. The recent identification of T-ALL risk groups, as defined by minimal residual disease (MRD) status,6,9,10 activating NOTCH1 mutations,11-13 and response to induction therapy,6,14,15 can be used to stratify treatment approaches. Nevertheless, the mechanisms of drug resistance that result in persistent disease and early treatment failure remain poorly understood
Gene expression microarrays are spotted with thousands of 25mer oligonucleotides, which correspond to transcripts of known and hypothetical genes within the human genome. By using microarrays for class discovery in hematopoietic malignancies, it has been possible to identify novel pathways in malignant transformation,16,17 explore heterogeneities among study populations,18-21 and segregate patients into prognostically relevant subsets.18,22 While numerous genes and genetic signatures predicting disease course have been identified for patients with acute myelogenous leukemia, precursor B-ALL, and lymphomas,16,19,23-27 far fewer microarray studies have been designed to specifically investigate expression profiles having prognostic relevance in T-ALL. These limitations prompted us to search for robust, genetically defined prognostic markers that might allow the early identification of patients who might fail treatment. In previous microarray and genetic profiling studies, aberrant expression of T-cell–specific transcriptional factors was shown to determine the stage of oncogenic arrest in thymocyte development, as well as contribute to leukemogenesis,2,16,17,28 but did not consistently predict outcome across age groups or treatment approaches.18,29
Because response-based therapy is becomingly increasingly important in ALL risk stratification, the identification of the genes that modulate drug resistance during induction is an important first step in understanding the mechanisms of early treatment failure. To gain insight into the genetic basis of treatment failure in T-ALL, we performed microarray analyses in 50 children and young adults who were treated in Children's Oncology Group (COG) 9404. We identified a genomic classifier that can distinguish patients who fail induction therapy and validated this classifier in an independent study. Seven genes were similarly upregulated in both the genomic classifier for IF patients and T-ALL cell lines having acquired resistance to neoplastic agents, identifying potential target genes for further study in drug resistance. These results indicate that genetic profiling may identify patients with T-ALL for whom induction therapy is likely to fail, and for whom an alternate treatment plan might be more beneficial.
Patients, materials, and methods
Study design, definition, case selection, and induction schemas
For children and young adults with newly diagnosed T-ALL, COG Study 9404 was open to accrual from June 1, 1996, to September 10, 2001, and enrolled 573 patients. Inclusion criteria included a diagnosis of T-ALL or T-lineage lymphoblastic lymphoma, age between 1 and 21.99 years, provision of written informed consent obtained in accordance with the Declaration of Helsinki, and no prior therapy except for emergency radiation or fewer than 48 hours of emergency prednisone. This study received institutional review board approval at each participating institution (Document S1, available on the Blood website; see the Supplemental Materials link at the top of the online article). The study design and results for COG T-ALL study 8704 have been reported elsewhere.1 Pretreatment leukemia samples from the bone marrow or peripheral blood were obtained from all patients through the COG 9400 or 9900 ALL Classification Study. Independent confirmation of T-ALL at a central reference laboratory required the presence of CD7, CD5, and/or CD2, or no coexpression of myeloid antigens in cases having expression of CD7 alone. While encouraged, cytogenetic analyses were not required for enrollment on either treatment study. Bone marrow cryopreservation was performed in cases having a sufficient sample size.
Using a retrospective, case-control design, samples from patients who either failed to achieve remission (induction failure [IF]), who relapsed within 4 years (relapse [RE]), or who remained in complete continuous remission (CCR) for at least 4 years were randomly selected for study. The induction drugs and response assessment time points for both studies are shown in Table 1. Patients were defined as having IF when they had 25% or more bone marrow blasts (M3) at day 22 or 43; at the discretion of the treating physician, those with an M2/M3 marrow could receive 2 additional weeks of therapy. Induction failure in COG 8704 was identified by the M3 marrow status at day 29. Relapse was defined as occurring when the marrow was M3 or if leukemic blasts could be identified in CSF with 5 or more mononuclear cells/μL at any time following first remission. CCR was defined as a continuous absence of T lymphoblasts (< 5%) in the bone marrow (M1) and CSF.
Sample preparation and expression profiling
Freshly acquired samples were enriched to more than 90% T-ALL blasts with Ficoll Hypaque centrifugation and stored in liquid N2 until thawed for RNA purification. Total RNA was extracted from 5.0 × 106 to 107 T-ALL cells using the RNeasyMini Kit (Qiagen, Valencia, CA). Expression profiling at the Keck-UNM Genomics Resource was performed using 2.5 μg total RNA for either a one-step approach to synthesize biotin-labeled cRNA for COG 9404 samples, or a 2-step approach for COG 8704 samples (Affymetrix, Santa Clara, CA). Using either approach, 15-μg aliquots of fragmented, labeled cRNA were hybridized to the U133 Plus 2.0 chip (50 COG 9404 samples), which has 54 675 probe sets, or to the U133A chip (42 COG 8704 samples), which has 22 283 probe sets.16,25 Chips were scanned using an Affymetrix microarray scanner (Hewlett Packard, Palo Alto, CA), while fluorescence intensity values were captured using GeneChip software (Affymetrix). Microarray processing was performed using the Affymetrix Microarray Analysis Suite (MAS version 5).30 Scan quality was assured based on a priori quality control criteria, including visible microarray artifacts. As recommended by others,31 chips were discarded and the procedure repeated if the scaling factor was found to be more than 50, the “present” calls were less than 20%, or the GAPDH/actin ratio was more than 3. To maintain consistency in approach, only cryopreserved samples were profiled.32 Of the 141 samples identified for analysis, 39 were excluded because of defects in the nucleic acids (poor quality RNA or hybridization artifacts), and 10 cases were excluded because the reason for death was unrelated to disease progression, the patient/family withdrew consent, or the outcome data were incomplete, leaving 92 cases available for profiling. Approval from the ethics committee was obtained (University of New Mexico Human Research Review Committee [HRRC] no. 03-183). We wished to further test our classifier, but could not identify additional IF cases in the COG Cell Bank repository. Our searches for testable IF microarrays in the GEO,33 ArrayExpress,34 or caARRAY35 databases were also unsuccessful.
Biocomputational and statistical methods for microarray analysis
Statistical analysis of raw Affymetrix microarray data at a probe set level was performed using robust multiarray average (RMA) normalization, which corrects arrays for background using a transformation metric, normalizes the data based on a normal distribution, and uses a linear model to estimate expression values on a log scale.36 We used prediction analysis of microarrays (PAM), which uses a nearest shrunken centroid classifier to develop a predictive signature for 2 or more classes. As an improvement to the standard nearest centroid classification, PAM also “shrinks” each of the class centroids toward their overall mean, termed a “threshold,” to improve the prediction accuracy. PAM also applies k-fold cross-validation inselecting optimal number of genes by controlling both overall and individual class cross-validation error rates.37 Hierarchic clustering was performed with the R package “gplots” (http://cran.rproject.org/src/contrib/Descriptions/gplots.html). Principal component analysis (PCA) was accomplished in R, for which a 3-dimensional plot was generated by projecting data to the first 3 principal components; the image was developed in the R package “rgl.” To search for gene network pathways, we searched BioCarta, KEGG, and Reactome pathways at (http://hatch.health.unm.edu/Genotation) and Ingenuity Systems software at (http://www.affymetrix.com/products/software/compatible/pathway.affx).
Development of T-ALL cell lines with resistance to daunorubicin and asparaginase
Jurkat and Sup T1 cell lines were developed for acquired resistance to therapeutic doses of daunorubicin (DNR) and l-asparaginase (l-asp), and represent the cortical and mature developmental stages of T-cell development, respectively (David A. Estes, S.S.W., and R.S.L., Genetic alterations determine chemotherapy resistance in childhood T-ALL: modeling in stage-specific cell lines and correlation with diagnostic patient samples, in press). Briefly, these cells were developed by maintaining initially chemosensitive parental cell lines in incrementally increasing concentrations of chemotherapeutic drugs for more than 6 months. Cells are continuously maintained in chemotherapeutic agents. The DNR-resistant Jurkat and Sup T1 cells have a 117-fold increase in the 50% inhibitory concentration to DNR, from 20.6 nM to 2.419 μM, while the l-asp–resistant cells showed increases in resistance to l-asp of 320-fold (0.003 IU/mL to 0.962 IU/mL) and 29-fold (0.042 IU/mL to 1.22 IU/mL), respectively. Jurkat cells that had acquired resistance to DNR also developed a 310-fold (0.8 nM to 250 nM) resistance to vincristine and a 120-fold (3.1 nM to 380 nM) resistance to prednisone.
Results
Patient demographics, karyotypic aberrations, and outcome characteristics
Our samples came from patients who were more likely to be males, and tended to present with NCI high-risk features (Table 2), as reported in other T-ALL studies.2,38,39 Overall, induction remission rates for the COG 8704 and 9404 studies were 96.5% and 91.7%, respectively. To develop our classifier, COG 9404 cases were used as the training set, and COG 8704 cases were used as the test set. In all cases, bone marrow samples were tested for karyotypic abnormalities and patient demographics were annotated for outcome-related correlative features. For COG 9404 patients, cytogenetic analyses showed a normal karyotype in 5 cases, deletions 6q (n = 2) and 5q (n = 1), translocations t(4;8), t(10p;11q), and t(12;22), nonspecific abnormalities (n = 6), and unknown results (n = 36). Of the 50 patient profiles in this study, 6 did not achieve remission during the induction phase of therapy, and 44 achieved remission by day 42. Three of 6 IF patients had more than 1000 absolute blasts/μL at day 8 of induction. The median end-of-induction bone marrow blast percentage was 61% (range: 29%-70%) in IF patients, and 1% (range: 0%-5%) in the RE and CCR groups. Among patients achieving a first remission, 14 relapsed within a median period of 245 days (range: 128 to 1669 days), and 30 have remained in CCR for a median period of 2200 days (range: 1833 to 2807 days).
Five of the 6 IF patients achieved remission with second-line therapies consisting of nelarabine, either alone (n = 2) or in combination with other agents (n = 1), cytarabine in combination with etoposide (n = 1), or with an unknown combination of drugs (n = 1). Three patients who achieved remission received allogeneic bone marrow transplantation; one patient died from posttransplantation infectious complications, another died from persistent disease, and one survives in CCR. In agreement with others, we found no correlation between cytogenetic aberrations and NCI features with early treatment failure.2,7,8
PAM distinguishes patients on COG 9404 for whom induction therapy fails to achieve first remission
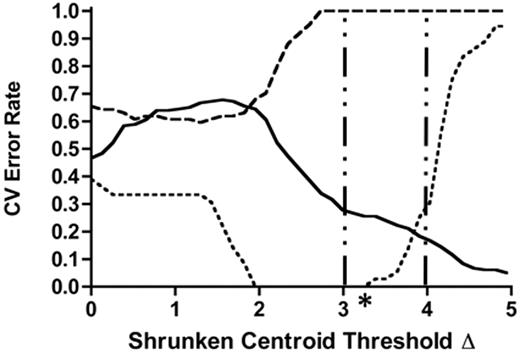
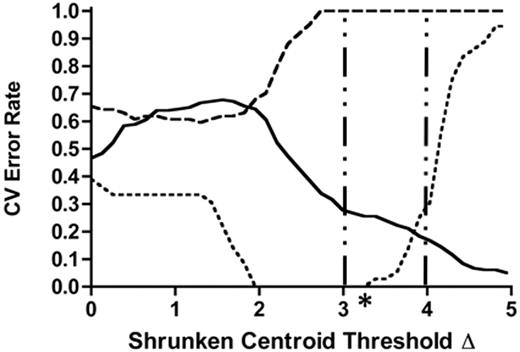
To identify a genomic classifier associated with treatment-dependent prognosis, we used RMA-normalized expression profiles from 50 COG 9404 patients who were divided into IF, RE, and CCR cohorts (Figure 1). Using PAM cross-validation (CV) least-discriminate analysis (LDA) error rates to distinguish gene sets having prognostic relevance for early treatment failure, we applied the following filtering criteria: a shrunken centroid threshold of at least 3.0, the lowest summative CV error rate across all outcome parameters, and the smallest gene set size identified by the first 2 filtering criteria (Figure 1). We found that the most efficient predictor of IF occurred at a shrunken centroid Δ of 3.25 (Table 3). Rank-ordered gene-set sizes that were greater or lesser than this threshold resulted in summative error rates that were larger than 1.25, and were no longer considered in the identification of a genomic classifier for IF. This prediction algorithm identified a 116-member genomic classifier in our COG 9404 training set that distinguished all 6 IF cases from 44 patients who achieved a first remission (Table 4; P < .001, 2-tailed Fisher exact test), but did not distinguish RE from CCR cases.
Identification of a genomic classifier for induction failure among 50 T-ALL samples. PAM cross-validation was used to identify genes that could distinguish patients who failed induction (⋯) from those who maintained a CCR for more than 4 years (—) and from those who relapsed after achieving remission (- -). A centroid level of 0 corresponds to 54 675 genes, ESTs, and probe sets, while a centroid level of 4.95 corresponds to 1 gene; the vertical bars indicate near-stable CV error rates between shrunken centroid threshold Δ values of 3.00 to 4.00. Asterisk indicates most efficient classifier.
Identification of a genomic classifier for induction failure among 50 T-ALL samples. PAM cross-validation was used to identify genes that could distinguish patients who failed induction (⋯) from those who maintained a CCR for more than 4 years (—) and from those who relapsed after achieving remission (- -). A centroid level of 0 corresponds to 54 675 genes, ESTs, and probe sets, while a centroid level of 4.95 corresponds to 1 gene; the vertical bars indicate near-stable CV error rates between shrunken centroid threshold Δ values of 3.00 to 4.00. Asterisk indicates most efficient classifier.
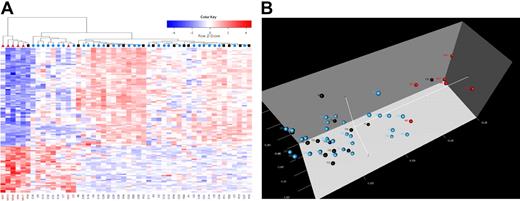
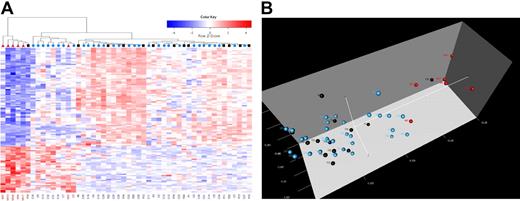
Because hierarchic clustering and principal component analyses can visually portray spatial distances in gene expression, albeit with less accuracy than nonvisual approaches, we next performed these analyses in our test set.40 We found that 5 IF cases clustered adjacent to each other, and 1 IF case clustered within a cohort of patients who have remained in CCR (Figure 2A). Interestingly, this one case differed from other IF cases for age (age of 4 years versus ages ≥ 9 years). Adjacent to the IF cases, one RE case appeared to have a similar genetic signature (Figure 2B). All IF, RE, and CCR cases were retained within this signature during subsequent analyses.
Cluster identification of patients for whom induction has failed in T-ALL. (A) Hierarchic clustering of samples (columns) and genes (rows) shows differential expression for patients in the IF (red triangle), RE (black square), and CCR (blue circle) cohorts. Red cells indicate high expression, and blue cells indicate low expression. (B) In 3-dimensional principal component analysis (PCA), 50 T-ALL samples were projected in the feature space based on differential expression for 116 probes. Each sphere represents a sample: red spheres denote IF patients, black spheres indicate RE postinduction remission, and blue spheres show patients who have remained in CCR for longer than 4 years. In panels A and B, the patients for whom induction failed clustered together, indicating a unique and shared genetic signature.
Cluster identification of patients for whom induction has failed in T-ALL. (A) Hierarchic clustering of samples (columns) and genes (rows) shows differential expression for patients in the IF (red triangle), RE (black square), and CCR (blue circle) cohorts. Red cells indicate high expression, and blue cells indicate low expression. (B) In 3-dimensional principal component analysis (PCA), 50 T-ALL samples were projected in the feature space based on differential expression for 116 probes. Each sphere represents a sample: red spheres denote IF patients, black spheres indicate RE postinduction remission, and blue spheres show patients who have remained in CCR for longer than 4 years. In panels A and B, the patients for whom induction failed clustered together, indicating a unique and shared genetic signature.
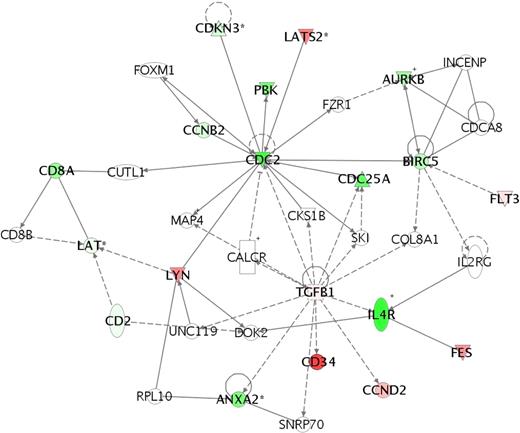
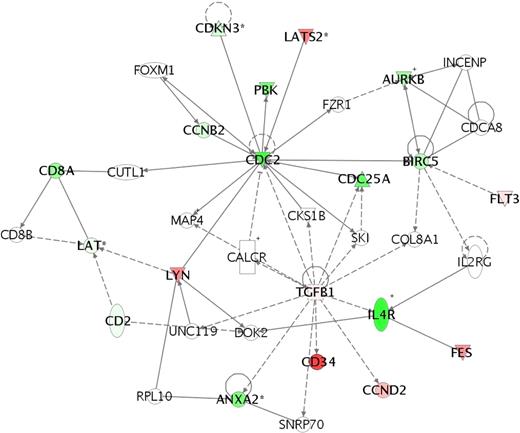
We identified 116 genomic classifiers (Table 5; Table S1) with diverse biologic roles. Because IF patients were the most genetically dissimilar from other outcome groups and to identify other gene pathways, we evaluated expression differences between the IF, RE, and CCR subsets. In comparison with RE and CCR cohorts, analysis of mean expression fluorescence values showed that 37 probe sets were upregulated and 79 were downregulated in the IF cohort. Identified within the 37 upregulated probes were genes with overlapping function in cytokine-cytokine receptor (TGFB1, TNFRSF18, and FLT3) and hematopoietic lineage differentiation (FLT3 and CD34) signaling pathways, cell cycle control (CCND2), and biosynthetic function (HK2, SPTLC2, and MGLL). In contrast, identified within the 79 downregulated probes were genes controlling T-cell differentiation (CD2, CD8A, and IL4R), junctional complexes (PARD3, MYH10, EPB41L2, and TJP2), and the G1/S checkpoint (CDC25A and CDC2). To evaluate whether specific molecular mechanisms might result in IF, we used the Ingenuity Pathways Knowledge Database to perform a network analysis on 70 “focus genes” within our 116-member classifier (Figure 3). Tissue growth factor beta 1 (TGFβ1), large tumor suppressor, homolog 2 (lats2), and v-yes-1 Yamaguchi sarcoma viral-related oncogene homolog (lyn) were identified as upregulated and functionally linked to CDC2 and CDC25 in arresting T-ALL cells at the G1/S transition. Notably, genes involved with apoptosis, proliferation, or drug metabolism were not identified, suggesting that T lymphoblasts within the IF cohort were in a state of cellular quiescence.
Network analysis of genetic pathways active in the IF cohort shows a checkpoint arrest at G1/S transition. To identify biologic function and interactions in the differentially expressed genes in the 116-member genomic classifier, we used Ingenuity Systems software to map gene networks and identifying potentially dysregulated pathways in the IF T-ALL cohort. Network analysis shows that TGFβ1, LYN, and LATS2 interact with and down-regulate CDC2 and CDC25 to result in a arrest of cell cycle progression at the G1/S checkpoint. Absent from this network analysis are genes governing drug metabolism (p450 enzymes) or apoptotic pathways. Each gene node represents a functional class, for which an upward-pointing triangle indicates a phosphatase, a downward-pointing triangle indicates a kinase, a vertical rectangle indicates a G-protein–coupled receptor, a square indicates a cytokine, and a circle indicates genes having other functions, to include surface receptors and adhesive ligands. Nodes that are colored in red indicate relative up-regulation, and nodes in green indicate relative down-regulation.
Network analysis of genetic pathways active in the IF cohort shows a checkpoint arrest at G1/S transition. To identify biologic function and interactions in the differentially expressed genes in the 116-member genomic classifier, we used Ingenuity Systems software to map gene networks and identifying potentially dysregulated pathways in the IF T-ALL cohort. Network analysis shows that TGFβ1, LYN, and LATS2 interact with and down-regulate CDC2 and CDC25 to result in a arrest of cell cycle progression at the G1/S checkpoint. Absent from this network analysis are genes governing drug metabolism (p450 enzymes) or apoptotic pathways. Each gene node represents a functional class, for which an upward-pointing triangle indicates a phosphatase, a downward-pointing triangle indicates a kinase, a vertical rectangle indicates a G-protein–coupled receptor, a square indicates a cytokine, and a circle indicates genes having other functions, to include surface receptors and adhesive ligands. Nodes that are colored in red indicate relative up-regulation, and nodes in green indicate relative down-regulation.
Comparison of genetic signatures between IF patients and drug-resistant cell lines identifies genes that are commonly altered in expression
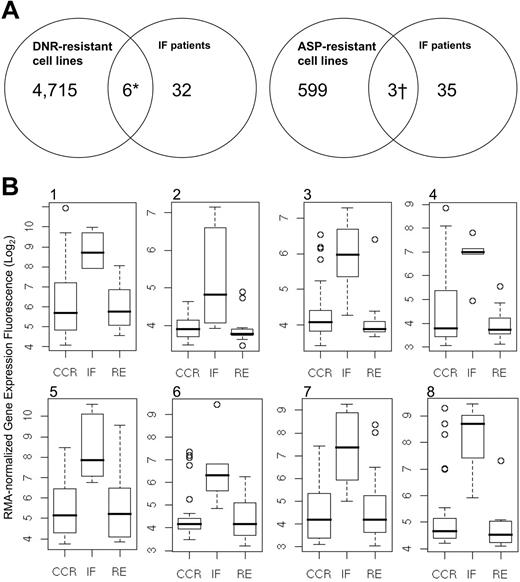
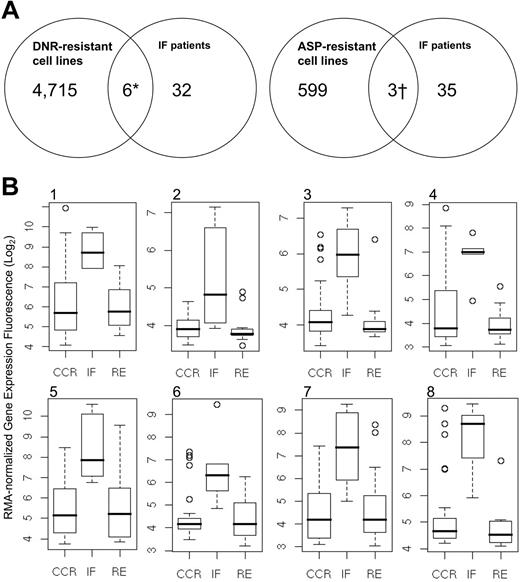
Because selective resistance to cytotoxic therapy can provide prognostically significant information,22,41,42 we developed cell lines having acquired drug resistance to DNR and ASP (Table 1). Nucleic acids from both Jurkat and Sup T1 cell lines, each of which had developed resistance to DNR and ASP, were harvested and hybridized to the U133 Plus 2.0 chip. Using a threshold of 1.5-fold or higher change in expression, earlier shown to correlate closely with quantitative reverse-transcription–polymerase chain reaction (QRT-PCR) measurement of mRNA levels,43,44 we performed intersection union testing between the DNR and l-asp–resistant cell lines and our 116-member genomic classifier (Figure 4; Figure S1). For genes that were commonly upregulated between the drug-resistant cell lines and the IF cohort, 6 classifiers were shared with both DNR-resistant cell lines, 3 classifiers were shared with both l-ASP–resistant cell lines, and one probe (FLJ39602) was shared between the DNR and l-ASP–resistant signatures (identified in Table 5 footnotes ‡ and ¶). Altogether, we identified 7 genes that were commonly upregulated between the drug-resistant cell lines and IF T-ALL cells (Table 6). We next developed box-plots comparisons for each gene within the IF, RE, and CCR cohorts (Figure 4Bi-viii). In all cases for upregulated genes, classifiers associated with IF patients were significantly more highly expressed than for the RE and CCR. Among the 79 downregulated probes, 2 classifiers were commonly shared with the DNR-resistant cell lines, and 7 were shared with ASP-resistant Jurkat and Sup T1 (identified in Table 5 footnotes ‡ and §; Figure S1). As in the upregulated gene set, classifiers that were downregulated within the IF cohort were significantly underexpressed in comparison with the RE and CCR patients (Figure S1A-I). Within the up- and downregulated gene sets, 5 ESTs were identified with functions that are currently unknown. Taken together, these data suggest that sets of genes are commonly up- and downregulated among T-ALL cell lines with acquired multidrug resistance and patients who did not enter a first remission.
Intersection union testing of genes up-regulated between IF patients and drug-resistant cell lines. (A) Differential expression between IF patients and drug-resistant cell lines identified 7 up-regulated genes. (B) Box-plot distributions for 8 up-regulated genomic classifiers, including 2 probes for LATS2: (i) 207761_s_at (DKFZ P586AD522), (ii) 216203_at (SPLTC2), (iii) 218618_s_at (FAD104), (iv) 218847_at (IMP-2), (v) 223380_s_at (LATS2), (vi) 226550_at (FLJ39602), (vii) 227013_at (LATS2), and (viii) 235824_at (EST). These data show differential expression in genes with shared upregulation in the IF cohort, compared with the RE and CCR subsets. Boxes indicate the range of data for each data set; vertical bars with horizontal lines represent the error bars associated with each data set.
Intersection union testing of genes up-regulated between IF patients and drug-resistant cell lines. (A) Differential expression between IF patients and drug-resistant cell lines identified 7 up-regulated genes. (B) Box-plot distributions for 8 up-regulated genomic classifiers, including 2 probes for LATS2: (i) 207761_s_at (DKFZ P586AD522), (ii) 216203_at (SPLTC2), (iii) 218618_s_at (FAD104), (iv) 218847_at (IMP-2), (v) 223380_s_at (LATS2), (vi) 226550_at (FLJ39602), (vii) 227013_at (LATS2), and (viii) 235824_at (EST). These data show differential expression in genes with shared upregulation in the IF cohort, compared with the RE and CCR subsets. Boxes indicate the range of data for each data set; vertical bars with horizontal lines represent the error bars associated with each data set.
Cross-validation of the genomic classifier in an independent T-ALL clinical trial identifies patients with progressive disease and early treatment failure
A recurring concern with expression profiles developed for a single study is that the genomic signature, however statistically significant, does not predict outcome classes in a different clinical trial.40 To address this concern, we next tested whether our 116-member genomic classifier could predict IF cases among 42 samples obtained from COG study 8704 study patients (Table 7), who share many karyotypic and demographic similarities with the cases tested from COG 9404 (Table 2). In this test set, bone marrow samples from patients with newly diagnosed T-ALL were hybridized to the Affymetrix U133A chip. Because only 85 probes of our original 116-member signature were shared between U133 Plus 2.0 and U133A chips, we used an approach described by others29,44 and again used PAM (Δ = 3.755) to constrain our classifier to the top 25 rank-ordered probes (identified as in Table 5). Interestingly, the 25-member classifier correctly identified 2 patients who continued to have a peripheral blood absolute blast count of more than 1000/μL for more than 14 days during the induction period (Table 7). A persistence of peripheral blood blasts has been previously reported as being prognostic of relapse in the 8704 study.14 The classifier correctly identified 1 of these 2 patients as the only induction failure in this case-control cohort, and the other patient with a slow disappearance of peripheral blood blasts relapsed at day of treatment 103. As in COG 9404, our classifier could not reliably differentiate between patients who entered remission but later relapsed from those who remained in a long-term CCR.
Discussion
Despite the recent identification that activating NOTCH1 mutations11-13 response to glucocorticoids6 and MRD determinants6,9,10 may allow risk stratification in T-ALL, there are no markers that reliably predict early treatment failure. To address this essential need, which is a necessary prerequisite in developing risk-adapted, individualized therapy, we identified a 116-member genomic classifier that distinguished patients who do not enter a first remission. To further explore the genetic mechanisms that may be responsible for IF, we developed 4 T-ALL cell lines, Jurkat and Sup T1, each with acquired resistance to DNR or ASP, and identified sets of genes that are commonly up- and downregulated. While others have used array profiling to investigate oncogenic pathways, molecular heterogeneity, and genomic features that distinguish T-ALL from other hematopoietic neoplasms,18,19,28,45-47 we are the first to develop a classifier that can predict IF, as well as guide further studies in drug-resistant T-ALL.
Of the 90% to 97% of patients with T-ALL who achieve a first remission, many are eventually cured of their disease,1,48,49 but among IF patients the mortality rate is nearly 90%.50 Unlike other investigators developing array-based classifiers,18,28 we were fortunate enough to have enough IF cases in this study to set them apart from RE and CCR patients, allowing us to highlight differences in these cohort-dependent expression profiles. Several IF patients in this study achieved remission with nelarabine, an ara-G prodrug that is efficacious against refractory T-ALL.51-53 Unlike other S phase–specific DNA nucleoside analogues, nelarabine selectively accumulates within T-ALL cells, and through upregulation of solubilized Fas ligand (sFasL) may induce S phase–independent cell death.54 The clonotypic elimination of T cells through the Fas-Fas ligand requires colocalization into lipid rafts55 along LAT56 and RasGRP,57 which we identified at rank-order positions 1 and 7 (LAT), and 4 (RasGRP) within our signature (Table 3). Based on these findings, and the observation that half of our IF patients achieved remission with nelarabine, we postulate that quiescent T lymphoblasts are subject to ara-G–induced S phase and non–S phase–dependent cell death. Through the development of a reliable genomic classifier for IF, we anticipate that specialized microarray platforms, gene cards, or flow-based assays of nucleic acid expression levels might further expedite the use of alternate treatment strategies for IF patients.
In the supervised classification approach used in PAM, support vector machine (SVM) and other computational tools, an algorithm optimizes a classifier to best fit a set of available cases for which a parameter has been predefined. Although others have investigated whether expression profiles might distinguish outcome in T-ALL,18,28,29 such studies based their predictions on fluorescence intensity alone, or did not test whether their classifier was predictive in an independent dataset and microarray platform. To address these issues, we used PAM to cross-validate the top 25 probes of our classifier in the COG 8704 test set, which was developed independently on a smaller chip. This classifier correctly identified 2 patients who continued to circulate peripheral blood absolute blast counts of more than 1000/μL for more than 14 days after starting induction. Both the speed and initial response to induction therapy have prognostic significance in T-ALL,14 6,58,59 and a persistence of peripheral blood blasts during induction was significantly associated with treatment failure in the 8704 study.14 One of these 2 patients was diagnosed with IF, and while the other achieved remission by day 29, he relapsed at day 103. Our cross-validation approach strengthens our findings that rare IF patients can be accurately identified with expression profiling, despite significant differences between induction regimens.
Although others have developed microarray signatures that can distinguish postinduction RE from CCR groups in precursor B-ALL,18,60,61 we and others have not been able to readily detect differences in signatures between these outcome groups in T-ALL. Using a variety of treatment protocols, array platforms, and computational approaches for class discovery in T-ALL, Yeoh et al,18 Chiaretti et al,29 and Ferrando et al28 have used microarrays to develop biomarkers associated with risk for relapse. Using the Affymetrix HG U95Av2 array (12 600 probes) and SVM, Yeoh et al found 7 genes that could distinguish between 8 RE and 26 CCR patients who were treated on St Jude Treatment studies TXIIIA and B. However, other investigators, including us, have not subsequently identified any of these same genes in patients who received different treatment approaches.28,29,62 While Ferrando et al did not study IF cases, they reported that the aberrant activation of LYL1, HOX11, and TAL1 results in leukemic arrest at specific stages of normal thymocyte development, and that each of these biomarkers had prognostic significance as well as a specific expression profile. Interestingly, while we and others29 did not identify LYL, HOX11, or TAL1 as having prognostic significance, we found that upregulation of CD34, Cyclin D2, and FES within our IF cohort was also overexpressed for patients who had LYL-mediated oncogenic arrest and primary drug resistance.28 Chiaretti et al profiled expression signatures in 51 adults treated on the Italian GIMEMA 0496 trial, and developed a 3-gene model, consisting of AHNAK, IL-8, and CD2, that could distinguish outcome.29 Our signature was largely dissimilar from that developed by Chiaretti et al, but shared CD2 as having prognostic importance. Although CD2 antigen expression has been reported to be predictive of EFS in T-ALL,29,63,64 we and others could not discriminate outcome in COG 8704 based on CD2 expression.2 While it is tempting to compare the shared identification of specific genes against other microarray studies, important differences in study design, treatment effect, patient demographics, and nonoptimized analysis of data may introduce significant confounding factors to these correlates.1,37,65 For these reasons, additional prospective testing of our and other classifiers is necessary in optimizing the efficacy of microarrays in risk assignment.
Several recent studies have suggested that cellular quiescence may be mechanistically linked with intrinsic drug resistance to multiagent therapy for both solid and liquid tumor cells.66,67 In support of this change in paradigm, we found that T lymphoblasts in patients with IF may have a genetic profile that suggests they resist multiagent therapy though cellular quiescence. A number of genes that control the G1/S transition in cell cycle progression, specifically TGFβ1, LYN, and LATS2, were upregulated (Figure 3). The Src family member lyn, growth regulatory factor TGFβ1, and putative tumor suppressor LATS2 have been shown to suppress CDC2- and CDC25-mediated cell cycle progression and apoptotic pathways in human T cells and hematopoietic progenitors.68-70 Through normal or deregulated signaling, we hypothesize that in IF patients these growth modulators maintain T lymphoblasts in a quiescent, nonproliferating state, in which the S phase–specific interference of cell cycle progression by DNR, l-ASP, VCR, and prednisone is either reduced or abrogated.
Relapse occurs with the acquired activation of cell survival pathways, to include ATP binding cassette (ABC) transporters, or enzymes that control nucleic acid biosynthesis, as described for DNR71-73 and ASP.74-76 To address the possibility that the genes mediating acquired drug resistance might also be active in IF patients, we developed Jurkat and Sup T1 T-ALL cell lines, each with resistance to DNR and ASP, but also to VCR and PRED, as in the case of DNR-resistant Jurkat cells. Our goal was to identify whether overlap exists between intrinsic and acquired multidrug resistance in T-ALL. Consistent with the findings of others, ABC transporters and genes regulating asparagine biosynthetic enzymes were not found to be specifically upregulated within our early treatment failure group.77-79 We instead identified fold increases for serine palmitoyltransferase (SPTLC2), monoglyceride lipase (MGLL), LYN, and TGFB1, all of which may participate in cell homing and migration. The role of cellular trafficking remains poorly understood in lymphoblastic leukemia, but may contribute to relapse in sanctuary sites or protection against cytotoxic agents.67,80 In the context of enhanced mobility and tissue compartment penetration, quiescent cells may not only escape S phase–dependent cytotoxicity, but also may dynamically upregulate drug efflux transporters and other pathways that are necessary for survival.40,81,82
Using standard light microscopy, IF can be easily identified in postdiagnostic bone marrow samples, but usually only after several weeks of treatment. This time lag is problematic, as patients who fail induction are likely to develop infectious complications and become refractory to salvage therapy. Alternatively, a genomic classifier might identify IF patients within a few days of diagnosis, sparing them the toxicities of ineffective therapy, and provide opportunities to pursue novel treatments, possibly in combination with stem cell transplantation. When used in combination with other risk-stratifying features, genomic classifiers may refine risk group assignment in T-ALL49,83,84 and allow the development of antineoplastic drugs that are not mechanistically tied to cell cycle progression.
The online version of this article contains a data supplement.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Acknowledgments
This work was supported in part by R01 CA114589–01, R21 CA098251–01 (R.S.L.); NIH/NCI U10 CA98543–03-14305, The Parents Leukemia Trust Fund of New Mexico (S.S.W.), and NIH/NCI U10 CA29139 (M.D.).
We wish to thank Mr Stephen Linda, COG statistician, for providing correlative data from the 8704 and 9404 T-ALL clinical trials, and Drs William Carroll and Stephen Hunger for providing T-ALL samples from the COG ALL Cell Bank Repository. The expression profiling experiments were performed using the facilities and services provided by the Keck-UNM Genomics Resource, a facility supported by a grant from the W. M. Keck Foundation and the State of New Mexico, the UNM Cancer Research and Treatment Center, and the New Mexico Center for Environmental Health Science.
National Institutes of Health
Authorship
Contribution: S.S.W. identified main hypotheses, oversaw microarray quality assurance, kept correlative patient databanks, and was chiefly responsible for writing this paper; Z.J. oversaw all computational aspects of statistics and data analysis; H.K. performed the technical aspects of microarray hybridization, analysis, and data management; T.G. provided insights into the clinical significance of persistent disease; M.D. is the lead statistician for the COG ALL studies and contributed to the 9404 study design as well as patient profiling case selection; B.L.A. is the overall prinicipal investigator of COG 9404 and provided insights into the biologic significance of induction failure; and R.S.L. is the overall principal investigator for the principal grants that funded this project.
S.S.W. and Z.J. contributed equally to this work.
A complete list of the members of the Children's Oncology Group is provided in Document S1, available on the Blood website; see the Supplemental Materials link at the top of the online article.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Stuart S. Winter, Pediatric Hematology-Oncology, MSC 10 5590, 1 University of New Mexico, Albuquerque, NM 87131-5311; e-mail: swinter@salud.unm.edu.