Key Points
Genetic deletion of JAK2 in vivo shows that MPN cells remain fully dependent on JAK2 signaling for survival.
Dual JAK2 targeting with JAK and HSP90 inhibitors offers increased efficacy in murine models and primary samples.
Abstract
The discovery of JAK2/MPL mutations in patients with myeloproliferative neoplasms (MPN) led to clinical development of Janus kinase (JAK) inhibitors for treatment of MPN. These inhibitors improve constitutional symptoms and splenomegaly but do not significantly reduce mutant allele burden in patients. We recently showed that chronic exposure to JAK inhibitors results in inhibitor persistence via JAK2 transactivation and persistent JAK–signal transducer and activator of transcription signaling. We performed genetic and pharmacologic studies to determine whether improved JAK2 inhibition would show increased efficacy in MPN models and primary samples. Jak2 deletion in vivo led to profound reduction in disease burden not seen with JAK inhibitors, and deletion of Jak2 following chronic ruxolitinib therapy markedly reduced mutant allele burden. This demonstrates that JAK2 remains an essential target in MPN cells that survive in the setting of chronic JAK inhibition. Combination therapy with the heat shock protein 90 (HSP90) inhibitor PU-H71 and ruxolitinib reduced total and phospho-JAK2 and achieved more potent inhibition of downstream signaling than ruxolitinib monotherapy. Combination treatment improved blood counts, spleen weights, and reduced bone marrow fibrosis compared with ruxolitinib alone. These data suggest alternate approaches that increase JAK2 targeting, including combination JAK/HSP90 inhibitor therapy, are warranted in the clinical setting.
Introduction
Myeloproliferative neoplasms (MPNs) are chronic myeloid malignancies characterized by the clonal expansion of myeloid lineage cells. The classical MPN include chronic myeloid leukemia (CML), polycythemia vera (PV), essential thrombocythemia (ET), and primary myelofibrosis (PMF). The majority of patients with PV, ET, and PMF harbor a highly conserved somatic mutation in the tyrosine kinase JAK2 (JAK2V617F).1-4 In addition, JAK2 exon 12 mutations are observed in JAK2V617F-negative PV5 and mutations in the thrombopoietin receptor (MPLW515L/K/A) are observed in JAK2V617F-negative ET/PMF.6 These mutations result in constitutive activation of the Janus kinase–signal transducer and activator of transcription (JAK-STAT) signaling pathway and their identification led to the clinical development of JAK kinase inhibitors for MPN patients.
In 2011, the dual JAK1/JAK2 inhibitor INCB18424 (ruxolitinib; Jakafi) was approved for the treatment of myelofibrosis (MF) based on the ability of ruxolitinib to markedly reduce splenomegaly and ameliorate MF-associated symptoms. Despite the important clinical benefits of ruxolitinib therapy, JAK inhibition with ruxolitinib and with other JAK inhibitors does not substantively reduce mutant allele burden as is observed with BCR-ABL kinase inhibition in CML.7 Similar results have been observed in murine models of MPN driven by activated Jak2/Mpl mutations, in which JAK inhibition leads to improved blood counts and splenomegaly but does not reduce mutant allele burden.8,9 The limited efficacy of JAK inhibitors in vivo might be due to incomplete pathway inhibition at clinically tolerable doses, the presence of other disease alleles, or incomplete dependence on JAK2 by the MPN clone.
We recently demonstrated that chronic exposure of MPN cells to ruxolitinib leads to the development of disease persistence and reduced sensitivity to JAK inhibition.10 We observed JAK inhibitor persistence (JAKPer) in MPN cell lines, mouse models, and primary samples from patients treated with ruxolitinib. Of note, MPN cells that are resistant to ruxolitinib were also insensitive to other JAK inhibitors including JAK inhibitor I and TG101348, and persistence was not associated with acquisition of secondary mutations in JAK2. In JAKPer cells, we found that JAK2 is stabilized by type I JAK inhibitors, which facilitates heterodimeric association of JAK2 with JAK1 and TYK2, JAK2 transactivation, and reactivation of JAK-STAT signaling. Although in vitro mutagenesis screens have identified a small set of JAK inhibitor resistance alleles in JAK2,11,12 to date, second-site mutations in JAK2 have not been identified in patients, consistent with incomplete inhibition of JAK-STAT signaling with existing JAK inhibitors.
These data have led investigators to question whether JAK2 represents an essential therapeutic target in MPN, and has led to diminished expectations of JAK-targeted therapy in MPN patients. We therefore sought to investigate whether JAK2 is a critical target in MPN in vivo using genetic studies, and sought to develop a therapeutic approach that improves JAK2 inhibition in vivo and increases therapeutic efficacy. Here we show that JAK2 is critically required for disease pathogenesis, both for initiation and maintenance of disease. Furthermore, we show that genetic deletion of Jak2 can overcome JAKPer in vivo. We have previously demonstrated that JAK2 is a heat shock protein 90 (HSP90) client protein and JAKPer cell lines remain sensitive to PU-H71, an HSP90 inhibitor.10,13 Based on these genetic and pharmacologic data, we investigated the efficacy of combined JAK2 and HSP90 inhibitors in preclinical MPN models such that we can inform the clinical development of improved treatment regiments for MPN patients.
Methods
Murine models and analysis of mice
Jak2f/f mice were a kind gift from Kay-Uwe Wagner (University of Nebraska Medical Center, Omaha, NE).14 They were backcrossed into C57BL/6 for 7 generations and then crossed to C57BL/6 Mx1-Cre mice. For Jak2 deletion studies, bone marrow (BM) cells from CD45.2 JAK2f/f Mx1-Cre positive and negative mice were enriched using CD117 microbeads from Miltenyi Biotec and transduced with viral supernatants containing MSCV-hMPLW515L-green fluorescent protein (GFP). One million transduced cells along with 500 000 CD45.1 CD117-positive BM cells were tail vein injected into CD45.1 recipients and bled 14 to 21 days after transplantation to assess engraftment and chimerism. For initiation experiments, mice received 4 intraperitoneal (IP) doses of 100 μL of polyI:polyC (pI:pC; 1 mg/mL) every other day starting at 14 days post–tail vein injection. For maintenance experiments, mice received pI:pC injections starting 19 days following tail vein injection. All mice were sacrificed 3 months after tail vein injection for histologic analysis and flow cytometry. For ruxolitinib experiments, mice were randomized to receive vehicle (20% Captisol in 58 mM citrate buffer, weight/volume), 60 mg/kg ruxolitinib twice daily by oral gavage or pI:pC by IP injection at day 18 after tail vein injection. All mice were euthanized 6 weeks later for further analysis.
For ruxolitinib and PU-H71 combination studies, BM cells were isolated from fluorouracil (5-FU)–treated Balb/C donor mice, transduced with hMPLW515L-IRES-GFP retrovirus and injected into lethally irradiated Balb/C recipients. Fourteen days following injection, mice were randomized to receive vehicle, 30 mg/kg ruxolitinib twice daily by oral gavage, 90 mg/kg ruxolitinib twice daily by oral gavage, and 30 mg/kg ruxolitinib with 75 mg/kg PU-H71 thrice weekly by IP injection. All mice were bled at day 14 following start of treatment. Two mice from each arm were euthanized for further analysis. At the 2-week time point, a subset of mice receiving 30 mg/kg ruxolitinib alone also started receiving 75 mg/kg PU-H71. Also, the ruxolitinib dose was increased to 90 mg/kg in a subset of mice receiving combination treatment. At 4 weeks from the start of drug treatment, all mice were euthanized for further analysis. Ruxolitinib was synthesized by the Bradner laboratory at the Dana-Farber Cancer Institute and PU-H71 was synthesized by the Chiosis laboratory at Memorial Sloan-Kettering Cancer Center as previously reported.15,16
Animal care was in strict compliance with institutional guidelines established by the Memorial Sloan-Kettering Cancer Center, the Guide for the Care and Use of Laboratory Animals, and the Association for Assessment and Accreditation of Laboratory Animal Care International. For histopathology, tissues were fixed in 4% paraformaldehyde and then embedded in paraffin for analysis. Tissue samples were stained using hematoxylin and eosin as well as a Gordon and Sweet stain for reticulin fibers (ammoniacal silver procedure).
Flow cytometry analysis
BM and spleen cells were filtered, and red blood cells were lysed and washed in phosphate-buffered saline (PBS). Cells were incubated with the following antibodies for 30 minutes on ice in PBS plus 2% bovine serum albumin (BSA). For staining of myeloid progenitors, the antibodies used were CD11b, Gr-1, Ter119, CD3, CD4, NK1.1, B220, CD19 conjugated to allophycocyanin Cy7 (APCCy7) (Biolegend), c-kit–phycoerythrin (PE) (Biolegend), Sca1-PECy7 (BD Pharmingen), CD16/32-eFluor 450 (eBioscience), CD34–Alexa Fluor 660 (eBioscience). For chimerism and mature leukocyte staining, the antibodies used were CD45.1–eFluor 450 (eBioscience), CD45.2-APC, CD11b-PECy7, Gr1-PE (Biolegend). Data were collected on LSRFortessa (BD Biosciences) and analysis was performed on FlowJo.
Immunoblotting for patient samples
The institutional review boards of Memorial Sloan-Kettering Cancer Center approved sample collection and all experiments. Informed consent was obtained from human subjects before study. The study was conducted in accordance with the Declaration of Helsinki. Mononuclear cells were freshly extracted using Ficoll separation from peripheral blood (PB) and used for studies. Cells were treated with 500 nM PU-H71 for 16 hours, washed in ice-cold PBS, and collected in lysis buffer (150 mM NaCl, 20 mM Tris [pH 7.4-7.5], 5 mM ethylenediaminetetraacetic acid, 1% Triton-X, 10% glycerol) containing Protease Arrest (G-Biosciences), Phosphatase Inhibitor Cocktail II (EMD Chemicals). Protein was quantified using the Bio-Rad Bradford protein estimation and 30 to 50 μg was loaded per well in 4% to 12% Bis-Tris electrophoresis gels (Invitrogen). Protein was transferred on to 0.45-micron nitrocellulose membranes. Antibodies used for western blotting included pSTAT5 and phosphorylated and total JAK2, pSTAT3, and pMAPK (all from Cell Signaling Technologies) and Actin (EMD Chemicals).
Results
JAK2 is required for initiation of MPLW515L-mediated disease
Retroviral expression of mutant MPLW515L in hematopoietic cells in vivo results in the development of a highly penetrant, lethal MPN with features of ET/PMF, including leukocytosis, thrombocytosis, extramedullary hematopoiesis, and extensive BM fibrosis.6 Given that this model depends on expression of a mutant cytokine receptor, we evaluated the effect of Jak2 deletion on disease development in vivo. Germline deletion of Jak2 results in embryonic lethality.17,18 We therefore used a conditional knockout model of Jak214 in which Jak2 is deleted in an inducible and hematopoietic-specific manner by Cre-recombinase expressed under the control of the Mx1 promoter. BM cells from Jak2 floxed (Jak2f/f) Mx1-Cre+ and Mx1-Cre− mice expressing the CD45.2 congenic marker were transduced with the GFP-tagged MPLW515L retrovirus and transplanted into irradiated CD45.1 recipients along with an equal number of CD45.1 support BM cells. Two weeks following transplantation, we documented engraftment by the presence of GFP-positive cells in PB of transplanted mice. Before mice developed overt disease, Jak2 was deleted by injection of pI:pC and we followed the proportion of GFP-positive cells and CD45.1:CD45.2 ratios in hematopoietic cells (supplemental Figure 1, available on the Blood Web site).
These data revealed marked differences in disease onset and progression in mice transplanted with Jak2f/fMx1-Cre+ and Mx1-Cre− cells. All mice receiving Jak2f/fMx1-Cre− cells developed severe, lethal MPN characterized by leukocytosis, thrombocytosis, splenomegaly, and MF. By contrast, we did not observe significant disease in mice transplanted with Jak2f/fMx1-Cre+ cells; white blood cell (WBC) (52.52 K/μL vs 10.27 K/μL,) and platelet counts (Plt) (1328 K/μL vs 988 K/μL) remained normal consistent with an absence of myeloproliferation (Figure 1A-B) More importantly, MPL mutant allele burden as measured by percentage of GFP-positive cells was significantly reduced compared with controls (4.9% vs 44.3%, P < .005) (Figure 1C). Spleen and liver sizes were also significantly reduced in mice transplanted with Jak2f/fMx1-Cre+ cells compared with mice transplanted with Jak2f/fMx1-Cre− cells (spleen 0.176 g vs 0.577 g, P < .05; liver 1.48 g vs 1.76 g, P < .05) (Figure 1D-E). Notably, BM fibrosis, a hallmark feature of this PMF model, was completely absent in mice transplanted with Jak2f/fMx1-Cre+ compared with marked reticulin fibrosis in mice engrafted with Jak2f/fMx1-Cre− cells (Figure 1F).
JAK2 is required for initiation of MPLW515L-mediated disease. Deletion of Jak2 following engraftment of MPLW515L-transduced BM leads to significant reduction in (A) WBC counts, (B) platelets, (C) mutant allele burden in terms of GFP-positive cells in PB (P < .005), and (D) spleen (P < .05) and (E) liver size (P < .05) as compared with controls. (F) BM fibrosis assessed by reticulin staining is absent in Jak2-deleted BM. (Data are represented at mean ± standard error of the mean [SEM]; Jak2+/+, n = 4; Jak2−/−, n = 5).
JAK2 is required for initiation of MPLW515L-mediated disease. Deletion of Jak2 following engraftment of MPLW515L-transduced BM leads to significant reduction in (A) WBC counts, (B) platelets, (C) mutant allele burden in terms of GFP-positive cells in PB (P < .005), and (D) spleen (P < .05) and (E) liver size (P < .05) as compared with controls. (F) BM fibrosis assessed by reticulin staining is absent in Jak2-deleted BM. (Data are represented at mean ± standard error of the mean [SEM]; Jak2+/+, n = 4; Jak2−/−, n = 5).
With extended observation, 1 mouse in the Mx1-Cre+ cohort manifested a persistent GFP-positive population in the PB, and subsequently developed elevated blood counts and an enlarged spleen (supplemental Figure 2A) consistent with disease progression. Polymerase chain reaction (PCR) analysis documented incomplete excision and expansion of unexcised GFP-positive cells over time, as can be seen by presence of the floxed allele in PB (supplemental Figure 2B). These data strongly indicate an absolute requirement for JAK2 in disease initiation in the MPLW515L model of MF.
JAK2 plays a critical role in survival of the MPN mutant clone
We then asked whether JAK2 was required in the maintenance of the disease clone in the MPLW515L model. Jak2 was excised by administration of pI:pC 4 to 6 weeks after BM transplantation, at which time all mice had evidence of myeloid expansion and overt disease. Similar to the results with Jak2 deletion at disease initiation, loss of JAK2 when mice had overt MF resulted in a rapid, potent reduction in leukocytosis and Plt compared with mice transplanted with Jak2f/fMx1-Cre− cells (WBC 18.71 K/μL vs 95.78 K/μL, P < .005; Plt 456 K/μL vs 1171 K/μL, P < .005) (Figure 2A-B). Jak2 deletion caused marked reduction in spleen sizes compared with mice transplanted with Jak2f/fMx1-Cre− cells (0.16 g vs 0.63 g, P < .005) (Figure 2C and supplemental Figure 3A). Most importantly, we observed a rapid, significant reduction in mutant allele burden (GFP-positive cells) in the PB and BM (PB 34.04% vs 77.12%, P < .005; BM 20.92% vs 84.56%, P < .005) (supplemental Figure 3B and Figure 2D). Of note, the marked reduction in mutant allele burden seen with Jak2 deletion is not observed with maximal kinase inhibition in this same model.9,10
JAK2 plays a critical role in the survival of MPN clone. Excision of Jak2 following disease establishment led to reduction in (A) WBC, (B) platelets, (C) spleen sizes, and (D) GFP-positive cells in BM (P < .005). Loss of JAK2 results in significant improvement in disease features including (E) restoration of splenic architecture, (F) decrease in myeloid infiltration in liver, and (G) reduction in BM fibrosis. (H) There was also a reduction in CD11b+Gr1+ cells and MEP population in MPLW515L-transduced mice lacking Jak2. (Data are represented at mean ± SEM; Jak2+/+, n = 5; Jak2−/−, n = 6).
JAK2 plays a critical role in the survival of MPN clone. Excision of Jak2 following disease establishment led to reduction in (A) WBC, (B) platelets, (C) spleen sizes, and (D) GFP-positive cells in BM (P < .005). Loss of JAK2 results in significant improvement in disease features including (E) restoration of splenic architecture, (F) decrease in myeloid infiltration in liver, and (G) reduction in BM fibrosis. (H) There was also a reduction in CD11b+Gr1+ cells and MEP population in MPLW515L-transduced mice lacking Jak2. (Data are represented at mean ± SEM; Jak2+/+, n = 5; Jak2−/−, n = 6).
Deletion of Jak2 led to reduced pathologic myeloproliferation, including a decrease in extramedullary hematopoiesis, restoration of normal splenic architecture, and abrogation of BM fibrosis (Figure 2E-G and supplemental Figure 4). These data indicate that elimination of JAK2 in mutant MPN cells can reverse BM fibrosis, contrary to what has been observed with JAK inhibition in the preclinical and clinical setting. Jak2 deletion normalized the expansion of the CD11b+Gr1+ myeloid lineages and megakaryocytic-erythroid progenitor (MEP) compartment observed in this MF model (Figure 2H and supplemental Figure 5). As in the studies done at disease initiation, we documented that the remaining GFP-positive mutant cells in the BM had incomplete excision of Jak2, based on genotypic analysis of sorted GFP-positive BM cells (supplemental Figure 6). These data demonstrate that JAK2 is required for maintenance of the mutant MPN clone.
Deletion of Jak2 is more effective than JAK inhibitor treatment alone
We have previously shown that although MPN cells can rapidly become insensitive to JAK inhibition, knockdown of JAK2 by RNA interference in JAKPer cells leads to growth suppression and inhibition of downstream signaling.10 By deleting Jak2 in vivo, we tested whether we could rescue the persistent phenotype in the MPLW515L model of ET/MF. BM from Jak2f/fMx1-Cre+ mice was retrovirally transduced with MPLW515L-IRES-GFP and transplanted into lethally irradiated recipients. After disease establishment, mice were randomized to receive vehicle, ruxolitinib, or pI:pC to delete Jak2. As reported previously,9 although drug treatment improved PB counts, ruxolitinib did not reduce the mutant allele burden in vivo. By contrast, deletion of Jak2 markedly reduced percentage of GFP-positive cells in the BM (5.03% vs 65.27%, P < .005) (Figure 3A). Deletion of Jak2 reduced blood counts (WBC 8.32 K/μL vs 17.06 K/μL, P < .005) (Figure 3B) and spleen size (0.19 g vs 0.40 g, P < .005) (Figure 3C) to a greater extent than observed with chronic JAK inhibitor treatment. Analysis of myeloid and progenitor populations revealed loss of JAK2 led to a significant reduction in MEP and CD11b+Gr+ proportions with a dramatic decrease in the contribution of mutant cells to myeloid progenitors and differentiated myeloid compartments (Figure 3D and supplemental Figure 7), which was not observed with ruxolitinib therapy. These results indicate that deletion of Jak2 is superior to JAK2 inhibitor therapy at reducing disease burden consistent with a dependence on JAK2, which is not amenable to inhibition with clinically available JAK inhibitors.
Deletion of Jak2 is more effective than JAK inhibitor therapy at reducing disease burden.MPLW515L-transduced mice were treated with vehicle, 60 mg/kg ruxolitinib or pI:pC to excise Jak2. (A) Deletion of Jak2 led to significant reduction in mutant allele burden in BM as compared with inhibitor treatment (P < .005). It also resulted in further decrease in (B) blood counts and (C) spleen size in Jak2-deleted mice compared with mice that received ruxolitinib alone (P < .005). (D) Loss of JAK2 also leads to reduction in MEP and CD11b+Gr+ proportions with a dramatic decrease in the contribution of mutant (GFP+) cells. (Data are represented at mean ± SEM; vehicle, n = 5; Rux, n = 9; Jak2 deleted, n = 6).
Deletion of Jak2 is more effective than JAK inhibitor therapy at reducing disease burden.MPLW515L-transduced mice were treated with vehicle, 60 mg/kg ruxolitinib or pI:pC to excise Jak2. (A) Deletion of Jak2 led to significant reduction in mutant allele burden in BM as compared with inhibitor treatment (P < .005). It also resulted in further decrease in (B) blood counts and (C) spleen size in Jak2-deleted mice compared with mice that received ruxolitinib alone (P < .005). (D) Loss of JAK2 also leads to reduction in MEP and CD11b+Gr+ proportions with a dramatic decrease in the contribution of mutant (GFP+) cells. (Data are represented at mean ± SEM; vehicle, n = 5; Rux, n = 9; Jak2 deleted, n = 6).
Combined JAK and HSP90 inhibition is more efficacious than JAK inhibitor monotherapy
The results so far suggest that inhibiting the kinase activity of JAK2 with existing, ATP competitive inhibitors might not sufficiently inhibit JAK2 to obtain adequate efficacy. However, since MPN cells remain dependent on JAK2 expression for their growth and survival, we hypothesized that combination approaches which increase JAK2 inhibition would lead to increased efficacy in vivo. We previously reported that JAK2 is an HSP90 client protein and that treatment of MPN cells with HSP90 inhibitors results in degradation of total and phosphorylated JAK2 and concomitant inhibition of downstream signaling.13 Of note, JAKPer cells remain sensitive to PU-H71, an HSP90 inhibitor that is efficacious in preclinical MPN models.10 We therefore decided to test the efficacy of combined ruxolitinib and PU-H71 in the MPLW515L model of ET/MF. The dosing regimens tested included vehicle, 2 different doses of ruxolitinib monotherapy, combined JAK/HSP90 inhibition from the onset and sequential therapy with ruxolitinib followed by combined ruxolitinib/PU-H71 therapy (supplemental Figure 8). We did not observe any hematopoietic or nonhematopoietic toxicity with combined JAK/HSP90 inhibition, and escalated both agents to their maximally tolerated monotherapy doses in combination studies.
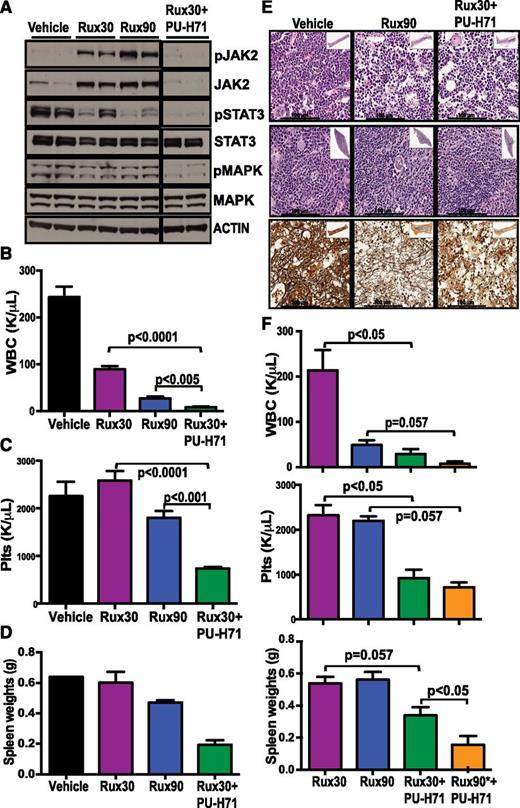
Combination therapy resulted in a potent reduction in total and phosphorylated JAK2 expression, whereas JAK inhibitor therapy alone resulted in increased phospho-JAK2 and JAK2 expression as previously reported.10,19 More importantly, we observed more potent inhibition of downstream signaling effectors including phospho- and phospho-MAPK (Figure 4A). Immunohistochemistry studies documented that combination therapy resulted in more potent inhibition of downstream signaling, concomitant with induction of HSP70 expression (supplemental Figure 9A-B). These data indicated that combination therapy results in greater inhibition of the JAK-STAT pathway than maximally tolerated JAK kinase inhibitor monotherapy.
Combination of JAK2 and HSP90 inhibition is more efficacious than JAK inhibitor monotherapy. (A) Combination of ruxolitinib and PU-H71 resulted in JAK2 degradation and inhibition of downstream signaling in splenocytes of MPLW515L-transduced mice (all lanes were run on the same gel). After 2 weeks of drug treatment, the ruxolitinib and PU-H71 combination arm had significantly lower (B) WBC and (C) Plt compared with ruxolitinib alone (P < .0001, n = 10). (D) Spleen size in the combination arm was also lower. (E) Improved histopathology and reduction in BM fibrosis in mice treated with combination of JAK and HSP90 inhibitors. (F) After 4 weeks of drug treatment, the combination group that had ruxolitinib dose increased to 90 mg/kg for 2 weeks (Rux90*+PU-H71) had lowest blood counts and spleen sizes compared with ruxolitinib alone. (Data are represented at mean ± SEM; n = 4).
Combination of JAK2 and HSP90 inhibition is more efficacious than JAK inhibitor monotherapy. (A) Combination of ruxolitinib and PU-H71 resulted in JAK2 degradation and inhibition of downstream signaling in splenocytes of MPLW515L-transduced mice (all lanes were run on the same gel). After 2 weeks of drug treatment, the ruxolitinib and PU-H71 combination arm had significantly lower (B) WBC and (C) Plt compared with ruxolitinib alone (P < .0001, n = 10). (D) Spleen size in the combination arm was also lower. (E) Improved histopathology and reduction in BM fibrosis in mice treated with combination of JAK and HSP90 inhibitors. (F) After 4 weeks of drug treatment, the combination group that had ruxolitinib dose increased to 90 mg/kg for 2 weeks (Rux90*+PU-H71) had lowest blood counts and spleen sizes compared with ruxolitinib alone. (Data are represented at mean ± SEM; n = 4).
We assessed inhibitor efficacy 2 weeks following the start of treatment; at that time point, combination therapy significantly reduced WBC and Plt compared with low-dose (30 mg/kg) or high-dose (90 mg/kg) ruxolitinib monotherapy (P < .0001) (Figure 4B-C). Combination treatment also led to a further reduction in spleen size compared with ruxolitinib monotherapy (Figure 4D) and increased histopathological improvement, with reduced BM and spleen cellularity, a reduction in megakaryocyte expansion, and a reduction in BM fibrosis (Figure 4E and supplemental Figure 9B). Of note, we observed a greater difference in efficacy with 4 weeks of combination therapy, as combined ruxolitinib/PU-H71 markedly reduced blood counts and organomegaly compared with ruxolitinib alone. Although we first used a suboptimal dose of ruxolitinib therapy in our combination studies, we found that combining the highest dose of ruxolitinib (90 mg/kg) with PU-H71 was the most efficacious strategy without any increased toxicity (Figure 4F).
Given that the data above were obtained in a MPLW515L-driven model of MPN, we investigated whether combined JAK/HSP90 inhibition would demonstrate increased efficacy in a Jak2V617F-driven model of MPN. We transplanted BM cells from Jak2V617F knock-in mice8 into secondary recipients, and following disease engraftment, treated mice with ruxolitinib or with combined ruxolitinib/PU-H71. Combination therapy resulted in a further reduction in blood counts and spleen size in vivo compared with ruxolitinib monotherapy, consistent with increased therapeutic efficacy (supplemental Figure 10).
Genetic or pharmacologic loss of JAK2 can overcome inhibitor persistence in vivo and in patient samples
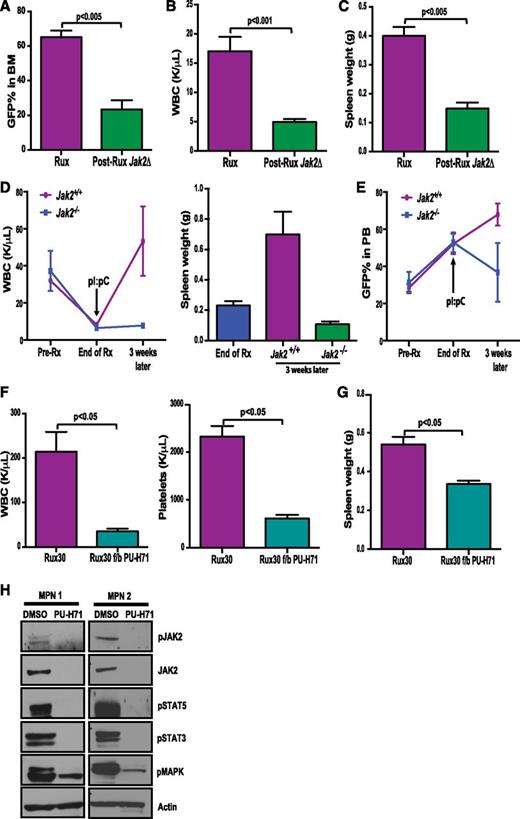
The approval of ruxolitinib for MF has led to its use as first-line treatment of MF patients with significant splenomegaly and symptoms. As such, we investigated whether improved JAK2 targeting following initial ruxolitinib monotherapy might lead to significant therapeutic benefit. We first assessed whether genetic deletion of Jak2 after ruxolitinib treatment could reduce myeloproliferation and disease burden in vivo. BM from Jak2f/fMx1-Cre+ mice was retrovirally transduced with MPLW515L-IRES-GFP and transplanted into lethally irradiated recipients. Following disease establishment, we initiated treatment with ruxolitinib (supplemental Figure 11). Although ruxolitinib treatment improved blood counts and reduced spleen size, we did not observe a reduction in mutant allele burden with 5 weeks of ruxolitinib therapy (supplemental Figure 12). By contrast, deletion of Jak2 following ruxolitinib treatment led to a significant reduction in mutant allele burden in BM (23.28% vs 65.27%, P < .005) (Figure 5A), blood counts (WBC 4.99 K/μL vs 17.06 K/μL, P < .001) (Figure 5B), and spleen sizes (0.149 g vs 0.40 g, P < .005) (Figure 5C) as compared with mice that continued to receive drug. Furthermore, deletion of Jak2 after long-term ruxolitinib treatment (5 weeks) prevented disease relapse, which is observed with cessation of treatment even after longer-term ruxolitinib therapy (Figure 5D). The percentage of GFP-positive cells, which persisted at a high-level despite 5 weeks of ruxolitinib treatment, was also decreased by deletion of Jak2 (Figure 5E). These results indicate that deletion of Jak2 in vivo can successfully eliminate mutant MPN cells that survive in the setting of chronic JAK2 kinase inhibition.
Genetic loss or pharmacologic degradation of JAK2 subsequent to JAK inhibitor monotherapy can overcome persistence. Deletion of Jak2 following 2 weeks of ruxolitinib treatment resulted in reduction of (A) mutant allele burden (P < .005), (B) bloods counts (P < .001), and (C) spleen weights (P < .005) compared with mice that continued to receive ruxolitinib. (Rux, n = 9; Jak2 deleted, n = 6). Deletion of JAK2 after long-term (5 weeks) of ruxolitinib treatment prevents disease relapse following cessation of treatment by reducing (D) WBC and spleen weights as well as (E) mutant allele burden in Jak2-deleted mice (n = 3). (F) Addition of PU-H71 to ruxolitinib monotherapy resulted in significant reduction in blood counts. (G) Spleen sizes were further reduced by PU-H71 therapy (P < .05, n = 4). (Data are represented at mean ± SEM). (H) Mononuclear cells from ruxolitinib-treated MPN patients were isolated and treated with 0.5 μM PU-H71 for 16 hours, which led to inhibition of JAK2 and downstream STAT-MAPK signaling.
Genetic loss or pharmacologic degradation of JAK2 subsequent to JAK inhibitor monotherapy can overcome persistence. Deletion of Jak2 following 2 weeks of ruxolitinib treatment resulted in reduction of (A) mutant allele burden (P < .005), (B) bloods counts (P < .001), and (C) spleen weights (P < .005) compared with mice that continued to receive ruxolitinib. (Rux, n = 9; Jak2 deleted, n = 6). Deletion of JAK2 after long-term (5 weeks) of ruxolitinib treatment prevents disease relapse following cessation of treatment by reducing (D) WBC and spleen weights as well as (E) mutant allele burden in Jak2-deleted mice (n = 3). (F) Addition of PU-H71 to ruxolitinib monotherapy resulted in significant reduction in blood counts. (G) Spleen sizes were further reduced by PU-H71 therapy (P < .05, n = 4). (Data are represented at mean ± SEM). (H) Mononuclear cells from ruxolitinib-treated MPN patients were isolated and treated with 0.5 μM PU-H71 for 16 hours, which led to inhibition of JAK2 and downstream STAT-MAPK signaling.
We then asked whether we could leverage this finding for therapeutic purposes. We initiated combination JAK2/HSP90 therapy following ruxolitinib monotherapy. After 2 weeks of ruxolitinib treatment, we added 75 mg/kg PU-H71 treatment to a subset of mice that were receiving monotherapy (supplemental Figure 8). Two weeks of combination treatment resulted in a significant reduction in leukocytosis and thrombocytosis compared with ruxolitinib monotherapy (WBC 35.22 K/μL vs 214 K/μL; P < .05; Plt 613 K/μL vs 2323 K/μL, P < .05) (Figure 5F). Addition of PU-H71 to ruxolitinib monotherapy also reduced splenomegaly to a greater extent than observed with ruxolitinib monotherapy (0.33 g vs 0.53 g, P < .05) (Figure 5G).
We have previously demonstrated that hematopoietic cells from MPN patients treated with ruxolitinib are insensitive to JAK2 kinase inhibition ex vivo and exhibit persistent downstream signaling with ex vivo JAK inhibitor exposure.10 We therefore asked whether treatment with an HSP90 inhibitor increased JAK-STAT pathway inhibition in samples from MF patients receiving ruxolitinib therapy. As postulated, PU-H71 treatment led to degradation of total and activated JAK2 and abrogation of downstream STAT and MAPK signaling (Figure 5H). Thus, PU-H71 is effective in inhibiting JAK-STAT signaling in primary samples that are insensitive to JAK inhibition.
Discussion
The discovery of mutations in the JAK-STAT pathway in a majority of MPN patients led to the hypothesis that this pathway activation is a central feature of MPN pathogenesis. Expression of mutant JAK2/MPL alleles transforms hematopoietic cells to cytokine-independent growth, and in vivo expression of these alleles recapitulates many features of MPNs. These experiments are consistent with the notion that JAK-STAT pathway mutations are indeed drivers of MPN pathogenesis. However, kinase inhibition of JAK2 has not resulted in the molecular responses that have been observed with ABL kinase inhibitors in CML. Although MPN patients experience an improvement in splenomegaly, and constitutional symptoms, treatment with JAK inhibitors is not curative and does not significantly decrease mutant allele burden. These studies therefore led investigators to question the role of activated JAK2 signaling in MPN pathogenesis and the ultimate role of JAK-targeted therapies as a long-term therapeutic strategy for MPN patients.
The identification of secondary resistance mutations in response to chronic inhibition is often considered a hallmark of effective targeted therapy consistent with sufficient target inhibition to select for genetic resistance. This has been observed with ABL kinase inhibitors in CML,20 EGFR inhibitors in lung cancer,21 and FLT3 inhibitors in acute myeloid leukemia.22 However, this has not been the case with JAK2 inhibitors to date, suggesting either incomplete dependence on JAK2 or insufficient inhibition of the target in the clinical setting. Our previous in vitro studies suggested a putative mechanism for insufficient JAK-STAT pathway inhibition based on activation of JAK2 by other JAK kinases. The underlying mechanism is based on the stabilization of activated JAK2 by the binding of type I inhibitors, including ruxolitinib.19 These data suggest that MPN cell lines require JAK2, at least in part, as a scaffold to maintain downstream signaling even in the context of inhibition of its catalytic activity. This can at least partially be overcome by type II JAK kinase inhibitors; however, type II JAK inhibitors have not been developed for in vivo studies to date. As such, there is a need to determine whether JAK2 remains a critical therapeutic target in MPN cells, and whether there are pharmacologic approaches that can be used to improve JAK2 targeting and therapeutic response.
In this study, we demonstrate that Jak2 is essential in vivo by deleting Jak2 in an inducible, tissue-specific manner at different stages of disease and following JAK inhibitor therapy. Jak2 deletion led to significant improvements in blood counts, organomegaly, and reduced mutant allele burden, which has not been shown using JAK inhibitors as monotherapy. We also showed that genetic loss of Jak2 was more efficacious than kinase inhibition alone in vivo. Furthermore, deletion of Jak2 was curative in this model, as disease did not recur with the exception of mice with incomplete deletion of Jak2, consistent with a genetic requirement for JAK2 signaling in MPN. Taken together, these data suggest that JAK2 remains an exceptional therapeutic target in MPN and that we have not seen the impact of maximal JAK kinase inhibition in MPN patients with existing agents to date.
We previously demonstrated that JAK2 is an HSP90 client protein and treatment with HSP90 inhibitors leads to degradation of total and phosphorylated JAK2 along with inhibition of downstream signaling.13 JAK inhibitors and HSP90 inhibitors likely have pleiotropic effects. It is possible that the increased therapeutic efficacy seen with combination therapy is due to HSP90-mediated effects on different client proteins, and JAK inhibitors may in part exert their effects through amelioration of inflammatory signaling. In these studies, we show that combination ruxolitinib/PU-H71 therapy was more efficacious than JAK inhibitor monotherapy at inhibiting JAK-STAT signaling and reducing myeloproliferation without attendant increases in toxicity. Preliminary observations from monotherapy trials with the HSP90 inhibitors AUY922 and PU-H71 for refractory MPN patients suggest significant clinical efficacy of HSP90 inhibition in MPN patients (R.R. and R.L.L., unpublished data). Combination studies in which HSP90 inhibition is added to JAK kinase inhibitor therapy are being planned subsequent to these trials.
Taken together, these data indicate that MPN cells are dependent on JAK2 expression for their growth and survival, and therapies that lead to increased inhibition of JAK-STAT signaling should be arduously investigated in preclinical and clinical settings. These may include combination JAK/HSP90 inhibitor therapy, type II JAK kinase inhibitors, and other combination approaches with additive inhibitory effects on the JAK-STAT pathway. Given the limited ability of JAK kinase inhibition to achieve disease-modifying activity in MPN patients, we predict that novel approaches which lead to increased JAK2 inhibition in the clinical setting will improve outcomes for patients with JAK-dependent malignancies.
The online version of this article contains a data supplement.
There is an Inside Blood commentary on this article in this issue.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Acknowledgments
We thank members of the Levine laboratory, Neal Rosen, and Ari Melnick for helpful comments and discussion. We thank Thomas Radimerski, Masato Murakami, and Vincent Romanet (Novartis) for help with immunohistochemical analysis. We thank Benjamin L. Ebert and Ann Mullally (both from Harvard Medical School) for providing Jak2V617F knock-in mice.
This work was supported by a Leukemia & Lymphoma Society Translational Research Program grant (R.L.L. and G.C.) and by National Cancer Institute at the National Institutes of Health grants 1R01CA151949-01 (R.L.L.). R.L.L. and R.R. are also supported by grants from the Geoffrey Beene Cancer Research Center.
M. Kleppe is a fellow of the Leukemia & Lymphoma Society and was previously supported by an EMBO Postdoctoral Fellowship. R.L.L. is a Leukemia & Lymphoma Society Scholar.
Authorship
Contribution: N.B., P.K., and R.L.L. conceived the project; N.B., P.K., S.M., and R.L.L. designed experiments; N.B., P.K., S.M., M. Keller, and K.S. performed experiments; N.B., P.K., S.M., and R.L.L. analyzed data; M. Kleppe and R.R. provided reagents and samples; H.J.P., S.K.S., T.T., and G.C. provided PU-H71 and assisted with design and performance of in vivo studies; J.Q. and J.E.B. provided ruxolitinib; and N.B., P.K., and R.L.L. wrote the manuscript with input from all other authors.
Conflict-of-interest disclosure: R.L.L. is a consultant and receives research support from Novartis Pharmaceuticals. Memorial Sloan-Kettering Cancer Center holds the intellectual rights to PU-H71. Samus Therapeutics, of which G.C. has partial ownership, has licensed PU-H71. The remaining authors declare no competing financial interests.
Correspondence: Ross L. Levine, Human Oncology and Pathogenesis Program, Memorial Sloan-Kettering Cancer Center, 1275 York Ave, Box 20, New York, NY 10065; e-mail: leviner@mskcc.org.
References
Author notes
N.B. and P.K. contributed equally to this work.

![Figure 1. JAK2 is required for initiation of MPLW515L-mediated disease. Deletion of Jak2 following engraftment of MPLW515L-transduced BM leads to significant reduction in (A) WBC counts, (B) platelets, (C) mutant allele burden in terms of GFP-positive cells in PB (P < .005), and (D) spleen (P < .05) and (E) liver size (P < .05) as compared with controls. (F) BM fibrosis assessed by reticulin staining is absent in Jak2-deleted BM. (Data are represented at mean ± standard error of the mean [SEM]; Jak2+/+, n = 4; Jak2−/−, n = 5).](https://ash.silverchair-cdn.com/ash/content_public/journal/blood/123/13/10.1182_blood-2014-01-547760/4/m_2075f1.jpeg?Expires=1767738147&Signature=L0xtyptFQXIm-yjmSlQzK2cPXIhtSO~7Zc-8b9dsKb26ZkWnXPVVufnIYQR3eyJh80jr6R6k4Yu1QOFI2afE5qdXZdO3AiOENwvOcv~Qsx7fiPJINqSMgRNKgVEMG5pF2fBPXmlPVlAbHwD5FL0FlHqmN9Cf7SyL3xyiHyrrtlzLBctY7M3q7EVpMLLAk1iHm4D76f5ss~Sdn67wB6huSwZIHkCeV~R0dMKwwi0WHp7GZ3X3d-DoFvn9GzaRf9Bb9FX1YjEed89svb4STTfdV7moO8BzTGuYlgMpO5Fq2S53lWU8HjW6RJ6N88Z3hrLRKc~BfMFejJJEhs0JoGLeQw__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)