Hematopoietic stem cell transplantation (HSCT) is followed by profound immunodeficiency. Thymic function is necessary for de novo generation of T cells after HSCT. Circulating CD45RA+ naive T-cell levels are predictive of antigen-specific T-cell responses in the absence of graft-versus-host disease (GVHD). These T cells may not represent recent thymic emigrants, since naive T cells may maintain this phenotype if not antigen-activated. To accurately measure thymic output after HSCT and determine the factors that influence thymic function, T-cell receptor excision circles (TRECs) were examined in CD4+ and CD8+ cells from a cross-section of patients following HSCT. TREC levels rose weeks after HSCT and could be detected in patients 6 years after HSCT. TREC levels correlated with the frequency of phenotypically naive T cells, indicating that such cells were not expanded progeny of naive T cells present in the donor graft. Chronic GVHD was the most important factor that predicted low TREC levels even years after HSCT. Patients with a history of resolved GVHD had decreased numbers of TREC, compared with those with no GVHD. Because few adults had no history of GVHD, it was not possible to determine whether age alone inversely correlated with TREC levels. Recipients of cord blood grafts had no evidence of decreased TREC induced by immunosuppressive prophylaxis drugs. Compared with unrelated donor grafts, recipients of matched sibling grafts had higher TREC levels. Collectively, these data suggest that thymopoiesis is inhibited by GVHD. Larger studies will be needed to determine the independent contributions of age and preparative regimen to post-transplant thymopoietic capacity.
Introduction
Following hematopoietic stem cell transplant (HSCT), there is a prolonged period of profound immune deficiency, which includes defects in thymopoiesis.1 This immune deficiency contributes to the high incidence of opportunistic infection, which continues for years after HSCT.2,3 The etiology of the immune defect is multifactorial. Thymopoietic defects resulting in decreased ability to generate new T cells after HSCT are important since complete immune reconstitution ultimately depends on the generation of new T cells from hematopoietic stem cell (HSCs), just as long-term myeloid and erythroid reconstitution depends on HSC engraftment. Transfer of committed progenitors or mature donor-derived T cells may permit short-term immune function. Analyses of patients after HSCT have demonstrated that the presence of immune function at 1 year or later was correlated with the number of CD4+CD45RA+ naive T cells, suggesting that immune function at later time points is dependent on the ability to generate new T cells.4 5
The factors that inhibit thymic function may include age, graft-versus-host disease (GVHD), and direct thymic damage from chemoradiotherapy. In multiple studies of normal individuals, recipients of high-dose chemotherapy, HSCT recipients, and patients infected with human immunodeficiency virus, age has been inversely correlated with thymopoiesis.4-7 GVHD after HSCT also leads to thymic insufficiency, possibly by direct attack on the thymic stroma by allogeneic effector cells.8,9 Confounding the understanding of the role of GVHD in the pathogenesis of post-HSCT immune deficiency are the immunosuppressive effects of the medications used to prevent or treat GVHD. Besides age and GVHD, direct damage to the subset of thymic epithelial cells leading to loss of production of the thymopoietic cytokine interleukin-7 (IL-7) and consequently decreased ability to support post-HSCT thymopoiesis has been shown in a murine model of HSCT (Chung et al, unpublished data, January 2001).10
Several methods have been used to measure thymopoietic capacity. Thymic size as measured by volumetric computerized tomographic measurements has been correlated with numbers of CD4+CD45RA+naive T cells.6,11 The number of phenotypically naive T cells has been shown to correlate with antigen-specific function.2,3 However, there are concerns about limitations of estimating thymic function on the basis of naive T-cell phenotype alone. T cells expressing a naive phenotype are not necessarily accurate surrogate markers of thymic function. Following thymic emigration, CD45RA+ naive T cells can have a long quiescent life span,12,13 may proliferate in an antigen-independent manner,14 or may rapidly convert to CD45RO+memory/effector phenotype T cells.15 Furthermore, naive T-cell markers may be acquired by memory T cells,15 and naive and memory cells may have overlapping phenotypes, especially among CD8+ T cells.16 17
A different assay that has been more recently used as a measure of thymopoietic capacity is the frequency of T-cell receptor excision circles (TRECs) among peripheral blood T cells. A byproduct of T-cell receptor rearrangement includes a circular episome of DNA, which is stable and does not replicate with cellular proliferation.7 18 Because recent thymic emigrants (RTEs) have TRECs at levels that would not have been affected by peripheral expansion and cellular replication, quantification of TREC levels after HSCT represents a sensitive measurement of thymopoietic capacity leading to the generation of mature peripheral blood T cells after HSCT.
In this study, we examined levels of TRECs in CD4+ and CD8+ lymphocytes from patients after HSCT. The first recipients studied were a cohort of patients in a cross-sectional analysis of TREC levels. As data from the cross-sectional analyses indicated an inverse relationship between GVHD and TRECs, we then prospectively analyzed a smaller cohort of recipients of umbilical cord blood cells (CBCs) who received prophylaxis for GVHD. The results indicate that GVHD and age were the most important predictors of low TREC levels after HSCT, but that the administration of standard immunosuppressive medications did not affect TREC levels.
Patients, materials, and methods
Patients
Patients were recruited for the study from 4 institutions: Childrens Hospital Los Angeles (43 subjects); the University of Minnesota Hospital (12 subjects); The University of Texas Southwestern Medical Center (10 subjects); and Baylor University Medical Center, Dallas (2 subjects). The subjects were children and adults who received HSCT after ablative pre-HSCT conditioning. The sources of HSCs included histocompatible sibling marrow, histocompatible sibling mobilized peripheral blood progenitor cells, HLA-matched unrelated marrow, and related or unrelated umbilical CBCs. The patients were conditioned for HSCT with either busulfan-cyclophosphamide, total body irradiation (TBI) and etoposide, TBI and cyclophosphamide, TBI and etoposide and cyclophosphamide, or cyclophosphamide and antithymocyte globulin (ATG) as previously described.19-26 The transplants were performed for the treatment of a variety of hematologic malignancies, aplastic anemia, or genetic diseases (summarized in Table1). The studies were performed in accord with research protocols approved by the Institutional Review Boards of the respective institutions.
Characteristics of patients in cross-sectional analysis
| . | No GVHD . | Previous episode of GVHD . | GVHD at time of sample . |
|---|---|---|---|
| HSCT recipients 25 years of age | |||
| Total no. | 21 | 17 | 18 |
| Diagnosis* | |||
| Acute leukemia | 6 | 9 | 10 |
| Chronic myelogenous leukemia | 3 | 2 | 1 |
| Lymphoma | 1 | 1 | |
| Aplastic anemia | 2 | 4 | |
| Primary immunodeficiency | 7 | 5 | 3 |
| Genetic hematologic disease | 2 | ||
| Source of HSCs† | |||
| Histocompatible | 12 | 5 | 5 |
| MUD | 7 | 10 | 11 |
| Mismatched family | 2 | 2 | 2 |
| Age at time of HSCT | 1.5-25 years (median = 8) | 0.3-20 years (median = 9.4) | 0.4-16.7 years (median = 8.1) |
| Time after HSCT of TREC analysis | 0.25-5 years (mean = 2.1) | 0.5-7 years (mean = 2.3) | 0.2-12 years (mean = 2.1) |
| Immunosuppression at time of TREC analysis‡ | 3/21 | 7/17 | 16/18 |
| HSCT recipients > 25 years of age | |||
| Total no. | 1 | 6 | 4 |
| Diagnosis* | |||
| Acute leukemia | 2 | ||
| Acute myeloid leukemia | 1 | ||
| Chronic myelogenous leukemia | 1 | 3 | 2 |
| Aplastic anemia | 1 | ||
| MDS | 1 | ||
| Source of HSCs† | |||
| Histocompatible | 1 | 5 | 2 |
| MUD | 1 | 2 | |
| Age at time of HSCT | 37 | 41-58 years (median = 51.5) | 44-53 years (median = 48.5) |
| Time after HSCT of TREC analysis | 2.25 | 0.6-2.6 years (mean = 1.3) | 0.7-7.3 years (mean = 2.4) |
| Immunosuppression at time of TREC analysis‡ | 0/1 | 3/6 | 4/4 |
| . | No GVHD . | Previous episode of GVHD . | GVHD at time of sample . |
|---|---|---|---|
| HSCT recipients 25 years of age | |||
| Total no. | 21 | 17 | 18 |
| Diagnosis* | |||
| Acute leukemia | 6 | 9 | 10 |
| Chronic myelogenous leukemia | 3 | 2 | 1 |
| Lymphoma | 1 | 1 | |
| Aplastic anemia | 2 | 4 | |
| Primary immunodeficiency | 7 | 5 | 3 |
| Genetic hematologic disease | 2 | ||
| Source of HSCs† | |||
| Histocompatible | 12 | 5 | 5 |
| MUD | 7 | 10 | 11 |
| Mismatched family | 2 | 2 | 2 |
| Age at time of HSCT | 1.5-25 years (median = 8) | 0.3-20 years (median = 9.4) | 0.4-16.7 years (median = 8.1) |
| Time after HSCT of TREC analysis | 0.25-5 years (mean = 2.1) | 0.5-7 years (mean = 2.3) | 0.2-12 years (mean = 2.1) |
| Immunosuppression at time of TREC analysis‡ | 3/21 | 7/17 | 16/18 |
| HSCT recipients > 25 years of age | |||
| Total no. | 1 | 6 | 4 |
| Diagnosis* | |||
| Acute leukemia | 2 | ||
| Acute myeloid leukemia | 1 | ||
| Chronic myelogenous leukemia | 1 | 3 | 2 |
| Aplastic anemia | 1 | ||
| MDS | 1 | ||
| Source of HSCs† | |||
| Histocompatible | 1 | 5 | 2 |
| MUD | 1 | 2 | |
| Age at time of HSCT | 37 | 41-58 years (median = 51.5) | 44-53 years (median = 48.5) |
| Time after HSCT of TREC analysis | 2.25 | 0.6-2.6 years (mean = 1.3) | 0.7-7.3 years (mean = 2.4) |
| Immunosuppression at time of TREC analysis‡ | 0/1 | 3/6 | 4/4 |
GVHD indicates graft-versus-host disease; HSC, hematopoietic stem cell; MUD, matched unrelated donor; HSCT, hematopoietic stem cell transplantation; TREC, T-cell receptor excision circle; and MDS, myelodysplastic syndrome.
Acute leukemias were acute lymphoblastic (22) and nonlymphoblastic (3) leukemia. Primary immunodeficiencies were severe combined immunodeficiency (8), Wiskott-Aldrich syndrome (3), CD40 ligand deficiency (1), and hemophagocytic lymphohistiocytosis (3). Genetic hematologic diseases were sickle cell disease (1) and chronic granulomatous disease (1).
Matched unrelated donors were 6/6 HLA matched (18) or 1 locus mismatched (11). Mismatched family donors were haploidentical and T-cell depleted (5), or 1 HLA locus mismatch without T-cell depletion (1).
Immunosuppression at time of TREC analysis included prednisone (29), cyclosporine or tacrolimus (36), and mycophenolate (11).
GVHD management and scoring
All patients received prophylactic immunosuppression after HSCT to prevent GVHD. GVHD prophylaxis included methotrexate and/or cyclosporine or tacrolimus for recipients of histocompatible marrow; methorexate, cyclosporine (CSA), and ATG for recipients of HSCT from matched unrelated donors (MUDs); and CSA and methylprednisolone for recipients of unrelated umbilical CBCs. Some patients were still receiving prophylactic immunosuppression at the time of analysis. Of the patients, 58% required continuing immunosuppression for the treatment of GVHD. GVHD was scored by standard criteria for acute or chronic GVHD.27
Cell isolation
Blood samples were collected in heparinized tubes for isolation of peripheral blood mononuclear cells (PBMCs). After Ficoll-Hypaque density centrifugation, the CD4+ or CD8+ T cells were isolated from the mononuclear cells by MACS magnetic microbead separation (Miltenyi Biotec, Auburn, CA).
Flow cytometry
To quantify phenotypically naive T cells in CBC recipients, PBMCs were stained with fluorochrome-conjugated antibodies against CD4, CD8, CD45RO, and CD27 (Becton Dickinson, San Jose, CA) and analyzed by 4-color flow cytometry (FACSCalibur, Becton Dickinson). Naive T-cell percentages in each CD4+ or CD8+ subset were measured by gating on CD45RO−CD27+ cells by means of Paint-a-Gate software (Becton Dickinson). To quantify phenotypically naive CD4+ T cells in the other study subjects, PBMCs were stained with fluorochrome-conjugated antibodies against CD4 and CD45RA, (Becton Dickinson). Naive CD4+T-cell percentages were measured by gating on CD45RA+CD4+ cells.
Measurement of TRECs in sorted cells
Quantification of TRECs in sorted CD4+ and CD8+ T cells was performed by real-time quantitative polymerase chain reaction (PCR) by means of the 5′ nuclease (TaqMan) assay with an ABI7700 system (PerkinElmer, Norwalk, CT) as previously described.7 28 CD4+ and CD8+ cells were lysed in 100 μg/mL proteinase K (Boehringer, Indianapolis, IN) for 1 hour at 56°C, and then 10 minutes at 95°C, at 107cells per milliliter. Real-time quantitative PCR was performed on 5 μL of cell lysate (equivalent to 50 000 cells) with the primers 5′-CACATCCCTTTCAACCATGCT and 5′-GCCAGCTGCAGGGTTTAGG, and probe FAM-5′-ACACCTCTGGTTTTTGTAAAGGTGCCCACT-TAMRA (MegaBases, Chicago, IL). PCR reactions contained 0.5 U Platinum Taq polymerase (Gibco, Grand Island, NY) and 3.5 mM MgCl2, 0.2 mM dNTPs, 500 nM of each primer, 150 nM probe, and Blue-636 reference (MegaBases). Conditions were 95°C for 5 minutes, 95° for 30 seconds, and 60° for 1 minute for 40 cycles. A standard curve was plotted, and TREC values for samples were calculated by the ABI7700 software. Samples were analyzed in duplicate.
Statistical analyses
Statistical analyses (2-tailed Mann-Whitney test [Pvalues]), and Spearman's rank correlation coefficients (rvalues) were calculated by means of Excel and Prism software. Anr value greater than 0.3, or less than −0.3, and aP value less than .05 were considered significant.
Results
Patient cohorts for cross-sectional analysis
As shown in Table 1, there were 67 patients with primary diagnoses of acute leukemia, chronic myelogenous leukemia, lymphoma, aplastic anemia, severe combined immunodeficiency (SCID) or other primary immunodeficiencies, or other genetic diseases. All patients, including those with SCID, received HSCT after ablative chemotherapy or chemoradiotherapy. For purposes of analysis, patients were divided into those younger or older than 25 years of age (Table 1). Patients 25 years of age or younger could be divided into approximately equal numbers of patients who either had never had clinical GVHD (21 patients), had had a previous history of an episode of GVHD but no clinical GVHD at the time of TREC analysis (17 patients), or had clinical GVHD at the time of analysis (18 patients). Of the 11 patients older than 25 years of age, only 1 had no history of GVHD; 6 had a previous history of an episode of GVHD, and 4 had clinical GVHD at the time of analysis.
TREC levels in patients without GVHD
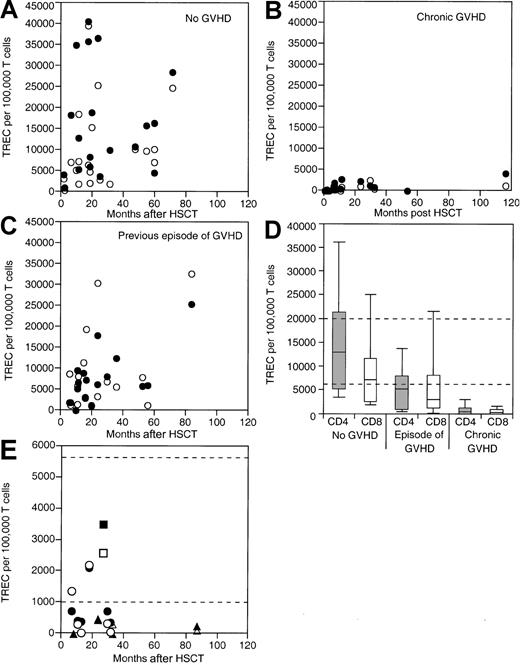
TREC levels were measured in a cross-section of children and adults through the age of 25 years following HSCT with bone marrow as the stem cell source (Figure1A,D). By 2 months after HSCT, the levels of TREC had risen, especially in those who had no GVHD. In the group of patients with no GVHD, TREC levels were highest 1 to 2 years after HSCT, but were found to be comparably high (10 000 to 30 000 TRECs per 105 T cells) in patients at 3 to 7 years. The rise in TREC levels was observed among both CD4+ and CD8+ T cells. These data (and other data not shown at time points soon after HSCT) indicate that TREC levels can increase substantially beginning 2 months after HSCT in children and adults who have no history of GVHD.
Cross-sectional analysis of TREC levels in relationship to GVHD.
(A) Individual CD4+ (●) and CD8+ (○) T-cell TREC levels are shown for a cross-section of patients through 25 years of age with no history of GVHD. (B) Patients through 25 years of age with chronic GVHD. (C) Patients through 25 years of age with a history of GVHD. (D) A composite box plot for TREC levels is shown for all 3 groups. The top, bottom, and line through the middle of the box correspond to the 75th, 25th, and 50th percentile (median) respectively. The whiskers on the bottom and top extend from the 10th percentile and top 90th percentile, respectively. (E) Individual CD4+ (closed symbols) and CD8+ (open symbols) T-cell TREC levels are shown for a cross-section of patients older than 25 years of age with no history of GVHD (squares), with chronic GVHD (triangles), and with a history of GVHD (circles). Normal range of TREC levels for individuals of ages in groups studied are shown lying between the dashed lines.
Cross-sectional analysis of TREC levels in relationship to GVHD.
(A) Individual CD4+ (●) and CD8+ (○) T-cell TREC levels are shown for a cross-section of patients through 25 years of age with no history of GVHD. (B) Patients through 25 years of age with chronic GVHD. (C) Patients through 25 years of age with a history of GVHD. (D) A composite box plot for TREC levels is shown for all 3 groups. The top, bottom, and line through the middle of the box correspond to the 75th, 25th, and 50th percentile (median) respectively. The whiskers on the bottom and top extend from the 10th percentile and top 90th percentile, respectively. (E) Individual CD4+ (closed symbols) and CD8+ (open symbols) T-cell TREC levels are shown for a cross-section of patients older than 25 years of age with no history of GVHD (squares), with chronic GVHD (triangles), and with a history of GVHD (circles). Normal range of TREC levels for individuals of ages in groups studied are shown lying between the dashed lines.
TREC levels in patients with GVHD
Patients with GVHD were divided into 2 groups for analysis. Some patients with no clinical evidence of GVHD at the time of study had prior histories of either acute or chronic GVHD; other patients were studied with concurrent chronic GVHD. In both patient populations, the levels of TRECs were lower than in the patients who had no history of GVHD (Figure 1B-D). The decreased levels of TRECs were most profound in the group of patients with active chronic GVHD at the time of study. In this group of patients, TREC levels were typically lower than 2000 TRECs per 105 T cells, irrespective of the time period after HSCT. TREC levels were significantly lower among both the CD4+ and CD8+ T cells in the active chronic GVHD group (P < .0001 for both CD4+ and CD8+ T cells).
In the patients with a history of GVHD but no current GVHD, TREC levels were also lower than normal, but were significantly higher than those observed in the chronic GVHD group (Figure 1C-D). The decrease in TREC levels in the patients with a history of GVHD compared with the group with no GVHD was significant for CD4+ T cells (P = .0017), but not CD8+ T cells (P = .70). The TREC levels were higher in the group of patients with a history of GVHD than in those with active chronic GVHD (P < .0001 for CD4+ T cells andP = .0003 for CD8+ T cells). Figure 1D shows a composite figure of the range and median levels of TRECs in the CD4+ and CD8+ cells for patients up to 40 months after transplant in the 3 GVHD groups.
Relationship of TREC levels to age
Because thymic function and TREC levels normally decrease with age, we examined whether TREC levels and age were inversely correlated after HSCT. In the group of patients with no GVHD, there was no correlation between TREC levels and age (r = −0.2631, 95% confidence interval [CI] = −0.6322 to 0.2035,P = .25, for CD4+ cells; andr = −0.1475, 95% CI = −0.5547 to 0.3160,P = .25, for CD8+ cells). However, because of the confounding relationship between older age and GVHD, there was only one patient older than 25 years of age who did not develop GVHD and who was excluded from the analysis. Thus, through age 25 years, age had no effect on TREC levels if there was no history of GVHD. Furthermore, in all patients with active chronic GVHD, TREC levels were similarly unaffected by age (r = −0.0792, 95% CI = −0.3557 to 0.4859, P = .272, for CD4+cells; and r = −0.2488, 95% CI = −0.0195 to 0.6078,P = .25, for CD8+ cells).
In all patients with a history of GVHD but no active chronic GVHD, there was a significant inverse relationship between TREC levels and age for both CD4+ T cells (r = −0.5682, 95% CI = −0.7991 to −0.1911, P = .0047) and CD8+ T cells (r = −0.72, 95% CI = −0.87 to −0.42, P = .0001). It is important to note that this negative correlation between age and TREC levels only became significant at ages above 16 years old (r = −0.8883, 95% CI = −0.95 to −0.2, P = .0031, for CD4+ T cells; and r = −0.7667, 95% CI = −0.87 to −0.39,P = .0041, for CD8+ T cells). Below the age of 16 years, there was no significant effect of age on TREC levels (r = −0.0945, 95% CI = −0.6 to 0.47,P = .748, for CD4+ T cells; andr = −0.4989, 95% CI = −0.82 to 0.06,P = .0694, for CD8+ T cells). Thus, increasing age alone did not appear to prevent the development of TREC after HSCT; rather, the combination of increasing age and GVHD was the major determinant of low TREC levels after transplant (Figure 1E). However, it should be emphasized that this conclusion derives from 2 patient groups (with and without GVHD) that have different age ranges. Thus, although it can be concluded that age per se is not necessarily a barrier to increased TREC levels after HSCT, one cannot ascertain with absolute certainty whether age-related changes might have occurred independently of GVHD.
CBC recipients
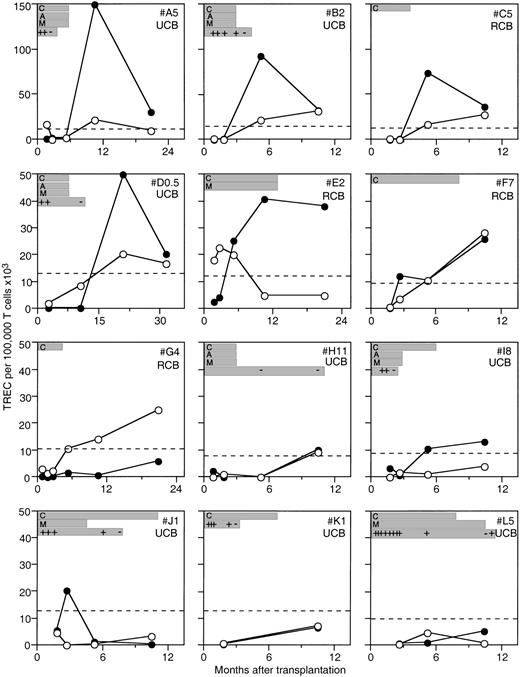
Because GVHD and age were such significant determinants of TREC levels, we next prospectively analyzed a cohort of patients with a low risk of GVHD. Umbilical CBC transplants are associated with a lower incidence of GVHD than marrow or mobilized peripheral blood stem cell transplants, for any given degree of histoincompatibility.23,24 The patients who had received CBCs were analyzed prospectively for TREC levels up to 33 months after transplant. These patients received prophylactic immunosuppression with cyclosporine and glucocorticoids, with or without ATG. The CBC recipients developed TREC at levels consistent with the cross-sectional analysis of the marrow transplant patients (Figure2). Indeed, TREC levels rose to supranormal levels for age in many of the patients, perhaps reflecting what has been termed thymic rebound.6 The presence of clinical GVHD was found to inhibit a rise in TRECs, but TREC levels increased when GVHD was not active. In 3 patients, although immunosuppression was continued for at least 6 months in the absence of GVHD, TREC levels nevertheless increased (Figure 2). These data indicate that in the absence of GVHD, prophylactic immunosuppressive agents did not preclude thymopoiesis from occurring, as measured by rises in CD4+ and CD8+ TREC levels. However, it should be stressed that although this conclusion may be valid, more patients and comparisons with an age-matched group without immunosuppression will be required before more definitive conclusions can be made.
TREC levels in a cohort of prospectively evaluated pediatric patients undergoing CBC transplantation.
Individual CD4+ (●) and CD8+ (○) T-cell TREC levels are shown for patients at time points after undergoing CBC transplantation. Normal TREC levels for age are shown by the dashed line. Duration of immunosuppressive therapy is shown for cyclosporine (C), methylprednisolone (M), and ATG (A). Recorded instances of active GVHD (+) and no active GVHD (−) are also shown. The patients are identified by a letter followed by their age, and whether they had related CBC (RCB) or unrelated CBC (UCB) in the upper right of each plot. Patients E2, F7, and I8 all had increases in TREC levels while on immunosuppression.
TREC levels in a cohort of prospectively evaluated pediatric patients undergoing CBC transplantation.
Individual CD4+ (●) and CD8+ (○) T-cell TREC levels are shown for patients at time points after undergoing CBC transplantation. Normal TREC levels for age are shown by the dashed line. Duration of immunosuppressive therapy is shown for cyclosporine (C), methylprednisolone (M), and ATG (A). Recorded instances of active GVHD (+) and no active GVHD (−) are also shown. The patients are identified by a letter followed by their age, and whether they had related CBC (RCB) or unrelated CBC (UCB) in the upper right of each plot. Patients E2, F7, and I8 all had increases in TREC levels while on immunosuppression.
Matched unrelated vs histocompatible sibling donors
Previous studies have indicated that HSCT from MUDs was associated with lower numbers of CD45RA+ cells after HSCT than when histocompatible sibling marrow donors were used, especially in the context of clinical GVHD.5 TREC levels in the younger age group (through age 25 years) were significantly lower in recipients of MUD-derived HSCT than in recipients of histocompatible cells when analysis was done across all GVHD groups (P = .0007 for CD4+ T cells andP = .0073 for CD8+ T cells). The lower levels of TRECs in the MUD recipients were observed whether or not the unrelated CBCs were included in the analysis (P = .0007 for CD4+ T cells and P = .007 for CD8+ T cells, when CBC recipients were excluded). However, when the comparison between TREC levels in recipients of MUD-derived HSCT and histocompatible cells was performed separately within the no-GVHD, history-of-GVHD, and active-chronic-GVHD groups, then there was no significant difference, and it became clear that the lower TREC levels in the recipients of MUD-derived HSCT resulted from their increased incidence of GVHD. At older ages, age itself became a confounding factor, and the correlations between donor type and TREC levels were not significant (P = .05 for CD4+T cells and P = .29 for CD8+ T cells).
Relationship between frequency of TRECs and CD4+CD45RA+ or CD8+CD45RA+ cells
Previous studies of immune reconstitution in chemotherapy and HSCT recipients have used the cell-surface molecule CD45RA as a phenotypic marker of naive T cells.4-6 The frequency of TRECs and CD45RA+ T cells was compared in the CD4+ T cells from all patients in the cross-sectional analysis of this study. TREC levels and CD45RA+ cell numbers were significantly correlated. The correlation was present regardless of whether the patient had no GVHD (r = 0.5353, 95% CI = 0.092 71 to 0.8013, P = .0182), a history of GVHD (r = 0.6882, 95% CI = 0.3639 to 0.8636,P = .0004), or chronic GVHD (r = 0.7065, 95% CI = 0.4043 to 0.8696, P = .0002). The percentage of CD45RA+CD8+ T cells was not determined in these patients. Among the prospectively studied CBC recipients, TREC levels correlated significantly with the number of naive (CD45RO−CD27+) CD4+ cells (r = 0.7262, 95% CI = 0.5435 to 0.8432,P < .0001) and naive CD8+ cells (r = 0.5235, 95% CI = 0.2598 to 0.7145,P = .0003). The correlation of RTEs, as measured by TRECs, with the number of naive T cells suggests that most of the naive T cells were generated de novo.
Discussion
The major findings of our study are the following: (1) TREC levels rose weeks after HSCT despite immunosuppressive GVHD prophylactic agents; (2) TREC levels correlated with the frequency of naive phenotype T cells, indicating that such cells were not the expanded progeny of naive T cells present in the donor graft; (3) active chronic GVHD was the most important factor that predicted low TREC levels even years after HSCT; (4) patients with a history of resolved GVHD had decreased numbers of TRECs compared with those with no GVHD; (5) age did not affect TREC levels in patients with no GVHD through the age of 25 and had no effect on TREC levels of patients with active chronic GVHD at all ages; and (6) in patients with resolved GVHD, there was an inverse correlation between age and TRECs above the age of 16 years. These data indicate that a history of GVHD, presence of GVHD, and age, but apparently not the administration of immunosuppressive agents per se, were critical determinants of thymic output after HSCT.
Immune reconstitution after HSCT is increasingly recognized as a critical problem because it is a determinant of morbidity and mortality from opportunistic infections in recipients. Post-transplant T cells are derived from both mature T cells present in the donor graft and T cells that develop de novo in the recipient from transplanted donor stem cells. It is likely that the latter pathway of differentiation leads to long-term immune reconstitution, whereas the former results in transient adoptive transfer of immunity as well as GVHD mediated by cells with alloreactivity toward recipient antigens.
Over the past decade, a variety of techniques have been used to characterize the development of donor-derived T cells in the recipient after HSCT. The measurement of peripheral T-cell TRECs allows the quantification of T cells that have not undergone post-thymic clonal expansion. Because TRECs are rapidly diluted by clonal expansion, TREC levels probably better reflect the RTE population. The TREC assays described in this study were performed on sorted CD4+ and CD8+ T lymphocytes. This technique makes it possible to examine each T-cell population, which may have different dynamics of production or peripheral expansion. Furthermore, analyses of enriched T cells prevents artifacts caused by varying levels of non-T cells, such as monocytes, which affect the results of TREC studies that use only unfractionated PBMCs.
In our cross-sectional analyses, TREC levels correlated with phenotypically naive T-cell numbers in the adult HSCT recipients and in the pediatric transplants. The time delay between HSCT and the appearance of TRECs, and the correlation between TRECs and numbers of phenotypically naive T cells, suggest that the majority of naive T cells were RTEs, rather than the products of clonal expansion of pre-existing naive T cells that had been infused with the donor HSC product. Because the naive phenotype can be maintained by cells for long periods after thymic emigration and even after expansion in the periphery, TRECs are a more accurate measure of post-transplant thymic output.11-13 Peripheral expansion of naive T lymphocytes in the absence of thymic function can occur, resulting in changes in naive T-cell numbers independent of changes in TRECs29; therefore, the measurement of TRECs can determine the origin of T cells bearing a naive phenotype. This is all the more important given the degree of overlap between phenotypes among naive and memory T cells.16,17 Furthermore, increases in TREC levels can be detected much earlier than increases in naive T cells.7 28Thus, the TREC assay will be useful in studies of thymic output after HSCT and can complement phenotypic analysis of T cells while avoiding the confounding variables associated with accurate definition of naive T-cell numbers.
We observed that TRECs rose to high levels, often above normal for age, early after allogeneic HSCT in children and adults in whom GVHD did not occur, indicating that the thymus was playing a role in immune reconstitution. The major correlate of low TRECs after HSCT that we observed was the presence or history of GVHD. Patients with chronic GVHD had no evidence of the early rise in TREC levels observed in the first 2 years after HSCT and had persistently low TREC levels up to 10 years after transplant. Patients with a history of prior acute or chronic GVHD also had lower CD4+ and CD8+T-cell TREC levels than the patients with no history of GVHD, and this was statistically significant for CD4+ cells but not CD8+ cells. MUD transplantation was also associated with lower levels of TRECs and reflected the observation that GVHD occurred more frequently in MUD recipients than in recipients of histocompatible sibling cells.
GVHD is a complex phenomenon that could affect thymic function in many ways. Because GVHD is treated with immunosuppressive drugs that may affect thymic function, any observed effect of GVHD on TREC levels could be due to such drugs. The relative roles of GVHD and immunosuppressive drugs can be examined more critically in the cohort of patients who received CBC transplants. Some CBC recipients did not develop GVHD, but did receive prophylactic immunosuppressive treatment with cyclosporine and glucocorticoids. In spite of immunosuppression, these CBC recipients who were followed serially developed TRECs normally in the first year after HSCT. Rodent studies have indicated that the administration of cortisone to animals undergoing a graft-versus-host response results in a reduction of mature thymocytes, particularly those bearing the CD4 antigen.30 Although exogenous glucocorticoids are known to cause thymocyte apoptosis in vitro, little is known of their effect on thymic output in humans.56 Thus, the observed continued increase in TREC levels in spite of continued glucocorticoids was unexpected. Cyclosporine administration also did not prevent the generation of RTEs as measured by TREC levels. Cyclosporine administration has been shown to alter thymic selection mechanisms in nontransplanted rodents and in syngeneic BMT recipients.31Cyclosporine administration to nontransplanted rats has been shown to reduce immature cortical thymocyte numbers, deplete medullary CD4+CD8− and CD4−CD8+thymocytes, and cause release of a wave of RTE.32 33However, we are unaware of studies documenting decreased thymic output caused by cyclosporine in humans. Thus, our data indicate that GVHD causes more thymic dysfunction than the immunosuppressive drugs used to treat or prevent GVHD.
GVHD is caused by alloreactive or autoreactive T cells transferred into the recipient along with the HSCs. As the pool of potential HSC donors expands to include those who are increasingly disparate from the recipients, the frequency and severity of GVHD are rising. Patients with GVHD most frequently succumb to opportunistic infections, yet the mechanisms responsible for the immunodeficiency associated with GVHD are unclear. Our data provide evidence that thymic dysfunction caused by GVHD is an important component of the observed immune deficiency. In rodents, GVHD has been shown to result in the elimination of both immature CD4+CD8+ and mature CD4+CD8− thymocytes.34 A number of investigators have shown that GVHD induces thymic dysplasia associated with thymic involution, depletion of cortical and medullary thymocytes, epithelial cell destruction, and loss of Hassall's bodies, which collectively result in T-cell lymphopenia, immunodeficiency, and a failure of negative selection of potentially autoreactive T cells.9,35-43 Lapp and Seemayer have reported that the inflammatory cells that mediated acute or chronic GVHD in murine models also attacked the thymic microenvironment.8,9,35 The destruction of thymic epithelial cells by GVHD may prevent HSC-derived thymic precursor cells from receiving appropriate signals from the thymic microenvironment. Of note, thymic epithelial cells are biologically similar to other epithelial cells typically damaged by GVHD, such as skin epithelial cells.44 The adverse effect of GVHD on T lymphopoiesis in humans has been observed by investigators for quite some time, although accurate quantitation of thymopoiesis has only recently become available for studies in humans.45The data in our study indicate that the same mechanisms of thymic damage shown in rodent GVHD may be operative in the clinical situation.
The evidence for a deleterious effect of GVHD on thymic function notwithstanding, it must be stressed that the TREC content in a cell population is affected not only by changes in thymic output but also by the proliferative history of the cells. A measured increase in TREC levels in the naive T-cell compartment is likely to reflect thymic output more directly when the rate of peripheral T-cell division is low or constant. However, in the setting of GVHD, it is quite likely that the rates of T-cell division may be altered and thus lead to an underestimate of the rate of thymic output. It should also be appreciated that in patients with previous but now quiescent GVHD, low TREC levels may indicate either that previous thymic injury prevents TREC generation, even in situations in which homeostasis may be reset, and/or that the new thymic emigrants are expanding and diluting out the number of TRECs until T-cell homeostasis is re-established. In the latter case, the true number of RTEs would be underestimated by the TREC analysis performed in the present study. To distinguish between the effects of peripheral T-cell turnover and thymic output on TREC levels, it will be necessary in future studies to measure changes in TRECs within naive T-cell subsets and an independent marker of cell turnover such as Ki67 expression.
An alternative interpretation of the relationship between GVHD and TREC levels, which we cannot exclude, is that antecedent thymic dysfunction occurring as a consequence of chemotherapy6 or the aging process7 hindered the development of thymic-derived regulatory T cells after HSCT, contributing to the development of GVHD. Indeed, studies in rodents have shown that thymectomized mice are more susceptible to GVHD induction than euthymic mice.46,47Other aspects of HSCT have also been proposed as causing thymic dysfunction. Increasing age has been previously shown to be negatively correlated with numbers of naive T cells observed after HSCT or high-dose chemotherapy.5-7 Age has also been shown to be correlated with the risk of GVHD after allogeneic HSCT. However, in our study, GVHD and not age was the most important predictor of TREC levels. Age was inversely related to TREC levels only among patients with a history of GVHD, but not in those with either no GVHD (through age 25 years) or chronic GVHD. In previous studies, age was negatively correlated with naive T-cell numbers in patients without GVHD as well as in recipients of MUD bone marrow.4,5Similarly, studies of recipients of autologous HSCT for multiple myeloma showed an inverse relationship between TREC levels and age.28 Because of the observed powerful effect of GVHD on TREC levels, our current study is confounded by the association of increased age with GVHD, so that in patients with no GVHD we were only able to make significant correlations with TREC levels through age 25. Critical examination of autologous HSCT recipients and larger numbers of allogeneic HSCT recipients will be necessary to determine whether the previously observed effects of age on thymic function after HSCT are independent of the increased incidence of GVHD. Similarly, our study was not able to determine whether there are pretransplant conditioning regimens, such as TBI, that cause more thymic damage than other regimens. Studies of TREC levels in larger cohorts of patients randomized among regimens will be necessary to address the question of whether some forms of conditioning are more deleterious to thymic function than others.
A recent report in a cohort of infants with SCID demonstrated that although TRECs appeared in the first year after haplo-identical bone marrow transplantation (BMT), TREC levels declined after the second year and became undetectable in patients receiving a transplant more than 10 years earlier.48 In the present study, patients had rising TREC levels in the first 1 to 2 years after HSCT, in a manner similar to that reported by Patel et al.48 However, at 3 to 7 years after HSCT, some of the patients whom we describe had TREC levels that were nearly 10 times higher than reported in the cohort of SCID patients reported previously. The patients in the present study differ from the previously reported SCID patients in that all of the patients in the present study, regardless of diagnosis, received ablative chemotherapy or chemoradiotherapy in order to achieve HSC engraftment. Thus, patients in our study had donor HSCs whereas the SCID patients reported by Patel, who did not receive pre-HSCT preparative therapy, may have had persistent recipient HSCs with intrinsic defects in T-cell maturation. An alternative explanation for the apparently poorer thymic function observed in the previously reported SCID patients is that the intrinsic defect in thymic progenitors in SCID results in severe, irreversible thymic damage, a phenomenon that has been observed in a murine model.49
Several strategies to improve post-HSCT thymic function are logical developments of our study. First, early successful prevention of GVHD may result in less thymic damage and improved long-term production of T cells. Increased thymic production of T cells is likely to increase the repertoire of T cells28 and possibly decrease the high incidence of infection that has been observed after HSCT.2,3 Novel strategies to prevent GVHD by induction of tolerance have been described in murine,50-52 preclinical human,53 and clinical human studies.52,54Successful tolerance induction by cytokine adminstration or blockade of costimulatory pathways may also be able to prevent the thymic damage that has been observed in GVHD. Thus, prospective analyses of TREC levels in studies of GVHD prevention and treatment will be useful in determining whether the intervention was successful at preventing thymic damage caused by GVHD. Secondly, administration of thymopoietic cytokines may be able to increase thymic function after HSCT. IL-7 is a stroma-derived cytokine that is a major growth factor for thymopoiesis and that is normally produced by thymic epithelial cells. Systemic administration of recombinant IL-7 to murine BMT recipients normalizes thymopoiesis and improves post-BMT immune function.55Clinical studies of IL-7 have not yet been instituted; monitoring of TREC levels in such studies will be a useful measure of the effects on thymopoiesis.
In summary, we have provided evidence that thymopoiesis as measured by peripheral blood CD4+ and CD8+T-cell TREC levels occurs in children and adults after allogeneic HSCT and is adversely affected by GVHD, even if the GVHD is not active. The presence of active chronic GVHD was a major barrier to thymopoiesis. Patients receiving MUD transplants had lower TREC levels than those receiving sibling donor grafts. Umbilical cord blood transplant recipients had no evidence of inhibition of thymopoiesis despite being maintained on immunosuppressive drugs. These data indicate that strategies to restore thymopoiesis will probably be required for patients who have a preceding history of GVHD.
The authors thank Drs Ami Shah, Neena Kapoor, Donald B. Kohn, Gay M. Crooks, and Robertson Parkman, Kathy Wilson, RN, and members of the BMT units at Childrens Hospital Los Angeles and Fairview–University of Minnesota Hospitals and Clinics for generously providing patient samples; and Roberta Nicklow, Cynthia Eide, Pamela Phillips, and Matthew Johnson for their assistance.
R.A.K. is an Elizabeth Glaser Scientist of the Pediatric AIDS Foundation.
Supported by the General Clinical Research Center at Childrens Hospital Los Angeles, National Institutes of Health (NIH) grants M01 RR00043, R01 HL54729, and R21 HD37598, and the T.J. Martell Foundation (K.W.); NIH grants R01 AI 34495, 2 R37 HL56067, and P01 AI-35225, and the National Marrow Donor Program (B.R.B.); The Leukemia Association of North Central Texas (R.H.C.); and Leukemia and Lymphoma Society of America Translational Research Award 6540-00 (D.C.D.).
K.W. and B.R.B. contributed equally to this paper.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.
References
Author notes
Kenneth Weinberg, Division of Research Immunology/Bone Marrow Transplantation, Mailstop #62, Childrens Hospital Los Angeles, 4650 Sunset Blvd, Los Angeles, CA 90027; e-mail:kweinberg@chla.usc.edu.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal