Introduction: Acute myeloid leukemia (AML) is a hematological neoplasm with poor clinical outcomes. The introduction of the BCL2 inhibitor venetoclax in combination with hypomethylating agent has improved response rates in older patients with AML by targeting the metabolism of leukemic stem cells (LSCs). Resistance, however, is unfortunately still common. Thus, there remains a need to enhance the clinical efficacy of venetoclax by combining it with novel agents. In diverse AML models, lysine-specific demethylase 1 (LSD1) inhibition promotes cellular differentiation and reduces LSCs. We therefore asked if LSD1 inhibition could enhance the efficacy of venetoclax in the treatment of AML.
Methods: We have studied the effect of single-agent and co-treatment with venetoclax and LSD1 inhibitor bomedemstat on cell proliferation in human AML cell lines (N=3), Hoxa9/ Meis1(H9M)- and MN1-transformed murine myeloid progenitor lines as well as primary AML patient samples (N=5). We have analyzed the effect of co-treatment with these two compounds on functional cellular bioenergetics in MOLM-13 cells using Seahorse extracellular flux analyzer and followed up by performing NMR spectrometry to study changes in metabolite abundance. Further, we have studied the efficacy of this drug combination in vivo in patient-derived xenograft (PDX) AML models (N=2).
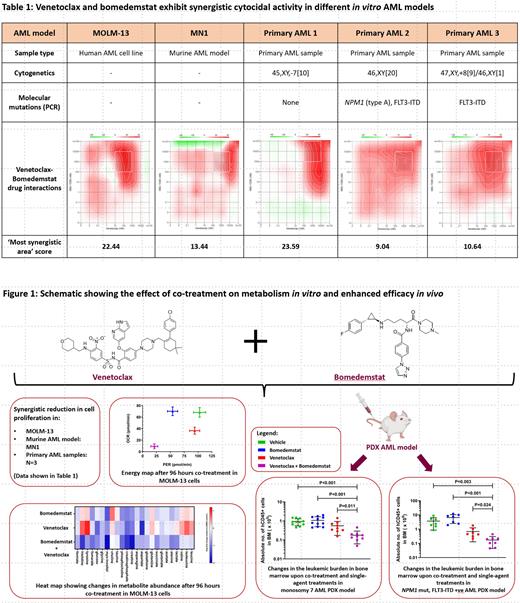
Results: Human AML cell lines showed a differential cytocidal activity at 96 hours (h) to venetoclax with MOLM-13 (IC 50 0.04 µM) being most sensitive and OCI-AML3 most resistant (IC 50 9.81 µM). A similar range (between 0.15 µM and 7.3 µM) was also observed in the primary AML samples. For bomedemstat, we observed a time-dependent response where the IC 50 decreased over time, in particular, in the most sensitive model systems. In murine H9M cells, the IC 50 decreased from 30.43 µM at 48 h to 0.034 µM at 96 h and further down to 0.0068 µM at 168 h. Combining bomedemstat and venetoclax, the IC 50 for venetoclax decreased; and synergism was observed in MOLM-13 cells, the most sensitive AML model, but no synergism was observed in venetoclax-resistant cells. Synergism was also observed in MN1 cells as well as 3 of the 5 primary AML samples studied (Table 1).
To investigate the mechanism of the observed synergism, we explored the effect of these treatments on metabolic pathways using the Seahorse extracellular flux analyzer. LSD1 inhibition had no effect on oxidative phosphorylation (measured as oxygen consumption rate (OCR)). However, glycolysis, measured as proton efflux rate (PER), was significantly reduced from 102.88 ± 11.03 pmol/min to 54.17 ± 6.18 pmol/min (P <0.01) after 96 h of treatment with 1μM bomedemstat. With the combined treatments, we observed a reduction in both OCR and PER. This synergistic effect was further investigated by studying metabolite abundance using NMR spectrometry where co-treatment showed a reduction in metabolite abundance across a broad spectrum of metabolic pathways (Fig. 1).
To determine if the observed combined effect of bomedemstat and venetoclax could enhance activity against AML cells in vivo, we employed a PDX model with two primary AML samples. In the first model system (monosomy 7), co-treatment resulted in a significant reduction in human CD45-positive cells as compared to vehicle (P <0.001) and single-agent treatments with venetoclax (P= 0.011) and bomedemstat (P <0.001). Similarly, in the second PDX model ( NPM1-mutated, FLT3-ITD positive), the co-treatment exhibited a significant reduction in leukemic burden in bone marrow as compared to vehicle (P=0.003) and single-agent treatments with venetoclax (P= 0.024) and bomedemstat (P < 0.001) (Fig. 1).
Conclusion: The combination of bomedemstat and venetoclax had synergistic cytocidal effects on AML cell line and primary AML cells in vitro. It significantly reduced the leukemic burden in PDX AML models and synergistically downregulated cellular energy metabolism. These findings suggest that combining venetoclax with LSD1 inhibition holds promise as a combination treatment in AML and warrants further clinical investigation.
Disclosures
Khalaf:Pfizer: Consultancy, Honoraria; Novartis: Consultancy, Honoraria; Paladin: Consultancy, Honoraria; Astellas: Consultancy, Honoraria; Jazz: Consultancy, Honoraria; Taiho: Consultancy, Honoraria; Abbvie: Consultancy, Honoraria; BMS: Consultancy, Honoraria. Garcia-Horton:Avir Pharma: Honoraria; BMS: Honoraria. Foley:Novartis: Speakers Bureau; Gilead: Speakers Bureau; Janssen: Speakers Bureau. Leber:Astex: Honoraria, Speakers Bureau; Celgene: Honoraria, Speakers Bureau; Otsuka: Honoraria, Speakers Bureau; Bristol Myers Squibb Canada: Honoraria, Speakers Bureau; Roche: Honoraria, Speakers Bureau; Alexion Canada: Honoraria, Speakers Bureau; Astellas: Honoraria, Speakers Bureau; AMGEN: Honoraria, Speakers Bureau; Palladin Canada: Honoraria, Speakers Bureau; Pfizer Canada: Consultancy, Honoraria, Speakers Bureau; Janssen Canada: Honoraria, Speakers Bureau; Novartis Canada: Consultancy, Honoraria, Speakers Bureau; Taiho Canada: Honoraria; Jazz Canada: Honoraria, Speakers Bureau; AbbVie Canada: Consultancy, Honoraria, Speakers Bureau; Treadwell: Honoraria, Speakers Bureau; Gilead/KITE: Honoraria, Speakers Bureau. Walker:Sanofi: Honoraria, Research Funding. Lepic:Sanofi: Honoraria. Kleppe:Imago Biosciences, Inc., a subsidiary of Merck & Co, Inc: Current Employment. Rienhoff:Imago Biosciences, Inc., a subsidiary of Merck & Co, Inc: Ended employment in the past 24 months. Berg:Incyte: Other: Travel Funding; Celgene: Honoraria, Other: Travel Funding; Abbvie: Other: Travel Funding; Riemser Pharma GmbH: Honoraria; Jazz Pharmaceuticals: Honoraria; Imago Biosciences (a subsidiary of Merck): Research Funding; Bristol Myers Squibb: Honoraria; Takeda Pharma: Honoraria; AVIR Pharma: Honoraria; Astellas: Other: Travel Funding; Alexion: Other: Travel Funding.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal