Background.
Acute graft versus host disease (GvHD) remains a major cause of treatment failure post allogeneic stem cell transplantation (HSCT). CD28-CD80/CD86 axis T cell co-stimulation blockade with CTLA4-Ig using abatacept (AB) is an emerging acute GvHD prophylaxis strategy1. After myeloablative conditioning (MAC), together with methotrexate and calcineurin inhibition, four doses of AB commencing day (d)-1 is being investigated in the unrelated HLA 7/8 and 8/8 mismatched/matched donor settings, mostly in children2. It is unknown if AB can be de-escalated in adults to three doses for HLA matched sibling (SIB) or unrelated 10-12/12 donors (MUD), or combined with post-transplant cyclophosphamide (PT-CY) GvHD prophylaxis3, or combined with treosulfan (TREO)-based MAC4, which are other emerging transplant strategies. We investigated the safety and efficacy of this combination, and compared outcomes to a standard anti-CD52 directed IgG1 alemtuzumab (CH) prophylaxis regimen5 in a reduced intensity conditioning (RIC).
Methods.
We report a single centre consecutive cohort study using peripheral blood stem cells from SIB or HLA 10-12/12 MUDs, for HSCT in adults with blood cancer indications (intuitional registration: 1920-31). One group received AB GvHD prophylaxis while the other group received CH.
During 2018-2019, the AB cohort received MAC with IV fludarabine 150 mg/m2, IV TREO 42 mg/m2, and 2 Gy total body irradiation (TBI). GvHD prophylaxis comprised three doses of IV AB 10 mg/kg on d+5, +14, and +28. This was combined with IV PT-CY 50 mg/kg on d+3 and +4, IV tacrolimus 0.03 mg/kg/d infusion from d+5, and IV mycophenolate mofetil 15 mg/kg tds from d+53. The MAC AB cohort was compared to a RIC CH cohort, chosen to be biased against AB. During 2017-2018 the CH cohort received RIC with IV fludarabine 150 mg/m2and IV melphalan 140 mg/m2. GvHD prophylaxis comprised IV CH 10 mg on d-7 to d-3, and IV ciclosporin 1.5 mg/kg bd from d-1.
The primary outcome measure was the incidence of acute GvHD6 at d100 and 180 post HSCT. Secondary outcomes measures were the incidence of graft failure, mixed donor chimerism, donor lymphocyte infusion (DLI), CMV reactivation, relapse, overall survival, at d100 and 180. The AB and CH cohorts were indirectly compared.
Results.
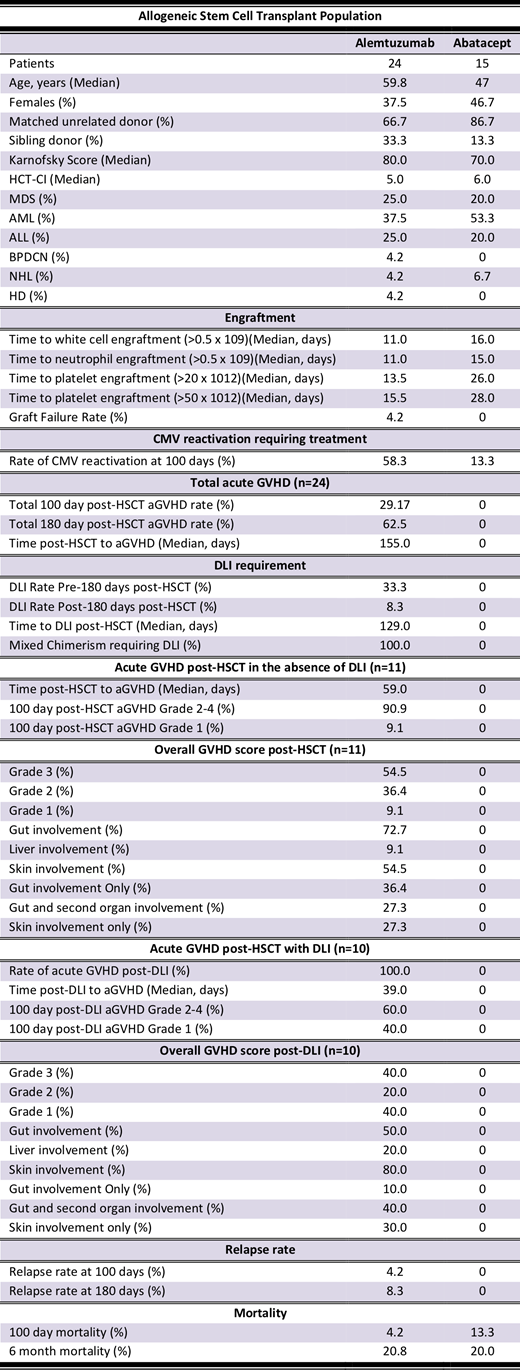
A total of 39 patients were included in this study, with 15 consecutive patients in the AB group (Table). The AB group had a median Karnofsky score of 70% and HCT-CI of 6; significant comorbidity greater than other reports1,2. The major cancer indications were acute leukaemia and myelodysplastic syndrome (MDS). The AB and CH groups were largely balanced. There were no AB related infusion reactions. As expected for MAC, the AB group had a numerically but not clinically significantly slower time to engraftment relative to the RIC CH group. There was no AB group graft failure.
Remarkably, the AB group did not experience acute GvHD compared to the CH group by d100 (P=0.03) and d180 (P<0.01). In contrast, by d100, 29% in the CH group experienced acute GvHD after a median of 59 days post HSCT. No AB group patients experienced mixed donor chimerism (P<0.01), nor required a DLI (P<0.01) compared to the CH group, where all had <99% T cell or whole blood values at d100. Consequently, 45% of the CH cohort without acute GvHD were treated with DLI, with all patients experiencing acute GvHD at a median of 39 days post DLI (Table).
Other secondary outcomes were similar. The AB group experienced a CMV reactivation rate of 67% in IgG seropositive patients. Unfortunately, 93% of CMV IgG positive patients in the CH group experienced CMV reactivation. No relapses were observed by d180 in the AB group, whereas in the CH group, 8% of patients experienced relapse. Mortality at d180 was 20% in the MAC AB group, and 21% in the RIC CH group.
Conclusions.
Acute GvHD prophylaxis with only three doses of AB and PT-CY post TREO-based MAC appears to be safe and efficacious in the SIB and MUD settings, in a highly co-morbid adult blood cancer population. This AB approach seems favourable compared to CH despite RIC, where acute GvHD without DLI or post DLI remains significant. A prospective multicentre clinical trial with AB in this setting seems warranted to confirm these remarkable findings of patient benefit.
References.
1. Biol Blood Marrow Transplant (BBMT) 2013;19:1638-49.
2. NCT01743131.
3. Lancet Haematol 2019;6:e132-43.
4. Cancer 2017;123:2671‐79.
5. BBMT 2017;23:805-12.
6. BBMT 2016;22:4-10.
No relevant conflicts of interest to declare.
Abatacept, alemtuzumab, cyclophosphamide all the for the indication of graft versus host disease (GvHD) prophylaxis.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal