Abstract

Background: Vosaroxin, is a first-in-class anti-cancer quinolone derived (AQD) DNA intercalator and topoisomerase II inhibitor, which is not a substrate for p53 or P-glycoprotein, and is currently under evaluation for the treatment of pts with AML and high-risk MDS.
Methods: Pts are eligible if they have AML or high-risk MDS (defined as having >/= 10% blasts), are 60 years of age or older, and have adequate performance status (ECOG </= 2) and organ function. In the phase I part of the study the first six pts received vosaroxin 90 mg/m2 daily on days 1 and 4 with decitabine 20 mg/m2 daily for 5 days repeated in approximately 4 to 5 week intervals for up to 7 cycles. This dose was well tolerated in the 6 patients. However, due to occurrence of 8 episodes of grade 3/4 mucositis in 7 of the subsequent 16 patients the induction dose of vosaroxin was reduced to 70 mg/m2 with the vosaroxin dose maintained at 70 mg/m2 or reduced to 50 mg/m2 in consolidation cycles. 34 patients were treated at this modified dose regimen. The primary endpoint was to determine the overall response rate including complete response (CR) + CR without platelet recovery (CRp) + CR with insufficient hematological recovery (CRi). Secondary endpoints were: CR duration, disease-free survival, overall survival, safety, and early mortality.
Results: To date, 56 pts (50 AML, 6 high-risk MDS) with a median age of 69 years (range, 60 - 78) have been enrolled. They included 19 (34%) pts with diploid cytogenetics, 21 (38%) with complex cytogenetic abnormalities including chromosome 5 and/or 7 abnormalities, and 16 (28%) with other miscellaneous abnormalities. Fourteen (25%) pts with AML had antecedent hematological disorders (AHD) including 7 with MDS, 4 with MPN, 2 with MDS/MPN, and 1 with CLL. Four pts with AHD had received prior therapy including 5-azacytidine (n=1), decitabine (n=1), ruxolitinib + 5-azacytidine (n=1), and lenalidomide (n=1). Additionally, 10 (18%) pts had therapy-related disease with prior exposure to chemotherapy or radiation therapy. Median bone marrow blast %, and median white blood cell, hemoglobin, & platelet counts were 40% (range, 11 - 97), 3.4 x 109/L (range, 0.4 - 57), 9.4 g/dL (range, 6.8 - 13.1), and 35 x 109/L (range, 7 - 333), respectively. All 56 pts have completed >/=2 cycles of therapy and were evaluable for response; 30 (54%) achieved CR, 8 (14%) CRp, and 5 (9%) CRi for an overall response rate of 77%. Minimal residual disease (MRD) by 19 color flow-cytometry was evaluable in 35 of the 43 responders. MRD was not detectable in 24 of 35 (66%) evaluable responders. All 56 patients had baseline cytogenetics and clinically validated next generation sequencing-based analysis for the detection of somatic mutations in the coding sequence of 28 genes commonly mutated in myeloid neoplasms. Response by baseline characteristics is shown in table 1. The median number of cycles to response was 1 (1 - 4); 13 pts have required >1 cycle to achieve response. Seven (13%) pts have proceeded to allogeneic stem cell transplant. The median follow-up is 4.7 months (1.3 - 20.8). The regimen was well tolerated with the main therapy related grade >/= 3 toxicities were mucositis in 10 (18%) pts and liver enzyme elevation in 8 (14%).
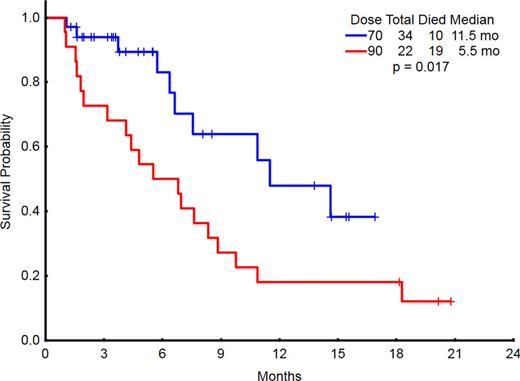
The median overall survival (OS) for all pts is 8.3 months. Four-week and 8-week mortality for all pts were 0 and 14%, respectively. The induction dose of vosaroxin was 90 mg/m2 in 22 pts and 70 mg/m2 in 34 pts. The lower induction dose of vosaroxin was associated with a reduced early mortality and an improved overall response rate and OS (Table 2 and Figure 1).
Conclusion: Combination of vosaroxin and decitabine is effective in older pts with AML and high-risk MDS. Reponses were encouraging in the pts with TP53 and complex cytogenetics. The lower dose of vosaroxin 70 mg/m2 on days 1 and 4 is better tolerated and is associated with significantly improved outcomes.
Response by baseline characteristics
| Parameter . | Category . | N . | Overall response (CR, CRp, CRi) . | CR . |
|---|---|---|---|---|
| Age | 60-74 | 44 | 80% | 57% |
| >/=75 | 12 | 67% | 42% | |
| Cytogenetics | Diploid | 19 | 86% | 57% |
| -5/-7/other adverse | 21 | 68% | 42% | |
| Miscellaneous | 16 | 75% | 63% | |
| Mutation Status | IDH2 | 11 | 91% | 73% |
| IDH1 | 14 | 57% | 43% | |
| TP53 | 11 | 73% | 55% | |
| RAS | 11 | 64% | 28% |
| Parameter . | Category . | N . | Overall response (CR, CRp, CRi) . | CR . |
|---|---|---|---|---|
| Age | 60-74 | 44 | 80% | 57% |
| >/=75 | 12 | 67% | 42% | |
| Cytogenetics | Diploid | 19 | 86% | 57% |
| -5/-7/other adverse | 21 | 68% | 42% | |
| Miscellaneous | 16 | 75% | 63% | |
| Mutation Status | IDH2 | 11 | 91% | 73% |
| IDH1 | 14 | 57% | 43% | |
| TP53 | 11 | 73% | 55% | |
| RAS | 11 | 64% | 28% |
Outcomes by induction dose of vosaroxin
| Induction dose (vosaroxin) . | N . | Med OS . | 8-week mortality . | Overall Response . | CR . | Need >1 cycle to response . |
|---|---|---|---|---|---|---|
| 90 mg/m2 | 22 | 5.5 mos | 25% | 73% | 41% | 23% |
| 70 mg/m2 | 34 | 11.5 mos | 6% | 79% | 62% | 23% |
| Induction dose (vosaroxin) . | N . | Med OS . | 8-week mortality . | Overall Response . | CR . | Need >1 cycle to response . |
|---|---|---|---|---|---|---|
| 90 mg/m2 | 22 | 5.5 mos | 25% | 73% | 41% | 23% |
| 70 mg/m2 | 34 | 11.5 mos | 6% | 79% | 62% | 23% |
Daver:ImmunoGen: Other: clinical trial, Research Funding. Off Label Use: Vosaroxin in the management of AML. Pemmaraju:Stemline: Research Funding; Incyte: Consultancy, Honoraria; Novartis: Consultancy, Honoraria, Research Funding; LFB: Consultancy, Honoraria. Konopleva:Novartis: Research Funding; AbbVie: Research Funding; Stemline: Research Funding; Calithera: Research Funding; Threshold: Research Funding. DiNardo:Novartis: Research Funding. Cortes:Pfizer: Consultancy, Research Funding; Novartis: Consultancy, Research Funding; Teva: Research Funding; BerGenBio AS: Research Funding; BMS: Consultancy, Research Funding; Ariad: Consultancy, Research Funding; Astellas: Consultancy, Research Funding; Ambit: Consultancy, Research Funding; Arog: Research Funding; Celator: Research Funding; Jenssen: Consultancy. Craig:Sunesis: Employment, Equity Ownership.
Author notes
Asterisk with author names denotes non-ASH members.

This icon denotes a clinically relevant abstract