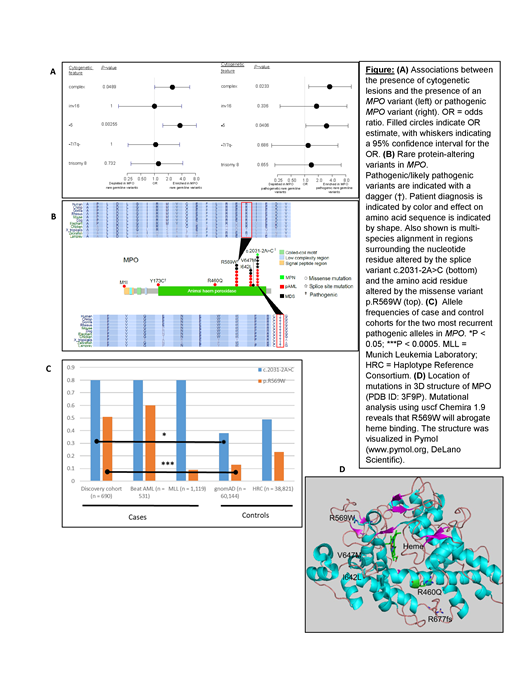
Myeloperoxidase (MPO) on chromosome 17q22 codes for a heme-containing protein produced following commitment and during myeloid differentiation. Ultimately, mature neutrophil azurophilic granules have MPO as their major component. Consistently, high MPO expression, suggestive of a more differentiated type, has been associated with favorable outcomes in acute myeloid leukemia (AML). MPO deficiency is an extremely rare autosomal recessive disorder caused by any of a number of known germline variants, though the propensity to develop myeloid malignancy has not been reported for affected individuals. Somatic MPO mutations have not be identified in myeloid neoplasia (MN).
In an effort to identify novel susceptibility genes and variants, we analyzed whole-exome sequence data from the germlines of 690 MN patients. Among all genes examined, MPO carried the highest burden of variants deemed pathogenic or likely pathogenic under the American College of Medical Genetics (ACMG) criteria; 19/690 MN patients (2.7%) carried a pathogenic/likely pathogenic MPO variant. Carriers of pathogenic MPO variants had higher rates of cytogenetic lesions, particularly -5 and complex karyotypes (Fig. A). The pathogenic variants found in our cohort are dominated by two substitutions: a splice site variant (c.2031-2A>C; eleven patients), and a missense variant (c.C1705T, p.R569W; seven patients) (Fig. B). Multi-species alignment shows that both of the corresponding wild-type alleles are highly conserved. Furthermore, both variants have significantly higher frequencies in our cohort (0.8% and 0.51%, respectively), as well as in the recently-published Beat AML patient cohort (0.8% and 0.6%), as compared to control populations (0.38% and 0.13% in gnomAD; OR 1.9, 95% CI 1.1-3.5, P<0.05 for c.2031-2A>C and OR 3.6, 95% CI 1.9-8.0, P<.0005 for p.R569W) (Fig. C). Of note is that the c.2031-2A>C variant has previously been reported in a family with hereditary MPO deficiency, with a splice variant-induced retention of intron 11 in MPO messenger RNA, inducing a premature stop codon. Another study showed that the R569W variant results in maturation arrest in the processing of MPO precursors. Analysis of the 3D structure of MPO shows that the R569W variant will abrogate heme binding (Fig. D).
We next sought to validate our findings in an independent cohort. To this end, we queried next-generation DNA and RNA sequence from 1,119 myeloid malignancy patients from the Munich Leukemia Laboratory (MLL). Similar to our discovery cohort and the Beat AML group, the c.2031-2A>C variant had significantly higher allele frequency in confirmatory cohort (0.8%) than in population controls (0.38%), though the R569W did not (Fig. C). We found that MPO expression is significantly lower in carriers of the c.2031-2A>C allele (P = 0.024), as expected since the variant affects mRNA processing.
There have been no prior reports of MPO as a MN susceptibility gene, but it is an appealing candidate. Taken together with evidence from earlier studies characterizing the impact of the variants highlighted here, our results support the hypothesis that inherited disruption of MPO confers myeloid malignancy susceptibility.
Haferlach:MLL Munich Leukemia Laboratory: Employment, Equity Ownership. Maciejewski:Alexion: Consultancy; Novartis: Consultancy.
Author notes
Asterisk with author names denotes non-ASH members.