Abstract
Thrombocytopenia is typically observed in patients undergoing hematopoietic stem cell transplantation. We hypothesized that delayed platelet count recovery might be ameliorated by increasing the number of megakaryocyte colony- forming units (CFU-Meg) in the hematopoietic cell graft. To test this hypothesis, we evaluated cytokine combinations and culture medium potentially useful for expanding CFU-Meg in vitro. We then examined the ability of expanded cells to accelerate platelet recovery in an animal transplant model. Depending on the cytokine combination used, we found that culturing marrow CD34+cells for 7 to 10 days in serum-free cultures was able to expand CFU-Meg ∼40 to 80 times over input number. Shorter incubation periods were also found to be effective and when CD34+ cells were exposed to thrombopoietin (TPO), kit ligand (KL), interleukin-1α (IL-1α), and IL-3 in serum-free cultures for as few as 48 hours, the number of assayable CFU-Meg was still increased ∼threefold over input number. Of interest, cytokine primed marrow cells were also found to form colonies in vitro more quickly than unprimed cells. The potential clinical utility of this short-term expansion strategy was subsequently tested in an in vivo animal model. Lethally irradiated Balb-C mice were transplanted with previously frozen syngeneic marrow mononuclear cells (106/mouse), one tenth of which (105) had been primed with [TPO, KL, IL-1a, and IL-3] under serum-free conditions for 36 hours before cryopreservation. Mice receiving the primed frozen marrow cells recovered their platelet and neutrophil counts 3 to 5 days earlier than mice transplanted with unprimed cells. Mice which received marrow cells that had been primed after thawing but before transplantation had similar recovery kinetics. We conclude that pretransplant priming of hematopoietic cells leads to faster recovery of all hematopoietic lineages. Equally important, donor cell priming before transplant may represent a highly cost-effective alternative to constant administration of cytokines during the posttransplant recovery period.
THROMBOCYTOPENIA remains a significant cause of morbidity in patients undergoing high-dose chemotherapy and hematopoietic stem cell transplantation. The platelet transfusions used to treat this problem are a potential source of blood borne pathogens, not infrequently lead to alloimmunization of the recipient,1 and consume millions of scarce health care dollars each year.2 For all these reasons, it is straightforward that an effective measure for ameliorating this problem would make an important contribution to the care of patients undergoing hematopoietic cell transplantation.
Although many factors could be hypothesized to be of etiologic importance in the pathogenesis of posttransplant thrombocytopenia, three that may be of particular importance are inadequate production of megakaryocyte growth factors, release of inhibitory cytokines after marrow damage, and inadequate numbers of megakaryocyte progenitor cells in the donor graft.3 The first two considerations have received the most attention to date. However, transplantation of peripheral blood stem cells harvested after cytokine stimulation has not led to consistent shortening of the thrombocytopenic period,4 and attempts to stimulate platelet recovery in the posttransplant period with cytokines such as interleukin-3 (IL-3), and IL-6 have been associated with unacceptable toxicity.5-7IL-11 and thrombopoietin (TPO) may prove more useful in this regard8,9 but this has not been formally proven. Regardless, administration of additional cytokines to transplant patients is likely to significantly increase the cost of an already expensive procedure. Avoiding platelet transfusions might decrease a patient's exposure to megakaryocyte colony-forming unit (CFU-Meg) inhibitory cytokines, such as PF4, which are released from platelet alpha granules,10-13 but platelets cannot be withheld when clinically indicated since the immediate consequences may be far more severe.
Our group has been interested in the technical feasibility of expanding CFU-Meg in the cells to be transplanted as a practical way of enhancing platelet production in the recipient until more primitive progenitor cells have had time to engraft and expand. We report herein in vitro and in vivo studies which suggest that this approach is feasible, economical, and likely to be successful in the population of interest.
MATERIALS AND METHODS
Human cells.
Light-density marrow mononuclear cells (MNC) were obtained from seven consenting normal human donors and depleted of adherent cells and T lymphocytes (A−T−MNC) as described.14-17 CD34+ cells were enriched from the A−T−MNC population by incubation with anti-HPC1 murine monoclonal antibody (Becton Dickinson, Mountain View, CA) and subsequent immunoselection of antibody-labeled cells with magnetic beads according to the manufacturer's protocol (Dynal, Oslo, Norway). Briefly, 2 × 107A−T−MNC were incubated (1 hour at 4°C on a rotating rack) in 1 mL of Iscove DMEM (GIBCO, Grand Island, NY) containing 5% (vol/vol) bovine calf serum (BCS; Hyclone, Logan, UT) and 50 μL of anti-CD34 antibodies (anti-HPC1). After incubation, the cells were washed 3× in Iscove DMEM + 5% BCS, then resuspended in 1 mL of Iscove DMEM + 5% BCS with 150 μL of sheep antimouse coated paramagnetic beads (Dynal). Cells were incubated again (45 minutes at 4°C on a rotating rack) and then transferred to 5-mL polystyrene plastic tubes for magnetic collection of tagged cells as directed by the manufacturer (Dynal). The CD34 enrichment procedure was performed a total of 3×. After the final magnetic separation, the remaining cells were resuspended in Iscove DMEM + 10% BCS. Cell viability and purity was >98%.
Murine cells.
Light density MNC were enriched from marrow flushed from the femurs of pathogen free, 4- to 6-week old female Balb-C mice (Taconic Labs, Germantown, NY) by Ficoll-Paque density gradient centrifugation. The light density MNC were further depleted of monocytes by adherence to plastic dishes for 2 hours.
Human CFU-Meg assay.
CFU-Meg were assayed in plasma clot cultures as previously described.14,17 CFU-Meg colony growth was stimulated with recombinant human TPO (50 ng/mL) and IL-3 (20 U/mL). Colonies were enumerated after 10 to 11 days of culture by immunofluorescence with an antiplatelet glycoprotein IIb/IIIa monoclonal antibody (Immunotech, Marseille, France) also as previously reported.14 17 Data points represent mean ± SD of quadruplicate culture dishes.
Megakaryocyte phenotyping of expanded cells.
Cells from expansion cultures were washed twice with Ca++and Mg++ free phosphate buffered saline (GIBCO-BRL, Grand Island, NY), stained with the Immunotech murine monoclonal antihuman IIb/IIIa antibody (1:100) for 30 minutes. The cells were then washed and binding of the primary antibody was detected with a phycoerythrin-conjugated sheep antimouse Ab (1:100) (Sigma, St Louis, MO). Expression of IIb/IIIa was evaluated by fluorescence-activated cell sorter (FACS).
Cryopreservation of MNC.
Cells (2 × 104 human CD34+ MNC/mL, or 5 × 106 murine A-MNC/mL) were suspended in freezing medium (Iscove DMEM with 10% dimethyl sulfoxide [Sigma] + 20% BCS [Hyclone] placed in cold 2-mL cryotubes (Corning, Corning, NY) and then transferred immediately to a −80°C freezer. Twenty-four hours later the vials were placed into liquid nitrogen (−196°C) for long-term storage. Four weeks later, samples were thawed and assayed for CFU-Meg as described above.
Cell thawing procedure.
Cryotubes were placed in a 37°C water bath for 2 minutes. The cell suspension was then transferred to 50-mL polypropylene tubes (Corning) containing 30 mL of ice-cold Iscove DMEM + 20% BCS. After 10 minutes of incubation on ice, cells were centrifuged (1,000 rpm, 10 minutes) and the pellet resuspended in fresh culture medium. Cell viability was assessed by trypan blue (0.5%) exclusion test and expressed as a percentage of 100 cells counted.
Serum free, ex vivo expansion of human CD34+ MNC.
CD34+ A−T− MNC were suspended in Iscove DMEM (5 × 103 cells/mL) supplemented with 25% (vol/vol) serum substitute or 25% animal sera (12.5% horse serum [HS] + 12.5% BCS). Cells were seeded into 24-well plates (1 mL/well) and incubated for 7 to 21 days (37°C, 95% humidity, 5% CO2). Serum substitute consisted of 1% delipidated, deionized, charcoal treated BSA, 270 μg/mL iron saturated transferrin, insulin (20 μg/mL), cholesterol (5.6 μg/mL) and 2 mmol/L L-glutamine (all from components from Sigma). Cytokines (R & D, Minneapolis, MN; unless otherwise indicated) added to the wells consisted of recombinant human (rH) TPO (50 ng/mL), rH kit ligand (KL) (10 ng/mL), rH IL-3 (20 U/mL, Genetics Institute, Cambridge, MA), rH interleukin-1α (100 pg/mL, Genzyme, Cambridge, MA), IL-6 (40 U/mL, Robert Wood Johnson Pharmaceutical Institute, Raritan, NJ), IL-11 (100 ng/mL, Genetics Institute).
Short-term cytokine priming of human MNC.
CD34+ cells (4 × 104/mL) were suspended for varying periods of time in 0.5-mL aliquots of expansion medium (Iscove DMEM [GIBCO; 25% (vol/vol) serum substitute; TPO [50 ng/mL], KL [10 ng/mL], IL-3 [20 U/mL], and IL-1α [100 pg/mL]) for 48 hours in 4-mL polypropylene tubes. Thereafter, an aliquot of cells (2 × 104/0.5 mL) was removed for CFU-Meg assay, while the remainder of the cells were stored in liquid nitrogen as described. On occasion, freshly isolated CD34+ cells were cryopreserved as described above, and upon thawing were suspended in expansion medium for 48 hours after which time cells were employed for evaluation of CFU-Meg numbers. Control cells were cultured briefly in expansion medium, washed, and then plated immediately in plasma clot containing rH TPO (50 ng/mL), and IL-3 (20 U/mL).
Short-term cytokine priming of murine MNC.
Murine A− MNC (fresh or newly thawed) were primed for 36 hours under serum-free conditions with the mixture of rH TPO (50 ng/mL), rH IL-1α (100 pg/mL), recombinant murine (rM) IL-3 (10 ng/mL, Genzyme), and rM KL (10 ng/mL, Genzyme). After priming, fresh cells were washed and then cryopreserved, while thawed cells after priming were mixed with newly thawed, unprimed cells in a ratio of 1:9 and then used for transplantation.
Marrow transplantation in lethally irradiated mice.
Balb-C mice were lethally irradiated from γ-irradiation source (900 cGy). Twenty-four hours later, mice were transplanted by tail vein injection with unprimed MNC (106 cells/mouse), or 106 total cells consisting of a mixture of cytokine primed and unprimed cells in a ratio of 1:9. Transplanted mice were bled at intervals from the retro-orbital plexus for leukocyte, platelet, and hematocrit determinations using unopette microcollection (Becton Dickinson, Rutherford, NJ) and heparinized microhematocrit capillary tubes (Oxford Labware, St Louis, MO), as described.18
Statistical analysis.
Arithmetic means and standard deviations of numbers of colonies, or cultured cells, were calculated using Instat 1.14 statistical software package (GraphPad 1993, San Diego, CA). Differences between means were judged for statistical significance (P < .01) using Student's t-test for unpaired samples. To analyze platelet recovery kinetics in the murine transplantation experiments mean nadir platelet values between the control and treatment groups were compared using an independent samplest-test. We also examined and compared the rate of platelet count recovery from day 9 (nadir) through day 16 using a random effects linear model that compares the fitted slopes of the linear increase between the groups. The linear models for each group were constructed based on individual animal data and were fit by maximum likelihood methods using a linear random effects model that compares the slopes of the line of increase between the groups. The lines for each group were constructed from the individual animal's data and were fit by maximum likelihood methods using the XTREG command in STATA version 5.0 m (STATA Corp, College Station, TX).
RESULTS
Serum free, ex vivo expansion of human CFU-Meg.
Our group,10,19 and others,11 12 have reported that megakaryocytes and platelets contain a variety of negative regulatory proteins in their alpha granules that are released upon activation of these cells. For this reason, and because serum is known to contain other hematopoietic inhibitory substances, we hypothesized that a serum-free culture system might be more conducive to megakaryocyte progenitor cell expansion than one containing serum. Proceeding from this premise, we isolated CD34+A−T− MNC and cultured the cells serum-free in Iscove DMEM containing different cytokine cocktails designed to stimulate human megakaryocyte growth. The effects of these cytokines on total megakaryocytes and CFU-Meg produced by the serum-free system was assessed after varying times in culture using a combination of immunocytochemistry, FACS analysis and colony forming assays.
Results of initial culture experiments are shown in Tables 1 and2. We first examined CFU-Meg expansion as a function of cytokine exposure kinetics. CD34+ cells were exposed to a maximally stimulating cytokine cocktail for 7, 10, 14, and 21 days. The number of assayable CFU-Meg, in comparison to colonies obtained after immediately plating cells in cultures containing the same cytokine cocktail is shown in Table 1. These experiments indicated that optimal CFU-Meg expansion was obtained by expanding cells for ∼10 days, which resulted in an ∼50-fold expansion of megakaryocyte progenitor cells. Of interest, the number of megakaryocytes/colony paralleled the kinetics of progenitor cell expansion in a similar but somewhat delayed fashion (Table 1). After a 7-day exposure to cytokines, when the number of assayable CFU-Meg had increased ∼15-fold, the number of cells/colony was not different from control colonies. However, after 10 and 14 days of cytokine stimulation, when expansion of colony-forming cells had peaked, the number of megakaryocytes/colony was ∼doubled. After 21 days, the number of CFU-Meg declined to ∼day 7 value, while the number of cells/colony declined to control values.
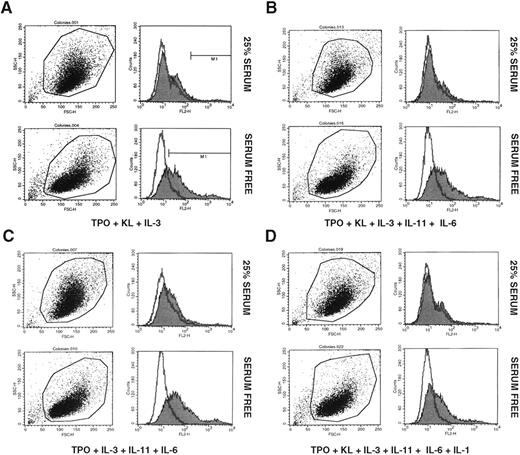
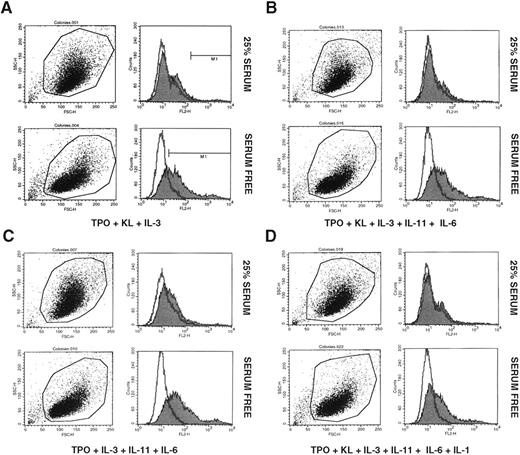
We then examined a number of different cytokine combinations for their ability to expand CFU-Meg after 10 days of culture (Table 2). Depending on the combination of cytokines employed, the number of megakaryocytic progenitors increased from 40 to 80 times over input number. Of interest, medium supplemented with animal serum was much less effective for CFU-Meg expansion purposes. In cultures supplemented with [12.5% HS + 12.5% BCS] the number of CFU-Meg increased only ∼2 to 3 times over input number. In addition, we also found that cells expanded in serum-free medium appeared to express IIb/IIIa at higher concentrations on their surface than cells that had been expanded in serum containing medium (Fig 1A through D).
Expression of glycoprotein IIb/IIIa by CD34+ MNC after 10 day exposure to TPO + KL + IL-3 (A); TPO + KL + IL-3 + IL-11 + IL-6 (B); TPO + IL-3 + IL-11 + IL-6 (C); and TPO + KL + IL-3 + IL-11 + IL-6 + IL-1α (D). Cells displayed in upper panels of A, B, C, and D were cultured in Iscove DMEM supplemented with 25% animal serum (12.5% BCS + 12.5% HS), whereas cells displayed in lower panels of A, B, C, and D were cultured in Iscove DMEM supplemented with 25% serum substitute. Representative data from three independent studies are shown.
Expression of glycoprotein IIb/IIIa by CD34+ MNC after 10 day exposure to TPO + KL + IL-3 (A); TPO + KL + IL-3 + IL-11 + IL-6 (B); TPO + IL-3 + IL-11 + IL-6 (C); and TPO + KL + IL-3 + IL-11 + IL-6 + IL-1α (D). Cells displayed in upper panels of A, B, C, and D were cultured in Iscove DMEM supplemented with 25% animal serum (12.5% BCS + 12.5% HS), whereas cells displayed in lower panels of A, B, C, and D were cultured in Iscove DMEM supplemented with 25% serum substitute. Representative data from three independent studies are shown.
Short-term cytokine priming of CD34+MNC− effect on human CFU-Meg.
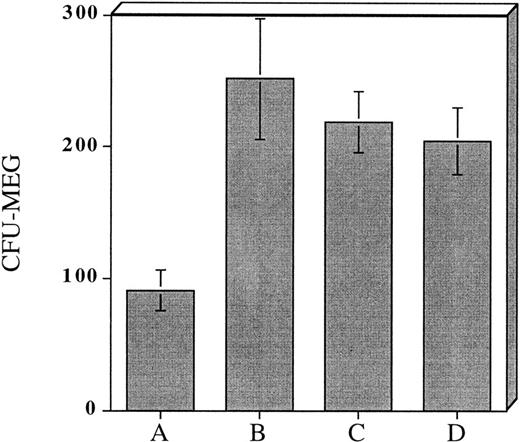
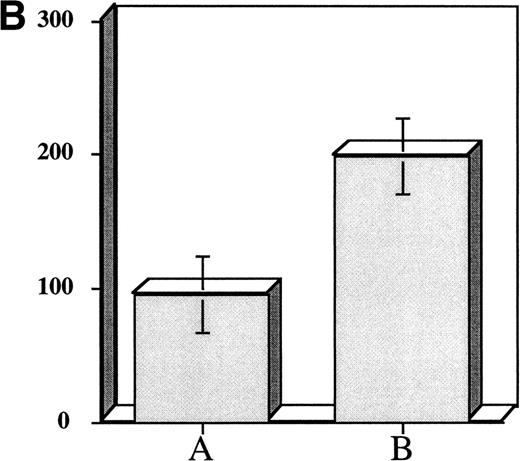
Incubation of CD34+ MNC for 7 to 14 days in serum-free culture appeared to be an effective strategy for expanding megakaryocyte progenitors and accelerating their maturation. Nevertheless, since the goal of our studies was to develop an efficient and practical approach for CFU-Meg expansion we began to investigate the utility of shorter cytokine preincubation periods. Accordingly, in the next series of experiments, we cultured cells serum-free for 48 hours in a cytokine cocktail consisting of (TPO + KL + IL-3 + IL-1a). As noted previously, this combination of cytokines was found to expand CFU-Meg >50-fold over the input number of CFU-Meg (Tables 1 and 2). Using these conditions, we found that CD34+ derived CFU-Meg were increased ∼threefold in comparison to untreated controls (Fig 2B).Further, when cytokine primed CFU-Meg were cryopreserved, subsequently thawed, and then plated in plasma clot cultures, the CFU-Meg not only remained clonogenic (Fig 2C), they appeared to form colonies faster than CFU-Meg present in an unmanipulated population of progenitor cells. Of interest, it was also possible to expand CFU-Meg in progenitor cells that had been previously frozen but unexposed to cytokines. The degree of expansion, ∼twofold to threefold over noncytokine exposed cells, was similar to that observed in cells exposed to cytokines ex vivo before cryopreservation (Fig 2D).
Effect of prefreezing or postthawing cytokine stimulation on CFU-Meg expansion from 104 CD34+ MNC. (A) CFU-Meg from cells cultured immediately. (B) CFU-Meg from cells cultured serum-free for 48 hours in the presence of TPO + KL + IL-1a + IL-3. (C) CFU-Meg from cytokine stimulated MNC before cryopreservation. (D) CFU-Meg from cytokine stimulated MNC after thawing. Data are pooled from four independent experiments, each performed in quadruplicate.
Effect of prefreezing or postthawing cytokine stimulation on CFU-Meg expansion from 104 CD34+ MNC. (A) CFU-Meg from cells cultured immediately. (B) CFU-Meg from cells cultured serum-free for 48 hours in the presence of TPO + KL + IL-1a + IL-3. (C) CFU-Meg from cytokine stimulated MNC before cryopreservation. (D) CFU-Meg from cytokine stimulated MNC after thawing. Data are pooled from four independent experiments, each performed in quadruplicate.
Developing a murine model for ex vivo cytokine priming of transplanted progenitor cells.
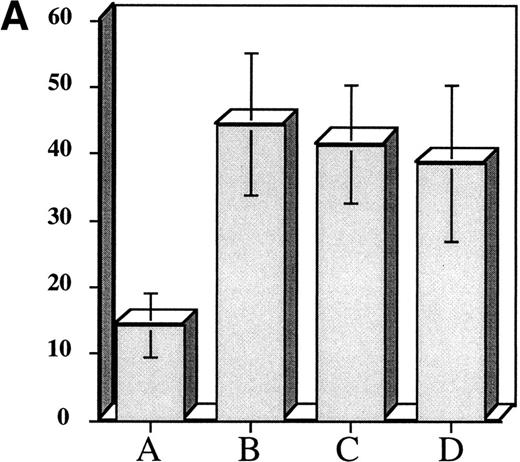
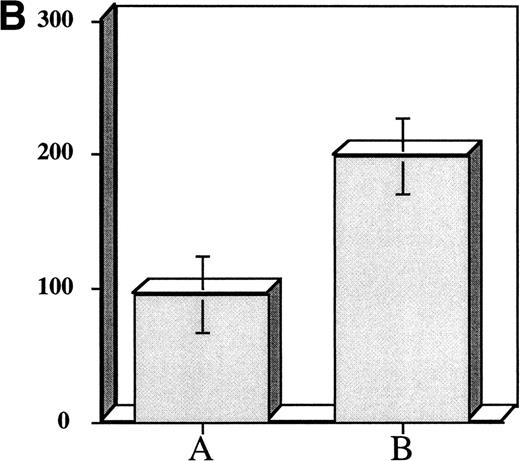
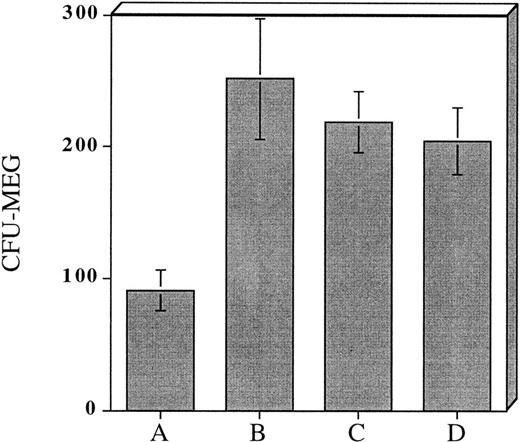
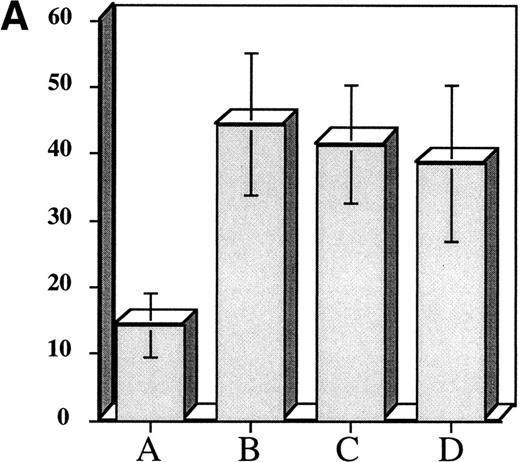
The above experiments suggested that exposure of hematopoietic progenitor cells to an appropriate cytokine cocktail before transplantation might lead to accelerated platelet count recovery in animals transplanted with these cells. To test this hypothesis we exposed 104 adherent cell depleted murine marrow mononuclear cells to a cytokine cocktail consisting of (IL-3 [10 ng/mL] + TPO [50 ng/mL] + IL-1α [100 pg/mL] + KL [10 ng/mL]) to determine if murine CFU-Meg would expand in a manner similar to human progenitor cells. We also assessed the effect of cytokine priming on expansion of CFU-GM. After cytokine exposure, the murine CFU-Meg were manipulated in the same manner as human CFU-Meg, ie, they were either cultured immediately following cytokine stimulation; frozen after stimulation and then thawed and cultured; or frozen, thawed, and then stimulated for 36 hours before immediate plating in plasma clots containing IL-3 + TPO. CFU-GM were plated immediately after cytokine exposure. We found that short-term cytokine exposure of murine progenitor cells expanded CFU-Meg ∼threefold, regardless of when in relation to freezing cells were treated (Fig 3A). CFU-GM were expanded ∼twofold (Fig3B).
Effect of short-term cytokine stimulation on expansion of murine CFU-Meg (Panel A) and CFU-GM (Panel B). Adherent cell-depleted murine marrow mononuclear cells were exposed to cytokines for 36 hours as detailed in the text. Panel A: (A) CFU-Meg colonies (mean ± SD) derived from cells (104) cultured immediately; (B) CFU-Meg colonies (mean ± SD) derived from cells (104) cultured serum-free for 36 hours in presence of (TPO + KL + IL-1α + IL-3); (C) CFU-Meg colonies (mean ± SD) derived from cells (104) cytokine stimulated before cryopreservation; (D) CFU-Meg colonies (mean ± SD) derived from cells (104) cytokine stimulated after thawing. Data are pooled from three independent experiments, each performed in quadruplicate. Panel B: (A) CFU-GM colonies (mean ± SD) derived from cells (104) cultured immediately; (B) CFU-GM colonies (mean ± SD) derived from cells (104) cultured serum-free for 36 hours in presence of (TPO + KL + IL-1α + IL-3). Data are pooled from three independent experiments, each performed in triplicate.
Effect of short-term cytokine stimulation on expansion of murine CFU-Meg (Panel A) and CFU-GM (Panel B). Adherent cell-depleted murine marrow mononuclear cells were exposed to cytokines for 36 hours as detailed in the text. Panel A: (A) CFU-Meg colonies (mean ± SD) derived from cells (104) cultured immediately; (B) CFU-Meg colonies (mean ± SD) derived from cells (104) cultured serum-free for 36 hours in presence of (TPO + KL + IL-1α + IL-3); (C) CFU-Meg colonies (mean ± SD) derived from cells (104) cytokine stimulated before cryopreservation; (D) CFU-Meg colonies (mean ± SD) derived from cells (104) cytokine stimulated after thawing. Data are pooled from three independent experiments, each performed in quadruplicate. Panel B: (A) CFU-GM colonies (mean ± SD) derived from cells (104) cultured immediately; (B) CFU-GM colonies (mean ± SD) derived from cells (104) cultured serum-free for 36 hours in presence of (TPO + KL + IL-1α + IL-3). Data are pooled from three independent experiments, each performed in triplicate.
Because murine and human marrow cells appeared to expand in an essentially identical manner, we employed a murine transplant model to test the ability of ex vivo stimulated marrow cells to accelerate platelet recovery. Lethally irradiated mice were transplanted with hematopoietic progenitor cells that were preincubated for only 36 hours with a cytokine mixture that consisted of (rHTPO + rH IL-1α + rM KL + rM IL-3). In addition, because we were aware of reports which suggested that exposure of progenitor cells to so-called “early acting” cytokines might impair long-term engraftment of transplanted cells,20-24 we primed only a small part of the marrow graft (∼10% of transplanted cells) to avoid this potential complication. Accordingly, marrow harvested from donor mice was divided into two aliquots. One, consisting of the majority of cells (∼90%) was cryopreserved after processing without exposure to cytokines. The other aliquot (∼10%) of cells was primed by 36 hours exposure to the cytokine combination just described. Priming was performed either before cryopreservation or immediately after thawing of cells to be transplanted. At the time of transplantation primed cells were mixed with unexposed marrow cells in a ratio of 1:10.
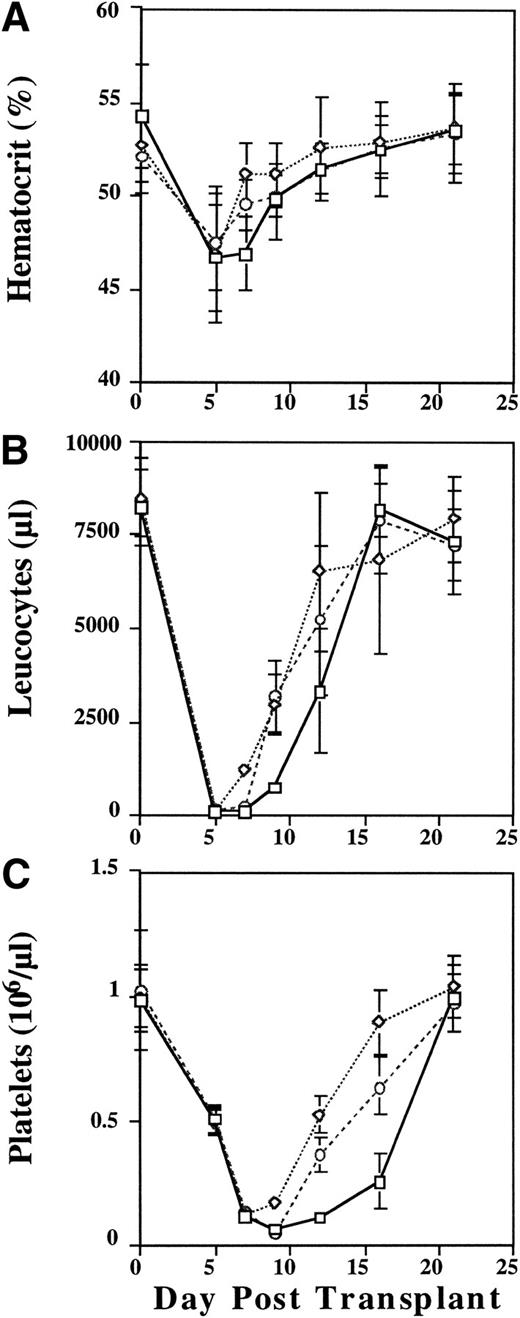
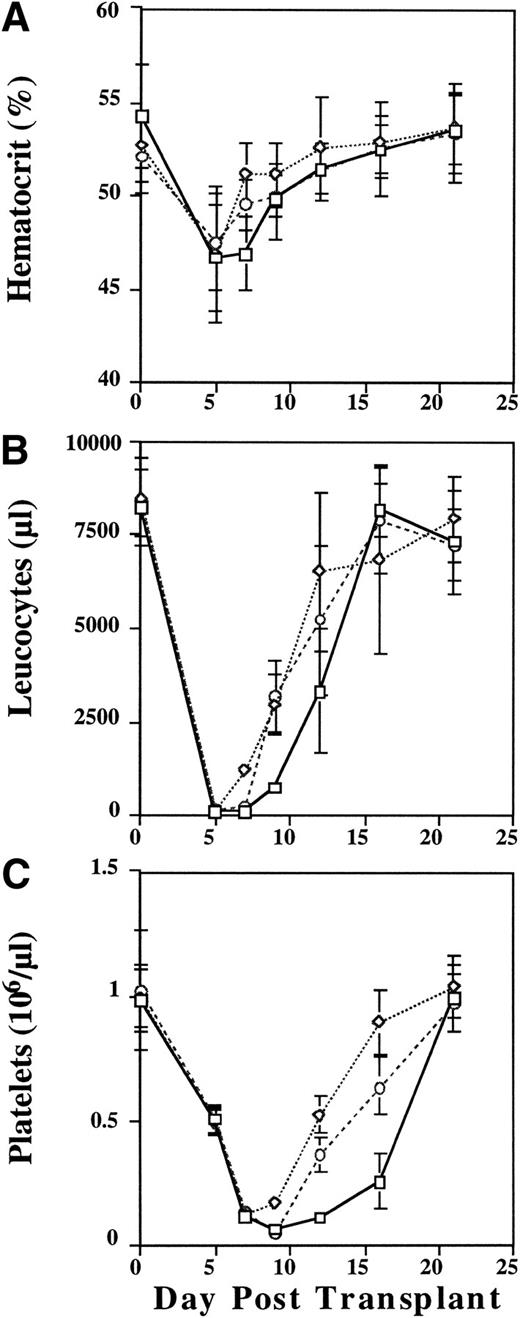
Mice transplanted with cytokine primed cells showed slightly improved rates of recovery of hematocrit (Fig 4A),and enhanced reconstitution of leukocyte counts (Fig 4B). Most germane to this report, however, were the effects of cytokine priming on the animals' platelet count nadirs and recovery kinetics (Fig 4C). As shown in Fig 4C, the nadir platelet count in the animals transplanted with unprimed donor cells occurred at day 9. While the mean day 9 platelet counts were not significantly different between the control group (56,500/μL) and the animals that were transplanted with cells that had been frozen/thawed and then cytokine stimulated (F/T/S) immediately before transplantation (48,200/μL), the difference between day 9 platelet counts in the control group and animals which had been transplanted with cells that were cytokine stimulated before being frozen, thawed, and then transplanted (S/F/T) was highly significant (125,300/μL; P < .001). In addition, while the platelet count nadirs occurred at day 9 for both the control animals and those which received F/T/S marrow cells, the platelet nadir count for those animals which received S/F/T marrow cells occurred on day 7 (P < .001).
Peripheral blood hematocrit (%) (A), leukocyte (B), and platelet (C) counts in lethally irradiated mice transplanted with unprimed (control) donor cells (□), donor cells that were cytokine stimulated, frozen, and then thawed (S/F/T) (◊), or cells that were frozen, thawed, and then cytokine stimulated (F/T/S) (○) before transplantation. Leukocyte counts are absolute per μL, platelet counts are (×106/μL). Data are pooled from two independent experiments each with 5 to 10 mice/group.
Peripheral blood hematocrit (%) (A), leukocyte (B), and platelet (C) counts in lethally irradiated mice transplanted with unprimed (control) donor cells (□), donor cells that were cytokine stimulated, frozen, and then thawed (S/F/T) (◊), or cells that were frozen, thawed, and then cytokine stimulated (F/T/S) (○) before transplantation. Leukocyte counts are absolute per μL, platelet counts are (×106/μL). Data are pooled from two independent experiments each with 5 to 10 mice/group.
We also examined the rate of platelet count recovery using a random effects linear model. Analysis of platelet count recovery from day 9 (nadir) through day 16 showed an average increase in platelet count of 28,300/μL/d in the control group of animals. In the F/T/S group the rate of increase was estimated to be 83,250/μL/d (z = 8.37,P < .001) while in the S/F/T group it was 103,150/μL/d (z = 11.23, P < .001). It is important to note that lethally irradiated control animals died by day 18 without signs of engraftment.
DISCUSSION
Depending on the source of hematopoietic stem cells, and the patients' underlying disease, restoration of hematopoiesis in transplant recipients can take as few as 7 days or up to a month or longer.4,25 During the period of marrow aplasia/hypoplasia, the patient is at considerable risk for infectious complications. Anemia and thrombocytopenia are more amenable to intervention, but transfusion therapy is not completely benign. Further, if a patient becomes alloimmunized, platelet transfusions are largely ineffective at controlling thrombocytopenic bleeding. To surmount these problems, patients are often infused with hematopoietic cytokines in an attempt to accelerate marrow growth.5-9 There is little doubt that presently available cytokines, in particular G-CSF and GM-CSF, accelerate neutrophil recovery by several days in comparison to patients who are not treated with hematopoietic growth factors.26,27 This strategy for accelerating myelopoietic recovery has been reported to be clinically useful by some investigators,28 but not by others.29 In no case have consistent effects on platelet count recovery, in the absence of clinically significant toxicity, been reported. Quite recently, IL-11 and TPO have entered into clinical trial but the utility of these growth factors remains unproved. However, even if effective it would not be unreasonable to expect that routine use of these, and other cytokines, would add significantly to the cost of transplantation and might also increase the morbidity of the procedure.
With the above considerations in mind, we began to explore alternative strategies for approaching the problem of posttransplant thrombocytopenia. We hypothesized that by expanding the number of megakaryocyte progenitors in the hematopoietic cell graft before transplantation, such cells might be primed to proliferate relatively rapidly upon homing to the host microenvironment. Proliferation in turn would be accompanied by maturation of platelet shedding megakaryocytes and consequent alleviation of thrombocytopenia. To test this hypothesis, we needed to design a viable strategy for CFU-Meg expansion. Our approach was based on the premise that use of serum-free culture medium, and the appropriate cytokines, would synergistically promote megakaryocyte progenitor cell expansion by stimulating cell proliferation in the absence of known inhibitors of megakaryocyte growth such as transforming growth factor-β and PF4.10-12There were reported already attempts to stimulate bone marrow cells before transplantation with cytokines yielding different results.28,29 We were also cognizant of reports that cytokines useful for stem/progenitor cell expansion, such as IL-1α, IL-3, IL-6 and KL, while immediately useful for progenitor cell expansion, might prove detrimental for long-term engraftment by downregulating cytoadhesion molecules necessary for stem cell homing.20-30 To obviate this problem we split the cells we wished to transplant into two separate aliquots. One of the aliquots consisted of the majority of cells (∼90%) and was transplanted unmanipulated. The other aliquot was exposed to a cytokine cocktail designed to promote the expansion of CFU-Meg.
In support of our hypothesis, and in accord with previously reported serum-free expansion systems designed to expand long-term culture initiating cells (LTCICs), colony-forming unit–granulocyte-macrophage (CFU-GM), and burst-forming unit erythroid,31-33 we found that megakaryocyte progenitor cells present in CD34+ enriched, as well as adherent cell depleted, marrow mononuclear cells were greatly expanded when cultured under serum-free conditions. We also found that with an appropriate cytokine cocktail, (TPO + KL + IL-1α + IL-3), the period of exposure required to obtain up to a threefold expansion of these cells was quite brief, between 36 and 48 hours. Cytokine exposed marrow cells also appeared to acquire an increased proliferative capacity as reflected by an increased number of megakaryocytes comprising colonies arising after 10 and 14 days of prestimulation (Table 1). Of interest, it also appeared to us that cytokine primed progenitor cells formed colonies in vitro at a faster rate (∼2 days) than cells which were not so treated. These results supported the idea that transplantation of primed cells in vivo might lead to early enhancement of platelet production and this in fact was shown in an in vivo model employing murine cells (Fig 4). To provide data that our approach might be useful in patients, we used a murine transplant model to test our hypothesis. This enabled us to show that transplantation of ex vivo expanded megakaryocyte progenitor cells resulted in platelet recovery 3 to 5 days faster than syngeneic animals who were transplanted with nonprimed cells. Interestingly, it appeared that these animals also had accelerated engraftment of CFU-GM as evidenced by the similar acceleration of neutrophil recovery in these animals (Fig 4B).
Presently, we cannot be certain of the mechanism(s) responsible for the observed acceleration of platelet engraftment. Since enhanced platelet recovery was observed in mice that received marrow cells primed for as little as 36 hours, it seems unlikely that enhanced proliferative capacity of progenitors is responsible for the effects we have observed (Table 1). Nevertheless, it was our impression that morphologically identifiable megakaryocyte colonies appeared earlier in cultures initiated with cytokine primed cells. We hypothesize then that enhanced platelet recovery is likely the result of transplanting an increased number of progenitors with an accelerated ability to complete their maturation program. Finally, while any regimen of cytokine priming appeared to accelerate platelet count recovery, statistical analysis of the data suggested that cells which were cytokine primed before being frozen engrafted and developed earlier as reflected by a higher nadir platelet count, and an earlier and more rapid recovery in animals transplanted with such cells (Fig 4C). Based on these results, we suggest that short-term stimulation of hematopoietic cells in serum-free cultures before cryopreservation will enhance platelet, and perhaps neutrophil, recovery in transplant patients in a timely and cost-efficient manner. Clinical trials designed to test this hypothesis appear warranted and we hope to begin such trials in the very near future.
Presented at the XXVIII Annual Meeting of the American Society of Hematology, held in Orlando, FL, December 6-10, 1996.
Address reprint requests to Alan M. Gewirtz and Mariusz Z. Ratajczak, Room 514 Stellar-Chance Laboratories, University of Pennsylvania School of Medicine, 422 Curie Blvd, Philadelphia, PA 19104.
The publication costs of this article were defrayed in part by page charge payment. This article must therefore be hereby marked “advertisement” in accordance with 18 U.S.C. section 1734 solely to indicate this fact.