To investigate the T-lymphopoietic capacity of human adult bone marrow (ABM) hematopoietic progenitor cells, CD34+Lin−, CD34+CD38+, and CD34++CD38− cells were cultured in a severe combined immunodeficient (SCID) mouse fetal thymic organ culture (FTOC). Direct seeding of these progenitors resulted in a moderate to severe cell loss, particularly for the CD34++CD38− cell fraction, and T cells could only be generated from the CD34+Lin− fraction. Preincubation for 36 hours with interleukin-3 (IL-3) and stem cell factor (SCF) led to an improved cell survival and proliferation, although T-cell development was seen only in the CD34+Lin− fraction. Addition of tumor necrosis factor (TNF)- to IL-3 + SCF-supplemented preincubation medium resulted in optimal cell survival, cell proliferation. and T-cell generation of all 3 cell fractions. The TNF- effect resulted in an up-regulation of CD127 (ie, the IL-7 receptor -chain) in a small subset of the CD34+ cells. No evidence could be generated to support the possibility that TNF- inhibits a cell population that suppresses T-cell differentiation. A quantitatively different T-cell generation potency was still seen between the 3 subpopulations: CD34+Lin− (100% success rate) > CD34+CD38+ (66%) > CD34++CD38− (25%). These data contrast with our previous findings using fetal liver and cord blood progenitors, which readily differentiate into T-lymphocytes in FTOC, even without prestimulation with cytokines. Our results demonstrate that adult CD34++CD38− cells, known to contain hematopoietic stem cells, can differentiate into T-lymphocytes and that a significant difference exists in T-lymphopoietic activity of stem cells derived from ontogenetically different sources.
Human CD34+ cells contain at least part of the hematologic progenitor and stem cells. Multipotent hematopoietic stem cells are characterized by their putative ability for self-renewal and differentiation into both the myeloid (ie, granulocytic, monocytic, erythrocytic, and thrombocytic) and the lymphoid (ie, T cell, B cell, and natural killer [NK] cell) lineages. Hematopoietic progenitors, on the other hand, are already committed to 1 or several of these lineages. For a long time, lack of appropriate assays allowing in vitro T-cell development has hampered progress in understanding T-lymphopoiesis. Recently, however, functional analysis of T-cell development has become possible after the introduction of techniques in which the thymic microenvironment is mimicked, such as the in vitro fetal thymic organ culture (FTOC) system.1-4 We have recently described a method in which human fetal liver (FL) precursor cells are transferred to severe combined immunodeficient (SCID) murine fetal thymic lobes, following which the lobes are cultured in a classical FTOC as described by Jenkinson.4,5 This in vitro method, combined with the possibilities of detailed phenotypic analysis, provides a unique tool to study early thymopoiesis and the critical factors involved in this process.6
In spite of intensive research on T-lymphopoiesis using these techniques, the question whether stem cells differentiate into committed T-cell progenitors before or after entry into the thymus remains unresolved. Although Rodewald et al7 showed that murine T-cell commitment can occur before entry into the thymus, recent data suggest that, in humans, CD34+ progenitors derived from cord blood (CB) and fetal bone marrow (FBM) do not undergo such a commitment before they migrate to the thymus.8
As shown by us and by others,4,8-10 human CD34+ FL and CB cells readily generate T cells in FTOC. Stem cells derived from ABM seem to be a more difficult source from which to generate T cells in vitro. Galy et al2,3 studied several subsets of ABM CD34+ cells and found that a phenotypically defined subset of CD34+Lin−CD45RA+ progenitor cells, which were CD38+CD10+HLA-DR+Thy-1−c-kit−, were able to produce T cells, though they were devoid of myeloid, erythroid, and megakaryocytic potential. Because at least part of the hematopoietic stem cells are contained within the ABM CD34++CD38− cell fraction,11-13 we compared the T-lymphopoietic capacity of 3 subpopulations of human ABM CD34+ cells. We first evaluated survival, proliferation, and T-cell generation of a CD34+ cell population devoid of the lymphocytic lineage-specific cell surface markers CD2, CD7, and CD19 (CD34+Lin−). Next, we investigated CD34++CD38− and CD34+CD38+ cells, the latter being less primitive cells containing differentiated progenitors. We furthermore studied the effects of a 36-hour preincubation of these cells with IL-3 + SCF ± TNF-α. It has indeed been shown that IL-3 + SCF can release hematopoietic stem cells from their G0 cell-cycle status.14Moreover, incubation of ABM CD34++CD38− cells with IL-3 + SCF with TNF-α resulted in a potent stimulation of myeloid proliferation and differentiation, significantly and markedly more than the combination of IL-3 + SCF without TNF-α.15 TNF-α has been shown by Zúñiga-Pflücker et al16 to be related to the induction of crucial events that lead to T-lineage commitment and differentiation in mice. Therefore, we investigated the effects of TNF-α on the generation of T-lymphocytes from IL-3 + SCF-stimulated human ABM.
Materials and methods
Adult bone marrow cells
Bone marrow samples were aspirated by sternal puncture from hematologically normal patients undergoing cardiac surgery and were obtained from healthy bone marrow donors who gave marrow for related family recipients. Cells were collected into sterile collection tubes containing complete medium (Iscove's modified Dulbecco's medium [IMDM] / 10% fetal calf serum [FCS]; Gibco, Paisley, UK) and heparin (100 U/mL; Novo Nordisk, Bagsvaerd, Denmark) and were isolated by density-gradient centrifugation over lymphocyte separation medium (ICN Biomedicals, Costa Mesa, CA). Cells were washed and resuspended in 1.8 mL 90% FCS / 10% dimethyl sulfoxide solution and frozen in liquid N2. Samples were obtained after informed consent according to the guidelines of the Medical Ethics Committees of the University Hospitals of Ghent and Antwerp.
Cytokines and monoclonal antibodies
Supernatant of the 43A1 hybridoma (IgG3, kindly donated by Dr H. J. Bühring, University of Tübingen, Germany) was used as a source of anti-CD34 antibodies.17 Fluorescein isothiocyanate (FITC)-conjugated rabbit antimouse (RAM) immunoglobulins (F[ab′]2 fragments) were purchased from Dako (Glostrup, Denmark). Phycoerythrin (PE)-conjugated anti-CD38 and anti-CD34 antibodies and FITC-conjugated anti-CD2, anti-CD7, and anti-CD19, as well as isotype-specific control antibodies, were purchased from Becton Dickinson (Erembodegem, Belgium), and mouse gamma-globulins were purchased from Jackson ImmunoResearch Laboratories (West Baltimore Pike, PA). Recombinant human (rhu) stem cell factor (SCF) (specific activity, more than 1 × 105 U/mg) and rhuTNF-α (specific activity, more than 1 × 108 U/mg) were obtained from Boehringer Mannheim GmbH (Penzberg, Germany), and rhu interleukin-3 (IL-3) (biological activity, 14 × 103 U/mL) was a kind gift of Dr S. C. Clark (Genetics Institute, Cambridge, MA).
Animals
C.B.-17 scid/scid (SCID) mice, originally purchased from Iffa Credo (l'Abresle, France), were bred in our own pathogen-free breeding facility. To obtain timed pregnancies, female and male mice were mated overnight and the day of the plug was considered day 0. Fetal thymic lobes were dissected from embryos at day 14 to 15 of gestation. Mice were treated and used in agreement with the institutional guidelines.
Cell sorting
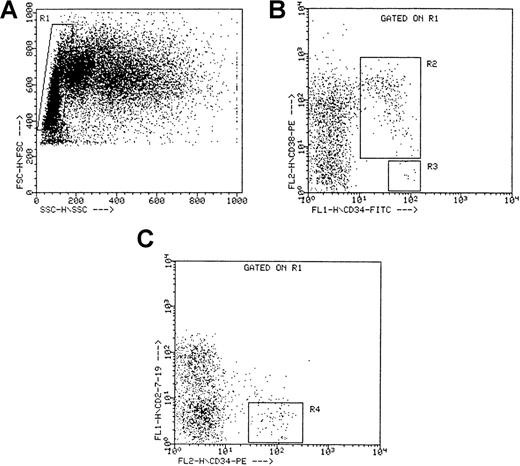
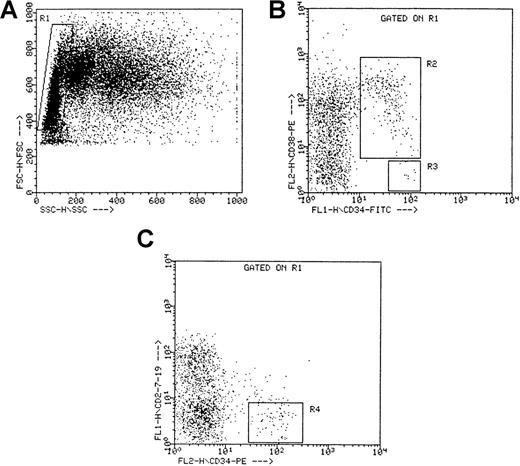
Viably frozen CB and ABM cells were thawed at 37°C, incubated for 10 minutes in a solution containing 100 μL 0.3 mol/L MgSO4, 100 μL DNase (specific activity, approximately 2000 U/mg; Boehringer Mannheim GmbH), 100 μL heparin (5000 U/mL), and 1.5 mL IMDM / 10% FCS, washed in IMDM and resuspended in IMDM / 10% FCS. After an incubation of another 20 minutes, the cells were washed again, resuspended at 107 cells/mL, incubated with 43A1 supernatant in a 1/10 dilution for 20 minutes at 4°C, washed twice in IMDM / 10% FCS, and incubated with RAM–FITC (1/50 dilution) for 20 minutes at 4°C. After washing twice in IMDM / 10% FCS, the cells were incubated with a 10-fold excess of mouse gamma-globulins for 10 minutes to avoid nonspecific staining and were incubated with anti-CD38−PE for 20 minutes at 4°C. After washing twice in IMDM / 10%FCS, the cells were sorted on a FACStarPlus Cell Sorter (Becton Dickinson) equipped with an air-cooled argon ion laser (ILT model 5500A; Ion Laser Technology, Salt Lake City, UT), tuned to 488 nm at 40-mW power. Cells with a low to medium forward scatter and a low side scatter, a highly positive green (CD34) fluorescence, and an orange (CD38) fluorescence signal lower than the mean fluorescence of cells labeled with an irrelevant isotype-matched control antibody plus 2 SD were retained as CD34+CD38− cells; cells with an orange fluorescence above this threshold were retained as CD34+CD38+ cells. For the CD34+Lin− cell fraction, thawed and washed cells were incubated with anti-CD34–PE and with FITC-conjugated anti-CD2, anti-CD7, and anti-CD19 for 20 minutes at 4°C and then were washed and sorted as described for the CD34+CD38− cells. Purities were always greater than 95%. Sorting regions are depicted in Figure1.
Sorting criteria for CD34++CD38− , CD34+CD38+, and CD34+Lin− cells.
(A) Side (SSC) versus forward (FSC) scatter for ABM mononuclear cells. The rectangular region (R1) defines the gated population used for sorting. (B) Fluorescent intensities of CD34−FITC and CD38−PE for all cells within the gated region R1. R2 defines the gated population used for sorting the CD34+CD38+ cells, and R3 defines the gated population used for sorting the CD34++CD38− cells. (C) Fluorescent intensities of CD34−PE and CD2/CD7/CD19−FITC for all cells within the gated region R1. R4 defines the gated population used for sorting the CD34+Lin− cells. Dot plots represent the sorting criteria for a single ABM experiment, but similar plots were obtained for the other ABM experiments.
Sorting criteria for CD34++CD38− , CD34+CD38+, and CD34+Lin− cells.
(A) Side (SSC) versus forward (FSC) scatter for ABM mononuclear cells. The rectangular region (R1) defines the gated population used for sorting. (B) Fluorescent intensities of CD34−FITC and CD38−PE for all cells within the gated region R1. R2 defines the gated population used for sorting the CD34+CD38+ cells, and R3 defines the gated population used for sorting the CD34++CD38− cells. (C) Fluorescent intensities of CD34−PE and CD2/CD7/CD19−FITC for all cells within the gated region R1. R4 defines the gated population used for sorting the CD34+Lin− cells. Dot plots represent the sorting criteria for a single ABM experiment, but similar plots were obtained for the other ABM experiments.
Phenotypic analysis
Phenotypic analysis at day 0 and after 36 hours was performed on a FACScan Cell Sorter (Becton Dickinson) equipped with an air-cooled argon ion laser (Spectra-Physics Lasers, Mountain View, CA) using the following antibodies: PE-conjugated—anti-CD127 (Immunotech, Marseille, France); anti-CD7 (PharMingen, San Diego, CA); anti-CD38 (Becton Dickinson); anti-CD2 (Becton Dickinson); anti-CD4 (Becton Dickinson); FITC-conjugated—anti-CD34 (Becton Dickinson); phycoerythrin–cyanin-(PC-)5-conjugated—anti-CD13 (Immunotech); anti-CD19 (Immunotech); anti-CD33 (Immunotech); and tricolor (TC)-conjugated—anti-CD14 (Caltag, Burlingame, CA). In most staining series, the appropriate isotype control antibodies were included and were found to be negative.
FTOC technique
Preincubation assays were started by performing liquid cultures in 24-well flat-bottomed plates in IMDM with 10% FCS and 100 ng/mL SCF + 20 U/mL IL-3 with or without 1 ng/mL TNF-α. Cells were transferred to fetal murine lobes by the hanging drop method for 48 hours, followed by culture of the lobes in FTOC as described previously.4 After 28 days of FTOC, the total cell number in each disrupted thymic lobe was assessed, together with the human cell fraction, which was measured by flow cytometry after staining with monoclonal antibodies directed against human CD45 and mouse CD45. Dead cells were gated out by propidium iodide exclusion.
Statistics
Statistical comparisons were validated using the nonparametric Wilcoxon signed rank test (paired samples) and the nonparametric Mann-Whitney U test (unpaired samples). Results are expressed as mean ± SEM.
Results
Phenotypic analysis and effects of preincubation on cell number and flow cytometric scatter profile
Different cell populations from the mononuclear ABM cell fraction were isolated by fluorescence-activated cell sorting. Combinations of CD34 with other cell surface markers (CD38, CD2, CD7, and CD19) were used. Three CD34-positive subpopulations were thus isolated: CD34++CD38− cells, CD34+CD38+ cells, and CD34+Lin− cells (CD2−CD7−CD19−). The CD38− fraction was CD34-positive with high density (CD34++CD38−) because the average CD34 fluorescence was 82.4% ± 10.4% higher than that for the CD34+CD38+ fraction (P < .001; Figure 1). Phenotypic analysis showed that 1.7% ± 0.3% of the ABM mononuclear cells were CD34+. Within this CD34+ population, 6.2% ± 1.8% were CD38−, 93.8% ± 1.8% were CD38+, and 91.1% ± 1.2% were Lin−. Sorting regions for each of the 3 cell fractions are depicted in Figure 1.
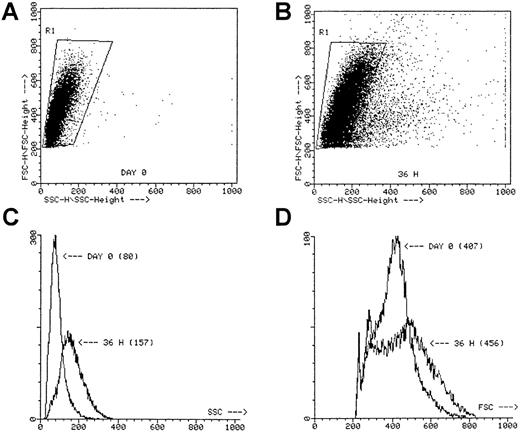
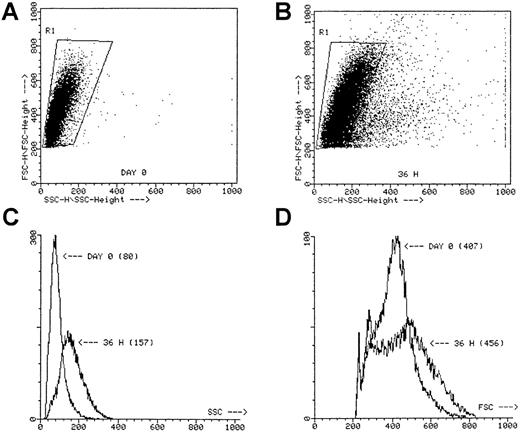
Incubation of these cell fractions for 36 hours in the presence of IL-3 + SCF with or without TNF-α did not result in significant cell proliferation, though the forward and side scatter profile strikingly shifted in all 3 fractions, especially after incubation with TNF-α (Figure 2). Moreover, microscopic evaluation of the cell fractions incubated with TNF-α always revealed the presence of numerous clusters containing 25 or fewer cells, whereas in the absence of TNF-α only dispersed cells were seen (not shown).
Scatter profile of sorted ABM CD34+ cells.
SSC versus FSC at day 0 (A) and after a 36-hour preincubation with IL-3 + SCF + TNF-α (B). The SSC histogram (C) and the FSC histogram (D) of the cells gated in R1. Numbers between parentheses, mean channel number. Dot plots represent the scatter profiles for a single ABM CD34+ experiment, but similar plots were obtained for the other ABM experiments when using CD34++CD38−, CD34+CD38+, and CD34+Lin− cells.
Scatter profile of sorted ABM CD34+ cells.
SSC versus FSC at day 0 (A) and after a 36-hour preincubation with IL-3 + SCF + TNF-α (B). The SSC histogram (C) and the FSC histogram (D) of the cells gated in R1. Numbers between parentheses, mean channel number. Dot plots represent the scatter profiles for a single ABM CD34+ experiment, but similar plots were obtained for the other ABM experiments when using CD34++CD38−, CD34+CD38+, and CD34+Lin− cells.
FTOC supports proliferation of human ABM CD34+ subpopulations
We first investigated whether FTOC supports survival and potential proliferation of the 3 different CD34+ subpopulations derived from human ABM. We seeded the CD34+ subpopulations, with or without a 36-hour preincubation in IL-3 + SCF or IL-3 + SCF + TNF-α, into single thymic lobes of day 14 to 15 fetal SCID mice (Table1). After 28 days of FTOC, we assessed the total cell number and the human cell fraction in each disrupted thymic lobe. Using these parameters, the total human cell number per 1000 input cells was calculated. As shown in Table2, seeding of cells without preincubation resulted in a moderate to severe loss particularly for the CD34++CD38− cell fraction. However, taking into account the fact that only 20% (ie, 200 cells/1000 input cells) of the seeded cells actually enter the thymic lobe during the hanging drop time period of 48 hours,4 our results clearly show that the FTOC assay at least sustains the survival of human ABM cells, and it may even promote proliferation (up to 3-fold) provided the cells have been preincubated with IL-3 + SCF with or without TNF-α (Table 2). This is in strong contrast with our previous findings whereby we showed that after 4 weeks of FTOC, FL and CB CD34+Lin− cells without cytokine prestimulation showed a 60-fold and a 20-fold cell expansion, respectively.4 9
CD34+ subpopulations from ABM differentiate into T cells in FTOC
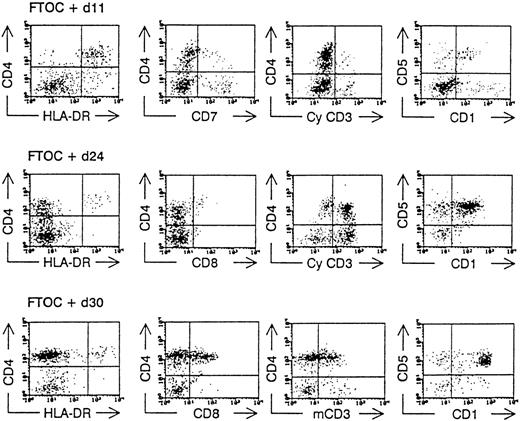
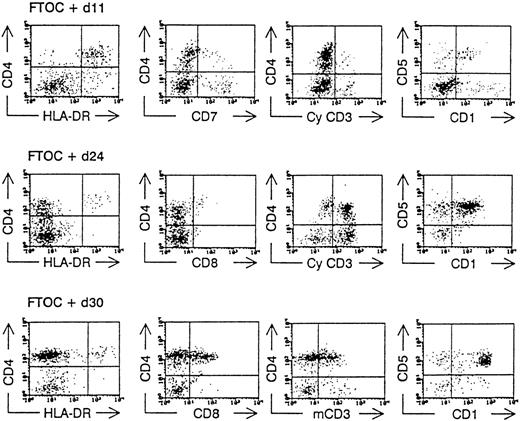
Human T-lymphopoiesis passes through different phenotypic maturation steps before giving rise to mature CD4+ and CD8+ T cells. As we have shown previously for FL and CB stem cells, the following stages can be demonstrated using FTOC: part of the CD34++CD38−CD4−CD8−CD1− cells develop rapidly into dendritic cells that are CD4+HLA−DR+cytoplasmic(cy)- CD3−CD7−, and CD1−/+. The T-cell differentiation proceeds through a CD4+cyCD3+CD7+CD1+HLA-DR− intermediate stage, followed by a CD4+CD8+membrane(m)CD3+CD1+CD5+ “double-positive” (DP) stage before achieving the end maturation stage, resulting in a CD4+CD8−CD3+CD1− and CD4−CD8+CD3+CD1− phenotype.4,9,18 Because of the low number of immature progenitor cells at the start of the FTOC (resulting from the low frequency of the CD34+ subpopulations in ABM) and because the proliferative potential of these cells was limited, we examined the presence of human CD4+CD8+ DP T cells, which were mCD3+ in the FTOC at 1 time point (day 28). The presence of these human CD4+CD8+mCD3+ cells indicates an active T-lymphopoiesis starting from the seeded stem cells.4,6 19 In 1 experiment, however, we were able to analyze the kinetics of the T-cell development from ABM CD34+Lin− cells and found the same differentiation markers as mentioned above (Figure 3).
Kinetic analysis of the development of ABM CD34+Lin− cells in hybrid FTOC.
CD34+Lin− ABM cells were sorted and cultured together with mouse thymic lobes. After various incubation times (as indicated), the lobes were minced and a flow cytometric analysis was performed after staining the cells with monoclonal antibodies directed against human antigens as indicated in the axis of the dot plot. CyCD3, cytoplasmic CD3; mCD3, membrane CD3.
Kinetic analysis of the development of ABM CD34+Lin− cells in hybrid FTOC.
CD34+Lin− ABM cells were sorted and cultured together with mouse thymic lobes. After various incubation times (as indicated), the lobes were minced and a flow cytometric analysis was performed after staining the cells with monoclonal antibodies directed against human antigens as indicated in the axis of the dot plot. CyCD3, cytoplasmic CD3; mCD3, membrane CD3.
As shown in Table 3, starting from unstimulated CD34+ cells, only the CD34+Lin− cells generated CD4+CD8+ DP T cells in 3 of 7 experiments analyzed at day 28, yielding 7.5% ± 4.0% DP cells. No DP T cells were generated in the CD34++CD38− and the CD34+CD38+ subfractions, except for 1 CD34+CD38+ experiment, in which traces (less than 0.1%) of DP cells were found. Preincubation of the CD34+Lin− cells during 36 hours in the presence of IL-3 + SCF or IL-3 + SCF + TNF-α did not result in a significant rise in DP T-cell generation. Starting from the CD34++CD38− and the CD34+CD38+ cell fractions, only traces of DP T cells were generated after preincubation with IL-3 + SCF. Addition of TNF-α to this preincubation cocktail resulted in a significantly higher output of DP T cells for CD34+CD38+ 5.9% ± 2.3% (P = .008) and CD34++CD38− 6.0% ± 4.1% (P = .03) cells. In terms of absolute DP T-cell generation/1000 input cells, Table 4clearly shows that we were able to generate T cells starting from all 3 different CD34+ subpopulations, with an optimal output after preincubation with IL-3 + SCF + TNF-α.
Preincubation with TNF- stimulates ABM stem cells to generate CD4+CD8+ T cells
To make a qualitative assessment of the effects of TNF-α on the generation of human T cells, we analyzed the pattern of T-cell development in the FTOC, which resulted in a classification with 3 distinctive growth patterns. In pattern A, only murine CD45+ cells and no or few (less than 0.1%) human CD45+ cells were found after 28 days of FTOC. Pattern B was defined by the presence of human CD45+ cells that could develop into CD4+CD8−CD3− single positive cells but of which less than 0.5% reached the CD4+CD8+ stage. In pattern C, a clearly distinguishable population of human double-positive CD4+CD8+ T cells could be observed. As summarized in Table5, only part (43%) of the CD34+Lin− samples could generate T cells without prior preincubation, whereas none of the CD34++CD38− or of the CD34+CD38+ fractions developed beyond the CD4 single-positive stage, not even after a 36-hour preincubation with SCF + IL-3. It was only after adding TNF-α to IL-3 + SCF that 25% of the experiments using CD34++CD38− cells, 66% of those using CD34+CD38+ cells, and 100% of those using CD34+Lin− samples generated CD4+CD8+ T cells (Table 5).
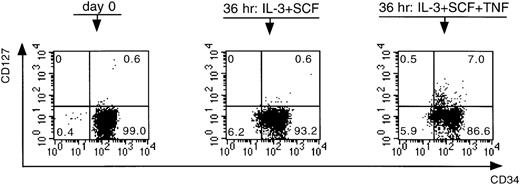
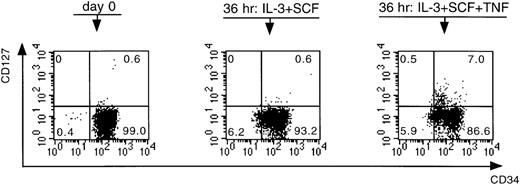
To understand the mode of action of TNF-α, we first performed extensive phenotypic analyses on CD34+Lin− cells preincubated in the presence and absence of TNF-α. We performed these experiments on CD34+Lin− cells for two reasons. First, to analyze a maximum number of markers on each bone marrow sample, an adequate cell number is required, making it almost impossible to use the low-frequency CD34++CD38− cell population for this purpose. Second, as shown in Table 5, preincubation of CD34+Lin− cells in the presence of TNF-α resulted in a C pattern in 100% of the experiments, which was in contrast to the outcome of the growth patterns of the CD34+CD38+ and the CD34++CD38− cell fractions, making the CD34+Lin− fraction the most interesting population to use for phenotypic analysis. The following markers were analyzed: CD34, CD33, CD13, CD38, CD2, CD4, CD7, CD14, and CD127 (ie, IL-7Rα). Preincubation of the cells resulted in a down-regulation of CD34, an up-regulation of CD13 and CD33, a slight up-regulation of CD38, and no effect on CD19 and CD7 expression. There was also an up-regulation of CD14 and of CD4, but all the CD4+ cells were also positive for CD14. Preincubation furthermore resulted in the appearance of a small population of CD2+ cells, though no difference was seen between the cells preincubated with or without TNF-α. Interestingly, in all samples analyzed, a small but clearly distinguishable population of CD127+ cells became evident only after preincubation in the presence of TNF-α (P = .01) (Figure 4).
Effect of IL-3 + SCF and IL-3 + SCF + TNF- on the expression of CD34 and CD127.
CD34+Lin− cells were analyzed at day 0 immediately after cell sorting and after a preincubation period of 36 hours. Numbers in quadrants refer to percentage of cells within that quadrant. This figure is representative of a series of six independent experiments.
Effect of IL-3 + SCF and IL-3 + SCF + TNF- on the expression of CD34 and CD127.
CD34+Lin− cells were analyzed at day 0 immediately after cell sorting and after a preincubation period of 36 hours. Numbers in quadrants refer to percentage of cells within that quadrant. This figure is representative of a series of six independent experiments.
In a second set of experiments, we performed 6 mixing experiments to examine the possibility that TNF-α might remove a cell population that suppresses T-cell differentiation (Table6). In these experiments, an equal number of cells preincubated in IL-3 + SCF (X in Table 6) and of cells preincubated in IL-3 + SCF + TNF-α (Y in Table 6) were mixed before they were transferred in the hanging drop. After 28 days of FTOC, the A-B-C growth pattern of these mixed cells was compared with that of each of the preincubation settings separately. These mixing experiments did not alter the positive effect of TNF-α on T-cell generation capacity, indicating that an inhibitory population that can be suppressed by TNF-α is not present (Table 6).
In a third set of experiments, we investigated the potential role of accessory cell populations such as dendritic cells. Figure 3 shows that at days 11 to 14, a population with a CD4+HLA-DR+ phenotype is present in the FTOC. This population can be further delineated according to a high forward and side scatter profile, and it is CD14− (data not shown). We have recently shown that these cells represent a dendritic cell population that is CD7− and cyCD3−.20 No difference in dendritic cell frequency was seen after preincubation with IL-3 + SCF with or without TNF-α (17.9% ± 8.1% vs 12% ± 5%, respectively;P = .17), though the absolute dendritic cell numbers in the presence of TNF-α were always higher (264.1 ± 138.6 vs 698.6 ± 220.9, respectively; P = .03).
Discussion
In this report we demonstrate that T cells can be generated from hematopoietic stem cell- and progenitor cell-containing fractions derived from human adult bone marrow in an ex vivo xenotransplant model. In this FTOC assay, human hematopoietic progenitor cells can differentiate to mature T cells in isolated fetal thymic lobes of SCID mice, which have a mutated scid gene, resulting in a deficient murine thymocyte maturation21 but allowing concomitant T-cell development of xenogeneic cells. We furthermore show that T cells can be generated from adult bone marrow, not only from the CD34+Lin− and CD34+CD38+ fractions, which still are rather heterogeneous populations containing differentiated progenitors, but also from the phenotypically and functionally most primitive hematopoietic cells, characterized by their high expression of CD34 and the absence of CD38 expression (CD34++CD38− cells).11-13,22 23
It has previously been shown by others and by us that human hematopoietic stem cell-containing populations derived from different ontogenetic sources such as CB, FBM, or FL can be stimulated to T-cell differentiation in a murine SCID fetal thymic environment. Res et al10 showed that only the CD34++CD38− FL cells had the potential to develop into T cells, whereas their CD34+CD38+ counterparts did not have this capacity. The same group, however, observed that both CD34++CD38− and CD34+CD38+ fractions from FBM and CB were able to generate T cells,8 which was recently confirmed by us.9 However, little information is available on whether T cells can be generated from ABM stem cells and on the possible differences herein between the CD34++CD38− and the CD34+CD38+ cell fractions. Freedman et al24 and Rosenzweig et al25 were able to generate T cells from human ABM cells by using a thymic stromal culture system derived from either fetal human or fetal rhesus macaque thymus glands. However, these authors used high concentrations (1 × 105 cells/well) of CD34+ cells, which are relatively heterogeneous cells containing progenitors already committed to T-cell differentiation.3In addition, because this CD34+ population is globally more differentiated than CD34++CD38− cells,11-13,22,23 it is likely that the early differentiation steps from multipotent progenitor cells to the T-lineage have been missed. On the other hand, Galy et al26 used a SCID–human thymus assay in which irradiated fetal thymuses were injected with human ABM cells, following which the thymuses were inoculated during 6 weeks under the kidney capsule of SCID mice. They found that lymphoid cells can be generated from immature progenitors such as CD34+Lin−Thy-1−CD45RA− cells and CD34+Lin−Thy-1+CD45RA− cells. Both these cell fractions were also able to generate erythroid and myeloid progeny, comparable with the lymphopoietic (this study) and the myelopoietic capacities of CD34++CD38− cells.11,15,22,27 28
Our data further indicate that 36-hour preincubation of ABM cells with IL-3 + SCF before seeding to FTOC improves cell survival and proliferation of the CD34++CD38− fraction (shift from pattern A → B). This is in strong contrast with the findings of Hirayama and Ogawa,29 who show that the addition of IL-3 to murine Lin−Ly-6A/E+ immature hematopoietic progenitors in methylcellulose cultures containing SCF + IL-11 + IL-7 for 8 days strongly suppressed T-lymphopoiesis. This may have been the result of an irreversible commitment of these progenitor cells to the myeloid lineage after 8 days of culture in the presence of IL-3 + SCF. A shorter preincubation period of 36 hours, however, may be just enough to release stem cells from their G0 cell-cycle status without directing them irreversibly to a specific lineage.14 This is supported by the results of Moore and Zlotnik,30 who found that a 3-day incubation of murine thymic CD4lo cells (CD44+CD25−CD3−CD4loc-kit+CD8−cells; ie, the first cells to seed the thymus from the bone marrow) with SCF + IL-3 resulted in the maintenance of cell survival, whereas the addition of IL-7 to this cocktail even induced a capacity for repopulation in FTOC. That IL-7 is also a critical growth factor in early human T-cell development in the FTOC system has recently been confirmed by us.31
The addition of TNF-α to SCF + IL-3 resulted in a T-cell generation success rate (pattern C) of 100% for the CD34+Lin− fraction and in a further shift from pattern B → C (66%) for the CD34+CD38+ cells and from patterns A → B and B → C (25%) for the CD34++CD38− cells (Table 5). These effects of TNF-α, which is produced within the thymic microenvironment, could be related to the induction of crucial events leading to T-lineage commitment and differentiation as shown by Zúñiga-Pflücker et al.16 However, this group could not conclude from their study whether TNF-α acted directly on murine T-cell progenitors or indirectly on a non–T-cell subset that is critical for T-cell maturation. We investigated both possibilities. First, we showed that in all CD34+Lin− samples analyzed, a small but clearly distinguishable population of CD127+ cells appeared only after preincubation with IL-3 + SCF + TNF-α. We hypothesize that TNF-α induces the up-regulation of CD127 on a small fraction of the preincubated cells, resulting in a positive effect (be it direct or indirect) on T-cell generation in the FTOC. This is in agreement with the 1997 findings of Kondo et al,32 who identified a clonogenic common progenitor in mouse bone marrow. This common lymphoid progenitor is Lin− IL-7R + Thy-1−Sca-1loc-Kitlo, and it has a lymphoid-restricted (T, B, and NK) reconstitution capacity in vivo but completely lacks myeloid differentiation potential. To address the role of this CD127+ cell fraction on T-cell generation in a direct manner, it would be interesting to isolate (sort) the CD127+ cells after 36 hours of preincubation and to seed them directly into the thymic lobes of the FTOC. Unfortunately, the low number of cells and the potential of CD127 antibodies to block the development of FL cells in the FTOC31 33 or of sorted CD127+CD34+ intrathymic precursor cells in FTOC (Plum J, unpublished data) present important obstacles to test whether CD127+ cells, generated after incubation with TNF-α , favor T-cell development. In addition, we examined the potential role of the dendritic cells that were generated from CD34+Lin− cells in the FTOC assay. We could not show a difference in dendritic cell frequency after preincubation with IL-3 + SCF with or without TNF-α , though the absolute dendritic cell numbers in the presence of TNF-α were always higher. Because of the overall low absolute cell numbers generated, it was not feasible to investigate further phenotypical and functional (eg, mixed lymphocyte culture assays) properties of these dendritic cells. Therefore, we cannot exclude that the dendritic cells generated in the presence or absence of TNF-α have different functional properties, resulting in an indirect TNF-α effect on T-cell generation. Finally, within the limitations of qualitative analysis when using the FTOC system, the evidence we gathered does not support the hypothesis that TNF-α inhibits a cell population that suppresses T-cell differentiation.
Based on our previous results concerning the stimulatory effect of TNF-α on the myeloproliferative capacity of CD34++CD38− cells, it is possible that TNF-α promotes the proliferation of stem cells and immature progenitor cells, making them more sensitive to T-lymphopoietic influences.15 Another explanation would be that TNF-α (in combination with SCF + IL-3) directly makes the ABM CD34+ cells more able to enter the thymic lobes. Further investigation is required to elucidate the exact role of TNF-α in this matter.
The results presented in this study also revealed important ontogenetic differences in T-cell generation potential between fetal, neonatal, and adult hematopoietic stem cells. From our previous findings, we know that highly purified CD34++Lin− cells from FL develop into T cells in 99% of the FTOC experiments, with a 60-fold cell expansion.4 CB progenitors show a success rate of around 85% (Plum J, unpublished data) and a 20-fold cell expansion.9 Our recent data demonstrate that for ABM stem cells the success rate progressed from 25% (CD34++CD38− cells) to 66% (CD34+CD38+ cells) to 100% (CD34+Lin− cells) and a 1 to 3-fold cell expansion (Tables2 and 5). Different hypotheses can be postulated to explain these differences in success rate and expansion capacity. They may develop because FL cells enter the thymic lobes far more easily than ABM cells, because FL contains more T-cell progenitors than ABM, or because there is an important difference between the proliferative capacity of T-cell progenitors derived from FL and those derived from ABM. Interestingly, the observed differences in T-cell generation potential of FL, CB, and ABM cells when using the FTOC model are comparable with the important variations that exist in their myelopoietic capacities. Indeed, reports28,34 have shown that, as far as myelopoiesis is concerned, there is an at least 100-fold difference in proliferative capacity between FL and ABM stem cells and that ABM cells have much higher cytokine requirements than their FL counterparts. It is, of course, still possible that ABM stem cells need an additional stimulus, unknown thus far, to enter or to grow and develop in the thymic environment. It is also possible that facilitating cells, which display a positive feedback mechanism for the developing stem cells,35 are generated from FL and CB cells but not from ABM cells. Our data, however, suggest that when using the FTOC system, providing an additional stimulus (eg, TNF-α) besides survival-enhancing factors (eg, IL-3 + SCF) could be of benefit to augment the T-lymphopoietic potential of ABM CD34+ subpopulations.
Finally, although ABM CD34+ subpopulations are used in a clinical set-up to try and purge malignant cells or to diminish the role of graft-versus-host reactions,36 37 transplantation of purified CD34+ cells could have the disadvantage of slowing down thymus repopulation, resulting in delayed T-cell immune responsiveness. Therefore, finding a system that would allow an accelerated graft take and T-cell repopulation, using ex vivo generated (and expanded) T cells from purified CD34+ cells, could obviate these disadvantages. Understanding the process that governs T-lymphopoiesis is also of importance in potential applications such as gene transfer to correct genetic disorders of the T-cell lineage and restoration of the T-lymphocyte compartment after depletion caused by HIV infection.
In conclusion, we show for the first time T-cell differentiation starting from CD34++CD38− human hematopoietic stem cells by using an in vitro FTOC model. This provides additional proof that at least some of the multipotent stem cells are contained within this cell fraction. We also demonstrate, again for the first time in the human system, that TNF-α promotes T-cell differentiation from different subsets of CD34+ progenitors and that TNF-α induces up-regulation of CD127. Finally, we show that a significant difference exists in the T-cell progenitor activity of hematopoietic stem cells derived from ontogenetically different sources.
Supported by grants G.0096.95, 3.0109.96, and G.0157.99 of the Fund for Scientific Research, Flanders, Belgium; by the Geconcerteerde Onderzoeksactie of the Special Research Fund of the University of Antwerp (GOA 21/1996); and by private charities organized for the HEBA Foundation by Martine Julien, Gerald Dauphin, and the musical friends of the late Luke Walter, Jr., to whose memory this article is dedicated. S.F.A.W. is a research assistant of the Fund for Scientific Research, Flanders, Belgium.
Reprints:Zwi N. Berneman, Laboratory of Experimental Hematology, Antwerp University Hospital (UZA), Wilrijkstraat 10, B-2650 Edegem, Belgium.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.