Abstract
In acute myeloid leukemia (AML), aberrant expression and mutations of transcription factors have been correlated with disease outcome. In the present study, we performed expression and mutation screening of GATA2, which is an essential transcription factor for regulation of myeloid lineage determination, in de novo pediatric AML patients. GATA2 mutations were detected in 5 of 230 patients, representing a frequency of 2.2% overall and 9.8% in cytogenetically normal AML. GATA2 expression analysis demonstrated that in 155 of 237 diagnostic samples (65%), GATA2 expression was higher than in normal BM. In complete remission, normalization of GATA2 expression was observed, whereas GATA2 expression levels stayed high in patients with resistant disease. High GATA2 expression at diagnosis was an independent poor prognostic factor for overall survival (hazard ratio [HR] = 1.7, P = .045), event-free survival (HR = 2.1, P = .002), and disease-free survival (HR = 2.3, P = .004). The prognostic impact of GATA2 was particularly evident in specific AML subgroups. In patients with French-American-British M5 morphology, inv(16), or high WT1 expression, significant differences in survival were observed between patients with high versus normal GATA2 expression. We conclude that high GATA2 expression is a novel poor prognostic marker in pediatric AML, which may contribute to better risk-group stratification and risk-adapted therapy in the future.
Introduction
Acute myeloid leukemia (AML) is a clinically and genetically heterogeneous disease that accounts for 15%-20% of childhood leukemia. Over the past decades, the prognosis for children with AML has improved considerably as a result of better (risk-adapted) therapeutic strategies. Currently, approximately 60%-70% of pediatric AML patients experience long-term survival.1,2 Current risk-group classification is based mainly on cytogenetic aberrations and response to induction chemotherapy.1 In addition, several molecular aberrations (acquired gene mutations and altered gene expression) that are correlated with clinical outcome have been identified in pediatric and adult AML. For various molecular aberrations, differences have been observed in the prevalence and prognostic significance between pediatric and adult AML.1,3,4 For the assessment of treatment response and early detection of relapse, monitoring of minimal residual disease (MRD) is important.5,6 Specific gene rearrangements and mutations can be used for MRD monitoring, but suitable MRD markers are lacking in the majority of AML patients.6
Genes that have been implicated in leukemogenesis are usually involved in the regulation of cellular survival, proliferation, and hematopoietic differentiation.7 These processes are tightly regulated by various transcription factors. The GATA-binding protein 2 (GATA2) gene, located at chromosome 3q21, encodes for a zinc-finger transcription factor that plays an essential role during the development and differentiation of hematopoietic cells. GATA2 knockout mice have profound defects in definitive hematopoiesis, resulting in embryonic lethality at day 10-11 of gestation due to a pan-hematopoietic deficit.8 The expression and function of GATA2 in hematopoietic cells depends largely on their differentiation status and is tightly regulated. GATA2 is expressed broadly in hematopoietic cells, with particularly high expression in hematopoietic stem and progenitor cells and early erythroid cells, megakaryocytic cells, and mast cells.9-14 In hematopoietic stem and progenitor cells, GATA2 functions as an important regulator of proliferation, differentiation, and survival and acts as a regulator of stem cell quiescence.15-19 During myeloid differentiation, GATA2 plays an important role in the regulation of lineage determination. GATA2 blocks terminal erythroid, monocytic, and granulocytic differentiation, and also promotes erythroblast proliferation, megakaryopoiesis, and mast cell formation.19-22 Tight regulation of GATA2 expression is essential in the process of myeloid lineage determination. Enforced expression of GATA2 in myeloid progenitors induces down-regulation of the crucial myeloid transcription factors PU.1 and CCAAT/enhancer binding protein-α (CEBPA), which are implicated in terminal granulocytic and monocytic differentiation.17,21,23,24 Moreover, GATA2 inhibits the ability of PU.1 to transactivate critical myeloid target genes and thereby prevents the induction of a myeloid gene expression program.25 Conversely, knockdown of GATA2 in myeloid progenitors results in significant up-regulation of the expression of PU.1 and CEBPA,20,21 which allows terminal granulocytic and monocytic differentiation to proceed.19
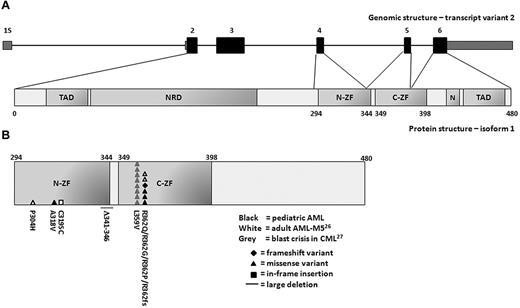
In sporadic and familial myeloid malignancies, acquired and inherited mutations in the highly conserved zinc-finger domains of the GATA2 gene have been described recently14,26-35 (Figure 1B and supplemental Figure 1, available on the Blood Web site; see the Supplemental Materials link at the top of the online article). These 2 zinc fingers play an essential role in DNA-binding and protein-protein interactions of GATA2 with target genes. During blast crisis in chronic myeloid leukemia (CML), 2 acquired GATA2 mutations were found in 9 of 85 (10%) patients.27 In adult monocytic AML (French-American-British M5 [FAB-M5]), 3 acquired GATA2 mutations were detected in 4 of 112 (3.6%) patients.26 In 3 other adult AML cohorts (N = 50, N = 225, and N = 268), no acquired mutations in the GATA2 coding sequence were found.14,28,29 So far, GATA2 mutations have not been reported in pediatric AML. In the present study, we investigated the relevance and prognostic value of GATA2 expression and mutations in a large cohort of pediatric AML patients.
Methods
Patient samples
Viable frozen bone marrow (BM) or peripheral blood samples taken at diagnosis from 261 children with de novo AML were provided by the Dutch Childhood Oncology Group (DCOG; The Hague, The Netherlands), the AML-Berlin-Frankfurt-Münster (BFM) study group (Hannover, Germany, and Prague, Czech Republic), and the Saint-Louis Hôpital (Paris, France). As controls, 18 BM samples form healthy donors and 62 BM samples from patients with AML (n = 42) or acute lymphoblastic leukemia (n = 20) in long-term complete remission (CR) were included. For a subgroup of 38 patients, follow up BM samples were provided by DCOG on the basis of available material for MRD analysis, including 10 paired diagnostic-relapse samples. The majority of the processed diagnosis samples (248 of 261) contained more than 80% leukemic cells as assessed morphologically on May-Grünwald-Giemsa–stained cytospin slides.
Mononuclear cells were isolated from BM or peripheral blood and contaminating nonleukemic cells were eliminated as described previously.36 Genomic DNA and total cellular RNA were extracted from at least 5 × 106 leukemic cells using TRIzol reagent (Invitrogen), RNA-Bee (Iso-Tex Diagnostics), or the GeneElute Mammalian Total RNA isolation kit (Sigma-Aldrich). Informed consent was obtained after institutional review board approval according to local laws and regulations and in accordance with the Declaration of Helsinki. Each national study group performed central review of the morphologic classification according to the FAB classification system, cytogenetic classification, and clinical follow-up.
Survival analysis was restricted to a subset of 205 pediatric AML patients who were treated according to the BFM and Dutch pediatric AML protocols (studies AML-BFM 98, AML-BFM 04, DCOG-BFM 87, DCOG 92/94, and DCOG 97). The median follow-up time for survivors was 4.6 years (range, 0.4-22.7). In the total cohort, 4-year probability for overall survival (pOS; time between diagnosis and death from any cause) was 60% ± 4%; probability for event-free survival (pEFS; time between diagnosis and first event) was 41% ± 4%; and probability for disease-free survival (pDFS; time between achieving CR and relapse) was 53% ± 4%.
Cytogenetic and molecular analysis
Leukemic samples were routinely investigated for cytogenetic aberrations using standard chromosome banding analysis by the national study groups. Leukemic samples were further analyzed for recurrent nonrandom genetic aberrations characteristic of AML as described by the World Health Organization (2008) classification of myeloid neoplasms and acute leukemia,37 including t(15;17)(q21;q22), inv(16)(p13q22), t(8;21)(q22;q22), and 11q23 translocations, using FISH and/or RT-PCR. 11q23 translocations were confirmed by RT-PCR or long-distance inverse PCR.38
Detection of molecular aberrations included mutation analysis of Wilms tumor 1 (WT1), nucleophosmin 1 (NMP1), CEBPA, FMS-related tyrosine kinase 3 internal tandem duplication (FLT3-ITD), NRAS, KRAS, PTPN11, KIT, and MLL-PTD, as described previously.4 If positive for MLL-PTD, this was confirmed by a multiplex ligation–dependent probe amplification analysis.39 High expression of EVI1 was established previously by gene-expression profiling and real-time quantitative PCR.40
Sequence analysis of GATA2
Sequence analysis of GATA2 was performed in 230 de novo pediatric AML patients with more than 80% blasts in their BM and for whom genomic DNA was available. Because all acquired GATA2 mutations in myeloid malignancies so far were present within the DNA-binding zinc-finger domains encoded by exon 4, 5, and 6 (Figure 1), we performed Sanger sequencing of the coding region of these exons and their associated splice sites. Genomic DNA was PCR amplified and direct sequencing of the purified PCR products was performed unidirectionally using the described primers (supplemental Table 1). The sequence data were analyzed using Variant Reporter (Applied Biosystems). Mutations were confirmed by an independent amplification of the fragment and direct sequencing of both strands.
Microarray-based gene-expression profiling
Gene-expression profiling data (Affymetrix HGU133 Plus 2.0 microarray) were available from 237 patients.41 The data files are available in the Gene Expression Omnibus repository (accession number GSE17855). Probe set intensities were normalized using the variance stabilization normalization (VSN) procedure (Bioconductor Package VSN). Details regarding RNA quality, microarray processing, data acquisition, and data normalization have been described previously.41
Three probe sets on the Affymetrix Human Genome U133 Plus 2.0 Array were annotated to the GATA2 gene (209710_at, 210358_ × _at, and 207954_at). All probe sets are located within the 3′ untranslated region of exon 6 in regions common for all isoforms of GATA2. VSN-normalized expression values of GATA2 obtained by microarray were validated with real-time quantitative PCR in a subset of patients (n = 44). VSN-normalized expression values of probe set 209710_at showed a good correlation with GATA2 expression values obtained by real-time quantitative PCR (Spearman correlation coefficient = 0.73, P < .00001), which enabled us to define a conversion factor (VSN-normalized expression = 1222.3*Q-PCR expression0.2955). GATA2 expression was considered to be high when the expression was 2 SDs above the median GATA2 expression obtained in normal BM as determined by microarray (n = 6) or real-time quantitative PCR (n = 74). Real-time quantitative GATA2 expression values obtained in normal BM (n = 74) were converted into VSN-normalized expression values using the conversion factor described in this section. The cutoff value for high GATA2 expression was determined to be 320 arbitrary units (AU).
Expression of WT1 was analyzed using VSN-normalized expression values of probe set 206067_s_at on the Affymetrix Human Genome U133 Plus 2.0 Array, which is annotated to the WT1 gene. Previously, we showed a good correlation with WT1 expression values obtained by real-time quantitative PCR.42 In the present study, we could define a conversion factor (VSN-normalized expression = 523.86*Q-PCR expression0.1613). WT1 expression was considered to be high when the expression was 2 SDs above the median WT1 expression obtained in normal BM as determined by microarray (n = 6) or real-time quantitative PCR (n = 69). Real-time quantitative WT1 expression values obtained in normal BM (n = 69) were converted into VSN-normalized expression values using the conversion factor described in this section. The cutoff value for high WT1 expression was determined to be 143 AU.
Real-time quantitative PCR
For quantification of GATA2 expression, a TaqMan Gene Expression Assay (Hs_00231119_m1; Applied Biosystems) was used. For quantification of WT1 expression, we used the European LeukemiaNet WT1 assay that was developed previously in our laboratory43 and was recently evaluated in normal control and AML patient samples by the European LeukemiaNet.44 For quantification of EVI1 expression, we used a forward primer (5′-GGTCAACAAACCAATTTAGACAGACA-3′), reverse primer (5′-TTCAGAATGAGGCGACGATGT-3′), and probe (5′-ATGGGAACATGTCCGGTAC-3′). For quantification of the AML1-ETO fusion-gene expression, we used a forward primer (5′-CCACAGAGCCATCAAAATCACA-3′), reverse primer (5′CAGCCTAGATTGCGTCTTCACAT-3′), and probe (5′-TGAGAAGCACTCCACAAT-3′). As a reference gene, we used GAPDH (premixed assay 4326317E; Applied Biosystems) or PBGD (forward primer: 5′-GCGGAGCCATGTCTGGTAA-3′; reverse primer: 5′-GGGTACCCAACGCGAATCAC-3′; probe: 5′-CTCATCTTTGGGCTGTTTTCTTCCGCC-3′). Samples were considered eligible for testing only when the threshold cycle value (Ct) of the reference gene GAPDH was lower than 26. Quantitative PCR reactions contained 2.5-5.0 μL of cDNA (10 ng/μL), 1.25 μL of premixed assay, or 0.75 μL of forward primer (10 pmol/μL), 0.75 μL of reverse primer (10 pmol/μL), and 0.35-0.5 μL of probe (10 pmol/μL). The 25-μL reactions were incubated in a 96-well plate for 2 minutes at 50°C, 10 minutes at 95°C, followed by 45 cycles of 15 seconds at 95°C, and 1 minute at 60°C. Quantitative PCR reactions were performed on the ABI/PRISM 7900 Sequence Detection system.
Statistical methods and analysis
To compare categorical variables, χ2 analysis and the Fisher exact test for small patient numbers were used. The nonparametric Mann-Whitney U test was applied for continuous variables. Correlations between continuous variables were calculated using the Spearman correlation coefficient.
Analysis of the FAB subgroup AML-M3 was restricted to patients harboring a translocation t(15;17)(q21;q22). Patients with rare recurrent cytogenetic aberrations such as t(7;12)(q36;p16), t(6;9)(p23;q34), t(16;21)(p11;q22), t(8;16)(p11;p13), monosomy 7, trisomy 8, and complex karyotype were classified as 1 cytogenetic subgroup entitled “rare cytogenetics.”
To assess outcome, the following parameters were used: CR rate, resistant disease (RD; defined as failure to achieve CR after ≤ 2 cycles of chemotherapy, excluding patients with early death), pOS (defined as time between diagnosis and death from any cause), pEFS (defined as time between diagnosis and first event, including relapse, death by any cause, second malignancy, or failure to achieve CR after ≤ 2 cycles of chemotherapy (included as an event on day 0), and pDFS (defined as time between achieving CR and relapse; only patients who achieved CR were included). pOS, pEFS, and pDFS were estimated by the Kaplan-Meier method and compared using the log-rank test. Two different analyses were performed according to the selected population: on the overall population and on the different morphological, cytogenetic, and molecular subgroups. The independence of prognostic factors was examined by multivariate Cox regression analysis, including in pediatric AML–established prognostic factors such as age, WBC count at diagnosis, favorable karyotype, NPM1 mutation, CEBPA double mutation (CEBPA-DM), and FLT3-ITD.45 Age and WBC count were included as continuous variables. Favorable karyotype was defined as patients harboring t(15;17)(q21;q22), inv(16)(p13q22), or t(8;21)(q22;q22). The results of univariate Cox regression analysis of the established prognostic factors are included in supplemental Table 4. P <.05 was considered statistically significant (2-tailed testing). All analyses were performed with SPSS Version 16.0 or 18.0 software.
Results
Genomic variations in the zinc-finger domains of the GATA2 gene
Sequence analysis of GATA2 was performed in 230 de novo pediatric AML patient samples. In 9 patients, a synonymous alteration (conversion of GCG to GCA) was observed within codon 411 of the coding sequence of exon 6, which is a known polymorphism (dbSNP release 132). A novel synonymous alteration was observed in codon 471 of exon 6 (supplemental Table 2). Nonsynonymous GATA2 variations were found in 5 of 230 (2.2%) pediatric AML patients (Figure 1B and Table 1). No correlation was observed between the presence of these variations and the expression levels of GATA2. The sequence abnormalities included 4 distinct heterozygous missense variants and 1 heterozygous single nucleotide deletion. In 1 patient, a heterozygous missense variant was detected in exon 4, leading to a A318V substitution. Neither this variant nor the affected codon has been described previously. The other 3 heterozygous missense variants and the single nucleotide deletion were present within codon 362 of exon 5. The 3 heterozygous missense variants resulted in 3 distinct amino acid substitutions (R362Q, R362G, and R362P). The single nucleotide deletion resulted in a frameshift with premature termination. Because neither germline nor remission material was available, the origin of the variations (acquired or inherited) could not be determined in the present study.
Sequence variations in the zinc-finger domains of GATA2 in myeloid malignancies. (A) Schematic representation of the human GATA2 gene and protein. The GATA2 gene consists of 6 exons. The coding exons (exon 2-6) are depicted in black and the 5′ and 3′ untranslated regions are depicted in gray. The GATA2 gene has 3 transcript variants that encode for 2 different protein isoforms with the same translation start site in exon 2. Isoform 1, encoded by transcript variant 1 or 2, contains 480 amino acids (50.5 kDa). Isoform 2, encoded by transcript variant 3, contains a smaller fifth exon resulting in a protein of 466 amino acids (49.1 kDa). Transcript variant 2, in which transcription is initiated from a distal first exon (1S), is specific for hematopoietic and neuronal cells. The encoded protein has 2 transactivation domains (TAD), a negative regulatory domain (NRD), a nuclear localization signal (N), and 2 zinc-finger domains (ZF). (B) Acquired mutations and sequence variations in the N-terminal (N-ZF) and C-terminal (C-ZF) zinc-finger domain of GATA2 in myeloid malignancies. Sequence variations in pediatric AML from the present study are depicted in black. Previously reported mutations in adult AML with FAB-M5 morphology or in blast crisis of CML are depicted in white and gray, respectively.26,27 Each symbol represents an independent GATA2 mutation or variation. Numbers indicate the amino acid affected by each mutation or variation. The type of mutation is indicated by the symbol.
Sequence variations in the zinc-finger domains of GATA2 in myeloid malignancies. (A) Schematic representation of the human GATA2 gene and protein. The GATA2 gene consists of 6 exons. The coding exons (exon 2-6) are depicted in black and the 5′ and 3′ untranslated regions are depicted in gray. The GATA2 gene has 3 transcript variants that encode for 2 different protein isoforms with the same translation start site in exon 2. Isoform 1, encoded by transcript variant 1 or 2, contains 480 amino acids (50.5 kDa). Isoform 2, encoded by transcript variant 3, contains a smaller fifth exon resulting in a protein of 466 amino acids (49.1 kDa). Transcript variant 2, in which transcription is initiated from a distal first exon (1S), is specific for hematopoietic and neuronal cells. The encoded protein has 2 transactivation domains (TAD), a negative regulatory domain (NRD), a nuclear localization signal (N), and 2 zinc-finger domains (ZF). (B) Acquired mutations and sequence variations in the N-terminal (N-ZF) and C-terminal (C-ZF) zinc-finger domain of GATA2 in myeloid malignancies. Sequence variations in pediatric AML from the present study are depicted in black. Previously reported mutations in adult AML with FAB-M5 morphology or in blast crisis of CML are depicted in white and gray, respectively.26,27 Each symbol represents an independent GATA2 mutation or variation. Numbers indicate the amino acid affected by each mutation or variation. The type of mutation is indicated by the symbol.
Patients who harbored nonsynonymous GATA2 variations were classified morphologically as FAB-M1 (n = 1), FAB-M2 (n = 1), or FAB-M4 AML (n = 3; Table 1). The nonsynonymous GATA2 variants were mainly present in cytogenetically normal AML (CN-AML) with a frequency of 9.8% (4 of 41). In addition, we detected a nonsynonymous GATA2 variation in a patient with monosomy 7. No GATA2 variations were found in the favorable cytogenetic subgroup (n = 67). In all cases, we observed cooccurrence of GATA2 variations with various other recurrent mutations, including NRAS mutations (n = 4), NPM1 mutations (n = 2), CEBPA-DM (n = 2), and WT1 mutations (n = 1). The outcome of patients with nonsynonymous GATA2 variations is described in Table 1. Because of the low frequency of nonsynonymous GATA2 variants in this pediatric AML cohort, prognostic implications of GATA2 variations could not be determined.
GATA2 expression in morphological and genetic AML subgroups
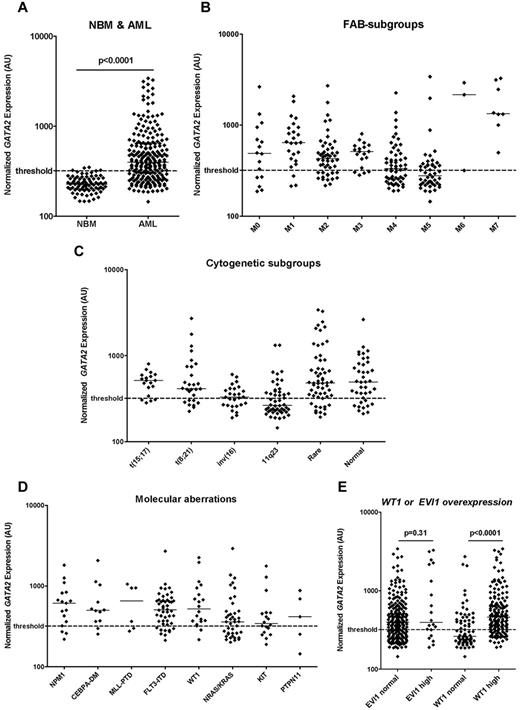
In 237 de novo pediatric AML patients, microarrays were used to determine the expression of GATA2 at diagnosis. In these AML samples, the expression of GATA2 varied more than 20-fold (range, 145-3405 AU; median, 379 AU). In 80 normal BM samples, a relatively homogeneous GATA2 expression was observed (range, 146-349 AU; median, 226 AU). The GATA2 expression was significantly higher in AML compared with normal BM (P < .0001; Figure 2A). GATA2 expression was not related to sex (P = .22), age (P = .52), or WBC count (P = .33; supplemental Figure 2).
GATA2 expression in normal BM and different morphologic, cytogenetic, and molecular AML subgroups. (A) Normalized expression levels of GATA2 in pretreatment samples of 237 children with de novo AML and 80 normal BM samples. Expression was significantly higher in AML compared with normal BM. Normalized expression levels of GATA2 in different morphological FAB subgroups (B), in cytogenetic subgroups (C), and in molecular subgroups including molecular aberrations (D) and AML with WT1 or EVI1 overexpression (E). GATA2 expression was determined using gene-expression arrays (Affymetrix HGU133 Plus 2.0 microarray, probe set 209710_at) or real-time quantitative PCR. Median values are depicted by the horizontal lines. The Mann-Whitney U test was used to compare expression levels between groups. The threshold for high versus normal GATA2 expression is based on median GATA2 in normal BM + 2 SD and is depicted as a dashed line.
GATA2 expression in normal BM and different morphologic, cytogenetic, and molecular AML subgroups. (A) Normalized expression levels of GATA2 in pretreatment samples of 237 children with de novo AML and 80 normal BM samples. Expression was significantly higher in AML compared with normal BM. Normalized expression levels of GATA2 in different morphological FAB subgroups (B), in cytogenetic subgroups (C), and in molecular subgroups including molecular aberrations (D) and AML with WT1 or EVI1 overexpression (E). GATA2 expression was determined using gene-expression arrays (Affymetrix HGU133 Plus 2.0 microarray, probe set 209710_at) or real-time quantitative PCR. Median values are depicted by the horizontal lines. The Mann-Whitney U test was used to compare expression levels between groups. The threshold for high versus normal GATA2 expression is based on median GATA2 in normal BM + 2 SD and is depicted as a dashed line.
In patients with FAB-M4 (n = 56) or FAB-M5 (n = 48) morphology, the median GATA2 expression was lower than in patients with other FAB subtypes (P = .019 and P < .0001, respectively), whereas higher expression was observed in patients with FAB-M1 (n = 25) or FAB-M7 (n = 8) morphology (P = .001 and P < .0001, respectively; Figure 2B). In cytogenetic subgroups, GATA2 expression was lower in patients with 11q23 translocations (n = 50) or inv(16) (n = 27; P < .0001 and P = .012, respectively), whereas higher GATA2 expression levels were observed in patients with CN-AML (n = 40) or rare cytogenetic aberrations (n = 58; P = .039 and P = .003, respectively; Figure 2C). No particular rare cytogenetic aberrations were associated with high levels of GATA2 expression. In addition, no chromosomal abnormalities involving the GATA2 locus (3q21) or located close to the GATA2 locus (eg, 3q26 aberrations) were present. No evident correlation between GATA2 expression levels and specific 11q23 translocation partners was observed. In the molecular subgroups, we observed that the median GATA2 expression was higher in patients with FLT3-ITD (n = 48), NPM1 (n = 17), CEBPA (n = 16), or WT1 mutations (n = 20; P = .003, P = .046, P = .02, and P = .035, respectively; Figure 2D-E) compared with the patients with wild-type FLT3, NPM1, CEBPA, or WT1, respectively. When CEBPA single mutants (n = 4) and double mutants (n = 12) were analyzed as 2 distinct entities, a trend toward higher GATA2 expression was observed in both groups compared with patients with wild-type CEBPA (P = .11 and P = .05, respectively). The expression of GATA2 showed a positive correlation with expression of the transcription factor WT1 (Spearman correlation coefficient = 0.51, P < .0001), which is known to be highly expressed in several hematopoietic malignancies including AML and has been identified previously as a molecular marker associated with disease outcome in AML.44,46,47
High GATA2 expression is an independent poor prognostic factor
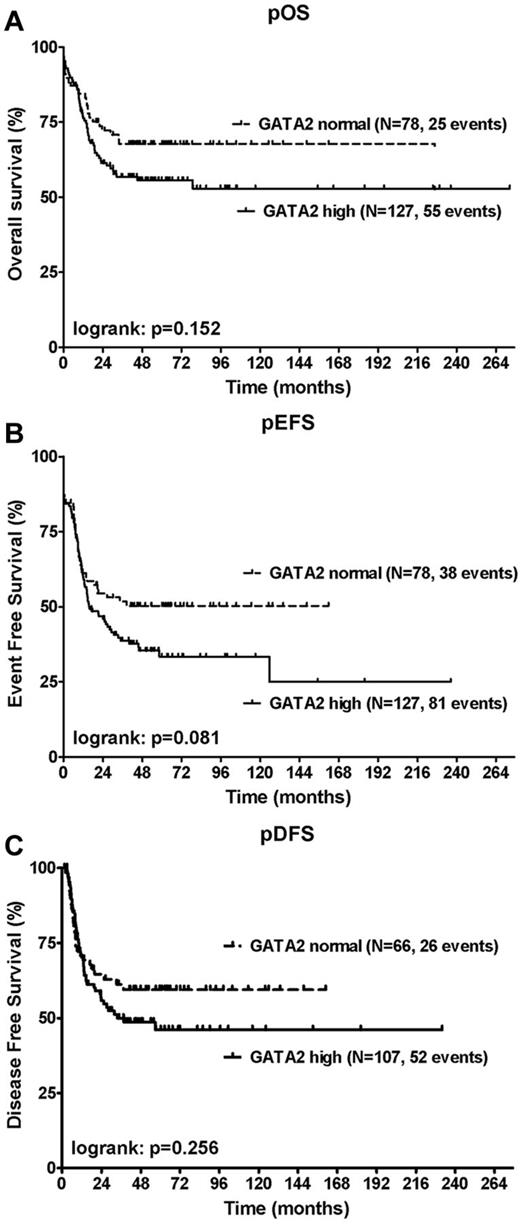
To determine the clinical significance of GATA2 expression in pediatric AML, we studied the relation between GATA2 expression at diagnosis and parameters of long-term outcome (pOS, pEFS, and pDFS). Because the level of expression of GATA2 appears to be a continuum, we defined a threshold for high versus normal GATA2 expression based on the expression of GATA2 in normal BM (threshold: median expression in normal BM + 2 SD). In 155 patients (65%) GATA2 levels were expressed above this threshold of 320 AU (range, 320-3405; median, 506 AU). The remaining 82 patients (35%) were classified as having normal GATA2 levels (range, 145-319 AU; median, 250 AU; Table 2).
Survival data were available for 205 patients, including 127 patients with high GATA2 expression (62%). High GATA2 expression was associated with worse pOS and pEFS, although the differences did not reach statistical significance at the P = .05 level in univariate analysis. No effect on pDFS was observed. The 4-year pOS was 56% ± 5% for patients with high GATA2 expression (n = 127) compared with 68% ± 6% for patients with normal GATA2 levels (n = 78; P = .15; Figure 3A); the 4-year pEFS was 35% ± 5% compared with 50% ± 6% (P = .08; Figure 3B) and the 4-year pDFS was 49% ± 5% compared with 59% ± 6% (P = .26; Figure 3C), respectively.
Correlation of GATA2 expression with OS, EFS, and DFS. Shown are Kaplan-Meier estimates for pOS (A), pEFS (B), and pDFS (C). Patients with high GATA2 expression were compared with patients with normal GATA2 expression using a log-rank test. High GATA2 expression suggested a worse pOS and pEFS, although the differences did not reach statistical significance at the P = .05 level in univariate analysis.
Correlation of GATA2 expression with OS, EFS, and DFS. Shown are Kaplan-Meier estimates for pOS (A), pEFS (B), and pDFS (C). Patients with high GATA2 expression were compared with patients with normal GATA2 expression using a log-rank test. High GATA2 expression suggested a worse pOS and pEFS, although the differences did not reach statistical significance at the P = .05 level in univariate analysis.
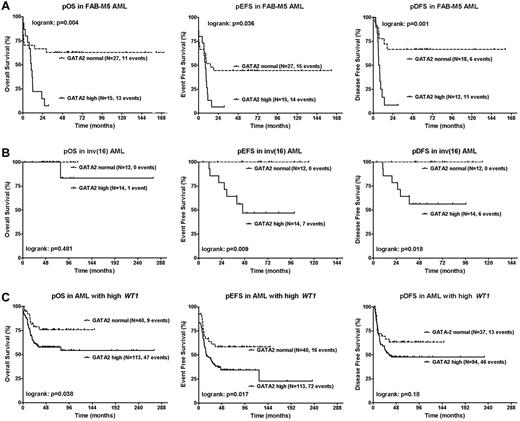
The prognostic impact of GATA2 was particularly evident in patients with FAB-M5 morphology (n = 42) and inv(16) (n = 26). In patients with FAB-M5 morphology, high GATA2 expression was associated with worse pOS, pEFS, and pDFS. The 4-year pOS was 7% ± 7% in FAB-M5 patients with high GATA2 expression (n = 15) compared with 63% ± 9% in FAB-M5 patients with normal GATA2 levels (n = 27; P = .004); the 4-year pEFS was 7% ± 6% compared with 44% ± 10% (P = .036) and the 4-year pDFS was 8% ± 8% compared with 67% ± 11% (P = .001; Figure 4A), respectively. In patients with inv(16), high GATA2 expression was associated with worse pEFS and pDFS, whereas no effect on pOS was observed. The 4-year pEFS was 47% ± 14% in inv(16) patients with high GATA2 expression (n = 14) compared with 100% in inv(16) patients with normal GATA2 levels (n = 12; P = .009); the 4-year pDFS was 56% ± 14% compared with 100% (P = .018; Figure 4B), respectively. Moreover, in patients with high WT1 expression, 11q23 translocations or FLT3-ITD positivity, a trend toward worse outcome was observed in patients with high GATA2 expression versus normal GATA2 expression (Figure 4C and supplemental Figure 3). Within other morphological, cytogenetic, or molecular subgroups, we did not observe differences in outcome between patients with high versus normal GATA2 expression (supplemental Table 3).
Relation of GATA2 expression with survival in morphologic and genetic AML subgroups. Kaplan-Meier estimates for pOS, pEFS, and pDFS in AML patients with FAB-M5 morphology (A), inv(16) (B), or high WT1 expression (C). Patients with high GATA2 expression were compared with patients with normal GATA2 expression using a log-rank test. High GATA2 expression was associated with worse outcome. P < .05 was considered statistically significant (2-tailed testing). Patient characteristics are described in supplemental Tables 5 through 7.
Relation of GATA2 expression with survival in morphologic and genetic AML subgroups. Kaplan-Meier estimates for pOS, pEFS, and pDFS in AML patients with FAB-M5 morphology (A), inv(16) (B), or high WT1 expression (C). Patients with high GATA2 expression were compared with patients with normal GATA2 expression using a log-rank test. High GATA2 expression was associated with worse outcome. P < .05 was considered statistically significant (2-tailed testing). Patient characteristics are described in supplemental Tables 5 through 7.
To assess the independent prognostic contribution of GATA2 expression, a multiple Cox regression analysis was conducted. In pediatric AML, several prognostic factors have been identified previously: age, WBC count, favorable karyotype, NPM1 mutation, CEBPA-DM, and FLT3-ITD.45 When GATA2 was entered into a multiple Cox regression analysis adjusting for these established prognostic factors, we observed a significant correlation between high GATA2 expression with worse pOS (HR = 1.7, P = .045), pEFS (HR = 2.1, P = .002), and pDFS (HR = 2.3, P = .004; Table 3 and supplemental Table 4).
GATA2 expression during treatment and follow-up
To study the prognostic relevance of GATA2 expression after induction and consolidation treatment, consecutive GATA2 expression levels were determined using real-time quantitative PCR in a representative subgroup of 38 patients (supplemental Table 10). Thirty-three patients reached CR, with a median time to first CR (CR1) of 1.4 months (range, 0.9-4.4), 3 patients died within 1 week after diagnosis (categorized as early death), and 2 patients had RD. Twelve patients relapsed, with a median time to relapse of 12.6 months (range, 6.0-65.4).
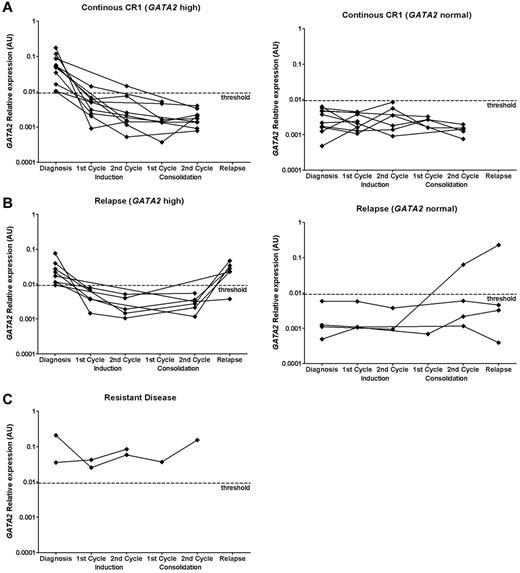
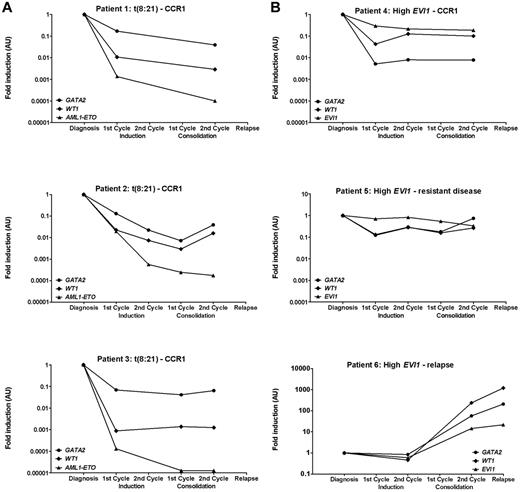
Both at diagnosis and clinical relapse, GATA2 expression was heterogeneous (> 2-log difference between highest and lowest expression). No differences were observed in median GATA2 expression at diagnosis versus relapse (P = .87). After induction and consolidation therapy, normalization of GATA2 expression was observed in patients who reached CR (Figure 5A-B), whereas GATA2 expression levels stayed high in patients with RD (Figure 5C). In 2 of 38 AML patients, GATA2 was sufficiently highly expressed to allow detection of at least a 2-log reduction (ie, sensitivity < 0.01). GATA2 expression was assessed in parallel with additional markers that have been used previously for the detection of residual disease: WT1 expression (n = 38), AML1-ETO fusion gene expression [patients with t(8;21); n = 3], and EVI1 expression (patients with high EVI1 expression, n = 3; Figure 6).6,44,47,48 The expression patterns of GATA2 were comparable to the expression patterns of the AML1-ETO fusion gene, EVI1 and WT1. However, the log reduction for WT1 and AML1-ETO was higher than for GATA2.
GATA2 expression in continuous CR, RD, and clinical relapse. Consecutive GATA2 expression at diagnosis and at different time points during treatment in a cohort of 38 pediatric AML patients from whom material was available for MRD analysis. GATA2 expression was determined using real-time quantitative PCR at diagnosis (n = 38), after the first cycle of induction therapy (n = 27), after completion of induction therapy (n = 23), after the first cycle of consolidation therapy (n = 12), after completion of consolidation therapy (n = 25), and at relapse (n = 10). Expression relative to GAPDH is plotted. GATA2 expression in patients with continuous CR1 (A; n = 21), relapse (B; n = 12), and RD (C; n = 2). Patients were subdivided into patients with high GATA2 expression at diagnosis versus normal GATA2 expression at diagnosis. GATA2 expression was considered to be high when the expression was 2 SD above the median GATA2 expression obtained in normal BM (n = 74), which was determined by real-time quantitative PCR. The threshold is depicted as a dashed line.
GATA2 expression in continuous CR, RD, and clinical relapse. Consecutive GATA2 expression at diagnosis and at different time points during treatment in a cohort of 38 pediatric AML patients from whom material was available for MRD analysis. GATA2 expression was determined using real-time quantitative PCR at diagnosis (n = 38), after the first cycle of induction therapy (n = 27), after completion of induction therapy (n = 23), after the first cycle of consolidation therapy (n = 12), after completion of consolidation therapy (n = 25), and at relapse (n = 10). Expression relative to GAPDH is plotted. GATA2 expression in patients with continuous CR1 (A; n = 21), relapse (B; n = 12), and RD (C; n = 2). Patients were subdivided into patients with high GATA2 expression at diagnosis versus normal GATA2 expression at diagnosis. GATA2 expression was considered to be high when the expression was 2 SD above the median GATA2 expression obtained in normal BM (n = 74), which was determined by real-time quantitative PCR. The threshold is depicted as a dashed line.
Relation between GATA2 expression and other residual disease markers. Consecutive GATA2, WT1, EVI1, and fusion gene expression at diagnosis and at different time points during treatment in pediatric AML patients with t(8;21) (A; n = 3) or high EVI1 expression (B; n = 3) determined by real-time quantitative PCR. Expression of GATA2 and WT1 relative to GAPDH is plotted. Expression of EVI1 and AML1-ETO relative to PBGD is plotted. For all of the different markers, expression at different time points during treatment was normalized to expression at diagnosis. CCR1 indicates continuous first CR.
Relation between GATA2 expression and other residual disease markers. Consecutive GATA2, WT1, EVI1, and fusion gene expression at diagnosis and at different time points during treatment in pediatric AML patients with t(8;21) (A; n = 3) or high EVI1 expression (B; n = 3) determined by real-time quantitative PCR. Expression of GATA2 and WT1 relative to GAPDH is plotted. Expression of EVI1 and AML1-ETO relative to PBGD is plotted. For all of the different markers, expression at different time points during treatment was normalized to expression at diagnosis. CCR1 indicates continuous first CR.
To determine whether GATA2 expression was related to outcome, we compared the GATA2 expression of patients who relapsed versus patients in continuous CR1 at different time points. Neither at diagnosis nor during chemotherapy were differences in GATA2 expression levels detected between the groups. After consolidation therapy, an increase of GATA2, WT1, and EVI1 expression was observed in 1 patient who was diagnosed with a clinical relapse 3 months later (Figure 6B bottom panel). In 2 other patients who suffered an early relapse with high GATA2 expression, we did not observe a rise in GATA2 expression levels after consolidation therapy (2.6-5.4 months before clinical relapse).
Discussion
In the present study, we investigated the relevance and prognostic value of GATA2 expression and mutations in a large cohort of de novo pediatric AML patients.
Mutational analysis was performed by sequencing of the DNA-binding zinc-finger domains of GATA2 in 230 patients. We detected nonsynonymous variations in the GATA2 gene with a frequency of 2.2% overall and 9.8% in pediatric CN-AML. All GATA2 variants affected highly conserved residues within the critical functional DNA-binding domain consisting of 2 zinc fingers. Functional studies have recently shown significant effects of 6 different mutations located within the N-terminal and C-terminal zinc-finger domain of GATA2 on transactivation of target genes, DNA-binding ability, protein-protein interactions, cellular differentiation, apoptosis, and global gene expression.27,29,35 In 1 patient, we detected a variation in codon 318 of the N-terminal zinc-finger domain adjacent to codon 319 in which an acquired mutation was described recently in an adult AML patient with FAB-M5 morphology.26 In codon 362 of the C-terminal zinc-finger domain, we identified 3 heterozygous missense variants and 1 heterozygous single nucleotide deletion resulting in loss of the C-terminal part of the protein. Because 4 of 5 GATA2 variants were present within codon 362, this suggests that these variations are indeed true mutations. One of these variants (R362Q) was identified recently as an acquired mutation in 2 adult AML patients with FAB-M5 morphology.26 In the highly conserved zinc-finger domains of the GATA2 gene, acquired and inherited mutations have been reported in myeloid malignancies. In (familial) myelodysplastic syndrome/AML and syndromes with a predisposition to developing myelodysplastic syndrome/AML, inherited GATA2 mutations are mainly clustered in codon 354 and codon 396-398 of the C-terminal zinc-finger domain (supplemental Figure 1).29-35 In contrast, in sporadic AML, the majority of acquired GATA2 variations are located within codon 362 in the C-terminal zinc-finger domain (Figure 1B),26 suggesting that this location may be a mutational hot spot.
The low frequency of GATA2 variations found in the present study is consistent with recent studies in adult AML, which reported acquired GATA2 mutations with a frequency of 3.6% in adult AML patients with FAB-M5 morphology.14,26,28,29 Interestingly, no GATA2 variations were observed in any of our pediatric patients with FAB-M5 morphology (n = 51). We detected mainly GATA2 variations in CN-AML. In all cases, we observed cooccurrence of nonsynonymous GATA2 variations with various other recurrent mutations, including NRAS mutations (n = 4), NPM1 mutations (n = 2), CEBPA-DM mutations (n = 2), and WT1 mutations (n = 1). In adult AML, cooccurrence of GATA2 mutations with other recurrent mutations (eg, WT1 mutation and CEBPA single mutation) has also been reported.26 Although the cooccurrence of these lesions remains a rare event in pediatric AML, this observation may suggest that these aberrations cooperate in leukemia development. Determination of the clinical significance of GATA2 variations in a larger cohort of pediatric CN-AML is highly recommended.
In the present study, GATA2 expression analysis demonstrated that in 155 of 237 diagnostic samples (65%), GATA2 expression was higher than in normal BM. Normalization of GATA2 expression was observed in patients who reached CR, whereas GATA2 expression levels stayed high in patients with RD. Although GATA2 expression during follow-up was studied in a limited number of patients, these data suggest that GATA2 may be an interesting marker for residual disease detection. However, because of the heterogeneous expression of GATA2 in AML and the relatively high background expression in normal hematopoietic cells, the sensitivity of GATA2 as a marker for residual disease may be limited. In only 2 of 38 AML patients was GATA2 sufficiently highly expressed to allow detection of at least a 2-log reduction (ie, sensitivity < 0.01). Assessment of quantitative fusion-gene expression was shown to be a more robust marker for residual disease. However, this strategy is limited to specific AML subgroups. In addition, MRD detection by multiparameter flow cytometry is a promising strategy that is applicable to the majority of AML patients.5,6
We have shown herein that high GATA2 expression at diagnosis is an independent poor prognostic factor for pOS (HR = 1.7, P = .045), pEFS (HR = 2.1, P = .002), and pDFS (HR = 2.3, P = .004) in pediatric AML overall. In adult AML, studies of the prognostic value of GATA2 have yielded conflicting results, which might be because of the relatively small patient cohorts and technical differences between studies in quantification of GATA2 expression.49-52 In a cohort of 112 adult AML patients, Vicente et al showed recently that high GATA2 expression was associated with worse outcome (pOS, pEFS, and pDFS). The prognostic value of high GATA2 expression was most evident in adult CN-AML.51,52 In the present study of pediatric AML, we did not observe a difference in survival based on GATA2 expression in CN-AML (n = 40), which may be because of differences in distribution of genetic aberrations underlying pediatric compared with adult CN-AML.
Interestingly, the prognostic impact of GATA2 was particularly high in specific morphologic and genetic AML subgroups. High GATA2 expression identified a group of patients with a significant worse outcome among patients with high WT1 expression, which is consistent with recent observations in adult AML.51 Moreover, in patients with FAB-M5 morphology and inv(16), we observed pronounced differences in survival between patients with high versus normal GATA2 expression, and a trend toward worse survival was observed in patients with 11q23 translocations or FLT3-ITD.
Based on the results of the present study, we conclude that GATA2 expression is a novel independent poor prognostic factor in pediatric AML. The negative correlation between GATA2 expression and outcome, especially in specific morphologic and genetic AML subgroups, suggests that GATA2 may be a useful marker for the identification of patients with a dismal prognosis, which could lead to better risk-group stratification and risk-adapted therapy in the future. In addition, we identified the presence of GATA2 mutations as a recurrent but infrequent event in childhood AML, which mainly occurs in CN-AML. The clinical significance of GATA2 mutations should be determined in a larger cohort of pediatric CN-AML patients, and the value of GATA2 expression as a predictor for poor OS, EFS, and DFS should be validated in prospective studies.
The online version of this article contains a data supplement.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Acknowledgments
The authors thank Mrs Patricia Hoogeveen for technical assistance.
This study was supported by The Dutch Children Cancerfree Foundation (KiKa; grant 70 to V.H.J.v.d.V.).
Authorship
Contribution: M.L., I.H.I.M.H., V.H.J.v.d.V., B.A.v.d.R., M.M.v.d.H.-E., C.M.Z., and J.H.J. designed the study; M.L., I.H.I.M.H., and R.H.J.N.K. performed the laboratory research; M.L., I.H.I.M.H., R.H.J.N.K., J.B.M.B., and J.H.J. analyzed and interpreted the data; V.d.H., J.T., A.B., and D.R. collected the patient samples and clinical data; M.L. and J.H.J. wrote the manuscript; and all authors critically reviewed the manuscript and gave their final approval.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Dr J. H. Jansen, Department of Laboratory Medicine, Laboratory of Hematology, Radboud University Nijmegen Medical Centre, Geert Grooteplein 8, 6525 GA Nijmegen, The Netherlands; e-mail: j.jansen@labgk.umcn.nl.