Abstract
Abstract 677
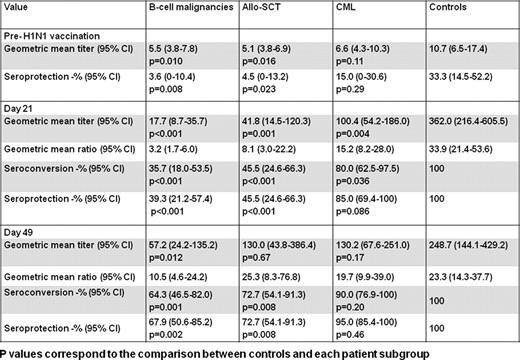
In 2009 the spread of influenza A (H1N1) satisfied the World Health Organization (WHO) criteria for a global pandemic and led to the initiation of a vaccination campaign to ensure protection for the most vulnerable patients. However, the immunogenicity of the 2009 H1N1 vaccine in immunocompromised patients has not been specifically evaluated. Furthermore, the number of doses of vaccine required for effective immunization against H1N1 has not been established. Whereas the European Medicines Agency (EMEA) and the UK Department of Health (DoH) recommended the injection of two doses of inactivated H1N1 vaccine 3 weeks apart in immunocompromised individuals, the Centers for Disease Control and Prevention recommended immunization with one dose of inactivated H1N1 vaccine for patients with cancer receiving chemotherapy, followed by a booster vaccine after completion of treatment if the pandemic continued. The aim of this study was to determine the safety and efficacy of the 2009 H1N1 vaccine in patients with hematologic malignancies. We prospectively evaluated the humoral and cellular immune responses to monovalent influenza A/California/2009(H1N1)v-like strain surface antigen vaccine in 97 adults with hematologic malignancies and 25 adult controls. Patients received two intramuscular injections of the vaccine 21 days apart and controls received one dose. Antibody titers, expressed as geometric mean, were measured using a hemagglutination-inhibition assay on days 0, 21 and 49 after injection of the first dose. The induction of virus-specific T-cell responses by H1N1 vaccination was assessed directly ex-vivo by flow cytometric enumeration of antigen-specific CD8+ and CD4+ T-lymphocytes using an intracellular cytokine assay for IFN-γ and TNF-α production on days 0 and 49. Of the 97 patients, 32 had chronic myeloid leukemia (CML) in chronic phase in complete cytogenetic response on the tyrosine kinase inhibitors imatinib or dasatinib, 39 had a B-cell malignancy in complete remission (CR) or untreated, and 26 were recipients of allogeneic hematopoietic stem cell transplantation (allo-SCT) in CR at least 6 months beyond transplant and without evidence of graft versus host disease. The vaccine was well tolerated, with no obvious difference in side effects for patients and controls. By day 21 post-vaccination, protective antibody titers of 1:32 or more were seen in 100% of controls compared to 39% of patients with B-cell malignancies (p<0.001), 46% of allo-SCT recipients (p<0.001) and 85% of CML patients (p=0.086). The effect of a booster dose was assessed with a paired sample analysis. After a second vaccine dose, the seroprotection rates increased to 68% (p=0.008), 73% (p=0.031), and 95% (p=0.5) in patients with B-cell malignancies, allo-SCT recipients and CML patients respectively. Patients vaccinated within 6 months of rituximab-based chemotherapy failed to mount a seroprotective antibody response. We also assessed the cellular response to H1N1 vaccine. Prior to vaccination, pre-existing T-cells against H1N1 could be detected in 10/23 controls compared to 2/25 allo-SCT recipients (p=0.007), 2/28 patients with B-cell malignancies (p=0.003) and 6/28 of CML patients (p=0.131). These pre-existing H1N1 T-cell responses may be related to previous exposure to 2009 H1N1 virus but more likely are due to the presence of cross-reactive seasonal and pandemic H1N1 specific T-cells. Following vaccination, H1N1-specific T-cells were induced in a significant proportion of allo-SCT recipient (10/25, p=0.008) and patients with B-cell malignancies (10/28; p=0.008), but not in CML patients or healthy controls. The limited ability of vaccines to significantly increase pre-existing influenza-specific T-cells has been previously reported although the mechanism for this phenomenon is not fully elucidated. These data demonstrate efficacy of H1N1 vaccine in the majority of patients with hematologic malignancies and unequivocally support the EMEA and the UK DoH official guidelines for the administration of 2 vaccine doses in immunocompromised patients to induce protective immune response against 2009 H1N1 influenza. Based on the WHO analyses, it is expected that the pandemic 2009 H1N1 virus will remain globally predominant in 2010–2011. These results may contribute towards the development of evidence-based guidelines for influenza vaccination in patients with hematologic malignancies.
Marin:Novartis: Consultancy, Honoraria, Research Funding; Bristol Myers Squibb: Consultancy, Honoraria, Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal