Abstract
BACKGROUND: We have previously shown that one target of hyper-methylation in AML is the promoter of the tumour suppressor and stress-response mediator Growth Arrest and DNA Damage inducible 45A (GADD45A) (GADD45AmeHI; 42% of AML). In mice Gadd45a has recently been shown to play a critical role in HSC stress responses. Gadd45a deficiency leads to enhanced HSC self-renewal, DNA damage accumulation in HSC, increased susceptibility to leukemogenesis, and impairment in HSC apoptosis after genotoxic exposure (Chen et al, Blood 2014). These findings suggest that hypermethylation of the GADD45A gene may play an important role in the altered properties of HSC, leukaemic initiation and progression. Promoter hypermethylation of this gene defines a patient group with poor survival on standard therapy (Perugini et al, Leukaemia 2012). To explore further the molecular basis of the GADD45AmeHI patient group weperformed genetic profiling of diagnosis samples using a Sequenom multiplex mutation panel, or using whole exome sequencing for broader coverage (n=95 patients).Sequenom MassARRAY was used for quantitative detection of GADD45A promoter methylation in patient samples. For a cohort of matched diagnosis and relapse samples we used CpG methylation data for GADD45A determined by ERRBS (Akalin et al, PLoSGenetics 2012). Response to cytotoxic drugs and assessment of drug combinations with 5-Aza-deoxycytidine (decitabine, DAC) and anthracycline (Daunorubicin, DNR) was performed in AML cell lines, and with primary leukemic cell populations.
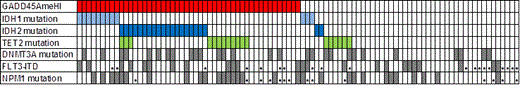
RESULTS: The association of the GADD45AmeHI patient group with poor outcome was validated in an independent AML patient cohort of 48 patients from the Alfred Hospital, Melbourne, Australia (p=0.003; HR3.35). Whole exome sequencing and Sequenom multiplex analysis of 95 AML patients revealed a striking co-occurrence of the GADD45AmeHI phenotype with mutations in IDH1, IDH2, and TET2 (p<0.0001, Fisher’s exact test, Fig. 1). To test the prediction that GADD45A hypermethylation may be an important factor for relapse we investigated GADD45A promoter DNA methylation levels in paired diagnosis and relapse samples. In a paired analysis of 39 patients we show that relapse samples display a significant increase in GADD45A promoter CpG methylation (p=0.035, paired t-test). This is consistent with emergence in many patients following chemotherapy of a chemoresistant clone that has increased GADD45A methylation and reduced GADD45A activity.
We next tested whether reactivation of GADD45A expression in GADD45AmeHI patient samples could be achieved through the use of hypo-methylation agents, and whether this is beneficial for response to chemotherapy. DAC treatment has been reported to induce DNA demethylation and GADD45A reactivation in primary AML samples (Klco et al, Blood 2013), and we observe reduced GADD45A promoter methylation and increased expression following DAC treatment of the GADD45AmeHI AML cell line (Mv4;11), consistent with DNA methylation-induced gene silencing of GADD45A. DAC pre-treatment of the GADD45AmeHI AML cell lines MOLM13 and Mv4;11, and three primary AML samples (GADD45AmeHI), resulted in increased GADD45A expression and increased DNR sensitivity.
CONCLUSIONS: DNA methylation of the GADD45A proximal promoter marks a large percentage of AML patients at diagnosis including the majority of those with IDH1/2 and TET2 mutations (collectively these occur in 28% of AML (Network CGAR, N Engl J Med, 2013)), and is an independent predictor of poor outcome in two independent patient cohorts. Our data shows that silencing of GADD45A through increased promoter CpG methylation maybe an important early event in leukaemogenesis associated with impaired TET2 activity. Based on recent studies describing the properties of Gadd45a-deficient murine HSC we suggest reduced GADD45A activity in this subset of patients may contribute to the properties of pre-leukaemic HSC that have been associated with IDH1/2 mutation and reported to display clonal expansion, resistance to chemotherapy, and ultimately a high risk of relapse.
In vitro drug experiments suggest that a priming schedule of DAC followed by DNR may provide a successful tailored treatment strategy for GADD45AmeHI patients, in combination with GADD45A expression as a biomarker predicting increased DNR sensitivity.
Co-association of GADD45AmeHI with IDH1/2 and TET2 mutations in 95 AML patients
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.