Abstract

BACKGROUND: Multiple myeloma (MM) is the most common indication for high-dose chemotherapy and autologous stem cell transplantation (ASCT). Post-transplant lenalidomide maintenance therapy doubles progression-free survival, but almost all patients eventually relapse. The immune system participates in the control of MM, whereas compromised immunity contributes to its evolution. Post-transplant immunotherapy to induce or restore antitumor immunity offers a promising approach to target residual MM and improve patient outcomes. The rational development of immunotherapeutic interventions after ASCT, however, requires a comprehensive understanding of the immunologic milieu. We therefore evaluated lymphocyte composition and function after ASCT to guide optimal timing of immunotherapy and to identify potential markers of relapse.
METHODS: Fifty-five MM patients undergoing ASCT were evaluated for at least one year. Peripheral blood from patients was obtained before ASCT and on d +12, +30, +90, +180, and +365 after ASCT, and at the time of relapse where applicable. Leukocyte concentrates were used as a source of healthy donor cells. Mononuclear cells were analyzed by flow cytometry for phenotypic assessment of lymphocyte subset composition. Functional assessment of dendritic cell and T cell activity in vitro was assayed in autologous and allogeneic mixed leukocyte reactions, cytotoxic T lymphocyte (CTL) lysis assays, and PD-1 blockade experiments.
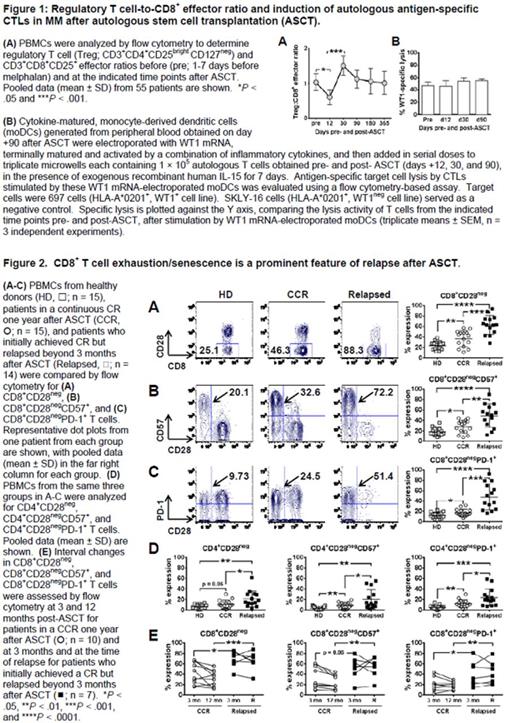
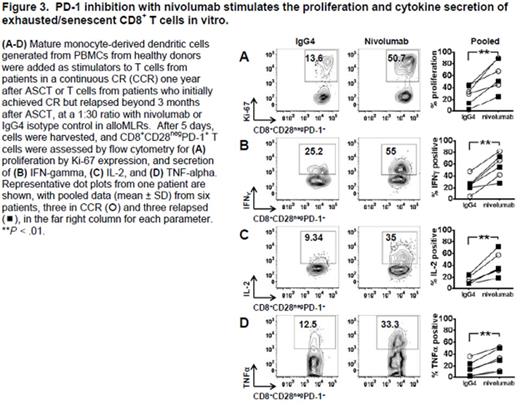
RESULTS: CD3+ CD4+ CD25bright CD127neg regulatory T cells (Tregs) decline as CD8+ T cells expand during early lymphocyte recovery after ASCT, markedly reducing the Treg:CD8+ effector T-cell ratio (Fig 1A) and providing a critical early window for the introduction of immune-based post-transplant consolidation therapies. CD8+ T cells can respond to autologous dendritic cells presenting tumor antigen in vitro as early as day +12 post-transplant, becoming antigen-specific CTL effectors and thereby demonstrating preservation of cellular reactivity (Fig 1B). CD4+ and CD8+ T cells express the negative regulatory molecules, CTLA-4, PD-1, LAG-3, and TIM-3, before and after ASCT (data not shown). A subpopulation of exhausted/senescent CD8+ T cells, however, down-regulates CD28 (Fig 2A) and up-regulates CD57 (Fig 2B) and PD-1 (Fig 2C), characterizing immune impairment and relapse after ASCT. CD4+ T cells show the same trends in the frequencies of CD28neg, CD28neg CD57+, and CD28neg PD-1+ cells, albeit at lower levels of expression (Fig 2D). Relapsing patients have higher numbers of CD8+ CD28neg PD-1+ T cells at +3 months after transplant (Fig 2E), but before detection of clinical disease, indicating their applicability in identifying patients at higher risk of relapse. PD-1 blockade revives the proliferation and cytokine secretion of the hyporesponsive, CD8+ CD28neg PD-1+ T cells in vitro (Fig 3).
CONCLUSION: These results identify T cell exhaustion/senescence as a distinguishing feature of relapse and support early introduction of immunotherapy to stimulate antitumor immunity after ASCT.
Lesokhin:Genentech: Research Funding; Bristol Myers Squibb: Consultancy, Research Funding; Aduro: Consultancy; Janssen: Consultancy, Research Funding; Efranat: Consultancy. Giralt:CELGENE: Consultancy, Honoraria, Research Funding; SANOFI: Consultancy, Honoraria, Research Funding; AMGEN: Consultancy, Research Funding; JAZZ: Consultancy, Honoraria, Research Funding, Speakers Bureau; TAKEDA: Consultancy, Honoraria, Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This icon denotes a clinically relevant abstract

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal