Abstract
To evaluate the prognostic significance of quantitative PML-RARA, AML1-ETO, and CBFB-MYH11 fusion transcript expression, real-time polymerase chain reaction was used to analyze bone marrow samples of 349 such patients at diagnosis and 522 samples of 142 patients also during therapy (total analyses, n = 859; median number of follow-up samples, 4/patient; median duration of assessment, 12 months). Lower expression levels at diagnosis correlated with better overall and event-free survival in all 3 leukemia subtypes. By combining the median expression ratio after consolidation therapy and the 75th percentile of the expression ratio at diagnosis, a new score was established that separates a group with 100% EFS from a significantly worse group (P < .0001) in each of the 3 acute myeloid leukemia subgroups. Eight patients showed increasing levels of expression during follow-up and all had relapse. In conclusion, patients at high risk for treatment failure can be identified by high levels of fusion gene expression at diagnosis or less than 3 logs of tumor reduction during the first 3 to 4 months of therapy. By combining the transcription ratios at these 2 checkpoints, a new powerful prognostic score has been established.
Introduction
The 3 most common reciprocal rearrangements in acute myeloid leukemia (AML), t(15;17), inv(16)/t(16;16), and t(8;21), occur in about 20% of all AML cases and are associated with a favorable prognosis.1,2 In these entities most patients achieve a complete remission (CR); however, 10% to 30% finally have a relapse.1,3-10 The corresponding leukemia-specific fusion transcripts, PML-RARA, CBFB-MYH11, and AML1-ETO, can be targeted by polymerase chain reaction (PCR)–based methods at diagnosis and to detect minimal residual disease (MRD).11-13 Thus, the presence of residual PML-RARA+ cells strongly predicts relapse and therefore is an important parameter for treatment decisions.6,14-20 In contrast, the prognostic impact of the mere detection of residual CBFB-MYH11 or AML1-ETO transcripts in CR has not been clarified yet.21-27
Today, the quantitative real-time PCR method allows the quantification of fusion transcript levels before, during, and after therapy.28-36 In chronic myeloid leukemia and acute lymphoblastic leukemia (ALL), quantification of MRD has been useful in predicting clinical outcome,31,37-39 although only limited data have been reported in AML.16,29,32,40-43 Most previous studies were focused on the validation of longitudinal monitoring MRD during the clinical course of the disease by real-time reverse transcription PCR (RT-PCR). In a limited number of cases a molecular relapse was demonstrated to precede the hematologic relapse by some weeks. In addition, it has been speculated that the transcription levels at diagnosis may be predictive for outcome in these AML cases.30 In a recent pivotal study on CBFB-MYH11+ AML, a transcription level above 0.25% CBFB-MYH11 transcripts as related to 100% cABL has been defined to be predictive of relapse.42 Overall, the available reports evaluating the AML-specific fusion genes for prognostic purposes are hampered by limited numbers of cases. In particular, in these prognostically favorable AML subtypes in which relapses occur infrequently, a large cohort of patients is needed to reliably define the impact of PCR-based prognostic markers. We designed primers and fluorescence hybridization probes necessary to apply LightCycler real-time PCR for the quantitative and sensitive detection of AML1-ETO, 2 different PML-RARA, and 6 different CBFB-MYH11 fusion transcripts and performed a total of 859 analyses in 349 patients with AML. Three major topics were addressed: (1) range of transcription levels at diagnosis, (2) kinetics of transcription reduction during therapy, and (3) early detection of molecular relapse. We were able to demonstrate that a new prognostic score can be defined by combining the transcription ratio at diagnosis with that after consolidation therapy.
Patients, materials, and methods
Patient samples
This study is based on 349 patients with CBFB-MYH11+, AML1-ETO+, or PML-RARA+ AML referred to our laboratory from July 1997 to December 2002 for reference diagnostics of AML. PCR analyses were performed in cases with inv(16), t(8;21), t(15;17), or a variant of any of these aberrations identified by karyotyping and in cases with AML M4eo, AML M2 with typical t(8;21) morphology,44 or AML M3/M3v identified by cytomorphology. Of the analyzed patients with AML1-ETO or CBFB-MYH11, 70% were enrolled in the protocols of the German AML Cooperative Group (AMLCG), and of the patients with M3/M3v, 63% were enrolled in the acute promyelocytic leukemia (APL) study of the AMLCG.14 All others were treated by similar regimens. After determination of the fusion types at diagnosis, a real-time PCR optimized for the distinct fusion type was performed on follow-up samples of the respective patients. Samples from 1997-1999 had been stored in RLT-buffer (Qiagen, Hilden, Germany) at –80°C and were analyzed retrospectively. Only bone marrow samples were considered for the presented analyses. All samples drawn after January 2000 were processed directly.
The studies adhered to the Declaration of Helsinki and were approved by the ethics committees of the participating institutions prior to the studies' initiation. Each patient provided informed consent.
Treatment protocol of the German AMLCG study
Treatment comprised the TAD9/HAM (thioguanine-cytosine arabinosidedaunorubicin/high-dose cytosine arabinoside-mitoxantrone [AMLCG92]) and the randomized comparisons of TAD9/HAM versus HAM/HAM (AMLCG99) double-induction therapy followed by TAD9 consolidation.45-47 Patients in CR after TAD9 consolidation were subsequently randomized for monthly maintenance therapy or sequential HAM (S-HAM) as a second course of consolidation in AMLCG92 and for maintenance or autologous stem cell transplantation in AMLCG99.48 Patients with APL were treated with TAD/HAM plus all-trans-retinoic acid (ATRA) followed by TAD9 consolidation and maintenance according to the APL protocol of the AMLCG.14
Cytogenetic and FISH analysis
Cytogenetic analyses were performed using standard techniques.49 For fluorescence in situ hybridization (FISH), commercially available AML1-ETO, CBFB, and PML-RARA probes were used according to the manufacturer's instructions (Vysis, Bergisch Gladbach, Germany). In addition, yeast artificial chromosome (YAC) 854E2 containing the MYH11 gene (Centre D′Etude du Polymorphisme Study Center, Paris, France) was hybridized as described.50
Sample preparation
Mononucleated bone marrow cells were obtained by Ficoll Hypaque density gradient centrifugation. Total RNA was extracted from 107 cells with RNeasy (Qiagen; 1997-2000) or mRNA with the MagnaPureLC mRNA Kit I (Roche Diagnostics, Mannheim, Germany; since January 2001). The cDNA-synthesis of 1 to 2 μg total RNA or mRNA from an equivalent of 107 cells was performed using 300 U Superscript II (Gibco BRL/Invitrogen, Karlsruhe, Germany) and random hexamer primers (Pharmacia, Freiburg, Germany).
PCRs at diagnosis
PCRs for AML1-ETO, CBFB-MYH11, or PML-RARA fusion transcripts were performed as previously described.13,51 For each sample a cABL-specific RT-PCR was performed to control the integrity of RNA using primers abl5′: 5′-GGCCAGTAGCATCTGACTTTG-3′ and abl3′ 5′-ATGGTACCAGGAGTGTTTCTCC-3′. Strict precautions were taken to prevent contamination. Water instead of cDNA was included as a blank sample in each experiment. Amplification products were analyzed on 1.5% agarose gels stained with ethidium bromide.
Quantification of the fusion transcripts by real-time PCR
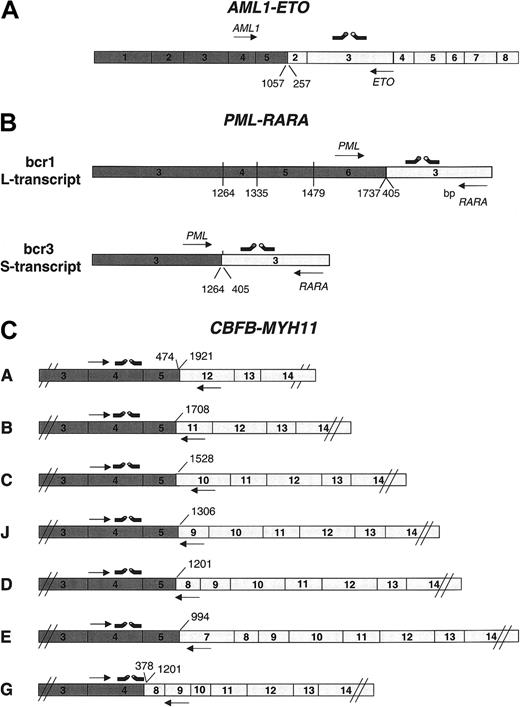
For quantification of the respective fusion gene in the individual sample, real-time PCR using the LightCycler technology was performed (Roche Diagnostics). All primers and hybridization probes used for this study are given in Table 1 and Figure 1. For AML1-ETO the forward primer was positioned to AML1 exon 4, whereas the reverse primer and the hybridization probes were positioned to exon 3 of the ETO gene (Figure 1A). PML-RARA transcript quantification was performed with forward primers in PML exon 6 for bcr1 (L-transcript) or in exon 3 for bcr3 (S-transcript) and reverse primers and the hybridization probes in RARA exon 3 (Figure 1B). Patients with bcr2 breakpoints were excluded from this study. To obtain maximal sensitivity for detection of all different CBFBMYH11 fusion transcripts, primer/probe sets were designed that are potentially able to detect all known fusion transcripts. For all CBFB-MYH11–specific sets, the forward primers, the 5′FL, and the 3′LCred probe were positioned in exon 4 of the CBFB gene. Only the reverse primers were variably positioned to MYH11 exons 7, 8, 9, 10, 11, and 12 to produce PCR fragments of similar sizes and to obtain similar PCR sensitivities for each fusion transcript type (Figure 1C). Running conditions for the respective detection sets were as summarized in Table 2. Each quantitative RT-PCR (Q-RT-PCR) was carried out in a 20-μL reaction volume with each 0.5 μM of forward and reverse primer, 0.25 μM Hyb-Probes, 4 mM MgCl2, and 2 μL LightCycler-FastStart DNA Master Hybridization Probes (Roche Diagnostics). LightCycler data were analyzed using the LightCycler 3.0 software and the second derivative maximum method. Each 20-μL reaction contains 2 μL cDNA, an equivalent of about 30 000 cells at diagnosis and of 200 000 cells of follow-up samples.
Primer and probe designs. Schematic presentation of primer and hybridization probe design for quantification of (A) AML1-ETO, (B) PML-RARA, and (C) CBFB-MYH11 fusion transcript types. Arrows to the right are forward primers; arrows to the left are reverse primers.
Primer and probe designs. Schematic presentation of primer and hybridization probe design for quantification of (A) AML1-ETO, (B) PML-RARA, and (C) CBFB-MYH11 fusion transcript types. Arrows to the right are forward primers; arrows to the left are reverse primers.
Quantification was done with the LightCycler relative quantification software 1.0 (Roche Diagnostics). A standard curve was produced for every target doing 10-fold dilution series of 5 different plasmid concentrations. The standard curve was saved in a standard-curve file. In all following runs a reference dilution was analyzed and the standard curve loaded over this reference sample.
To assess the quality and quantity of the isolated RNA as well as the efficiency of cDNA synthesis, each sample was normalized against the expression of cABL.33 ABL was chosen as a housekeeping gene because it was previously shown to be the most adequate for quantitative analysis in AML.52 A relative quantification was performed by calculating the ratios of the target fusion gene using a plasmid dilution containing the respective fusion target as a standard curve in relation to the housekeeping gene. For simplicity the ratios were multiplied with factor 100. Thus, all expression ratios are given as: 100 × target fusion gene/cABL.
All samples that turned out to be cABL negative or had a cABL value of less than 1 fg/2 μL cDNA (< 1.3 × 104ABL copies), isolated from 2 × 105 cells, were excluded from subsequent analysis.
Reproducibility and accuracy of assays
Positive and negative controls were included in all assays. In particular, RNA from any diagnostic sample was used as a positive control. Negative controls included reactions with no cDNA. Precautions that were taken to avoid contamination included the use of a UV cabinet (Appligene Oncor, Heidelberg, Germany) and PCR-designated pipettes with filtered tips. All tests were conducted at least in duplicate to confirm the results.
Efficiencies, reproducibility, and sensitivities of the PCR assays
The sensitivity and reproducibility of the PCR assays were assessed in triplicate by performing limited dilution series of respective plasmid dilutions and RNA from diagnostic patient samples with near to median transcription ratios in RNA of Jurkat cells that are negative for all the tested fusion transcripts. PCR efficiencies were calculated from the slope of the curves produced from the dilution samples (PCR efficiency = 10–1/slope; Table 3).
Clinical end points and statistical analysis
CR was defined as less than 5% blasts in a normocellular bone marrow with peripheral neutrophil counts more than 1500/μL and platelet counts more than 100 000/μL. Overall survival (OS) was calculated from the first day of therapy to death and event-free survival (EFS) to nonresponse, relapse, or death using Kaplan-Meier estimates.53 Survival curves were compared using a 2-sided log-rank test. Results with a P value of less than .5 were considered significant. The Pearson χ2 statistic and Student t test were used to test for differences in distribution of dichotomous variables and in the mean values of continuous distributions. The normalized expression ratios at diagnosis were correlated to EFS and OS using Cox regression analysis with the initial expression ratio as a covariate and by the Spearman rank correlation. Time points of increasing expression level during follow-up until clinical relapse were analyzed by the Spearman rank correlation. All analyses were performed using the SPSS 11.0.1 software package (SPSS, Chicago, IL).
Results
PCR assays
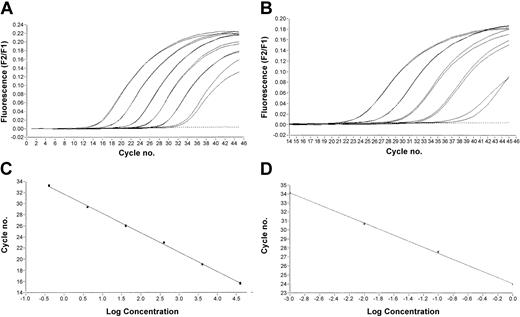
All PCR assays had a dynamic range of at least 6 orders of magnitude. Sensitivities as estimated by limited dilution assays of patient RNA in RNA of Jurkat cell lines were between 10 000 and 100 000 for all assays. The PCR efficiencies were comparable when using plasmids in comparison to patient samples as templates (Table 3 and one example in Figure 2).
CBFB-MYH11 dilution series. (A) Dilution series of CBFB-MYH11 plasmid of fusion type A and (C) the standard curve produced from it. (B) In analogy, a dilution of a patient sample with CBFB-MYH11 with the first 4 samples defined as standard showing nearly the same PCR efficiency (D). All dilutions were done in duplicate.
CBFB-MYH11 dilution series. (A) Dilution series of CBFB-MYH11 plasmid of fusion type A and (C) the standard curve produced from it. (B) In analogy, a dilution of a patient sample with CBFB-MYH11 with the first 4 samples defined as standard showing nearly the same PCR efficiency (D). All dilutions were done in duplicate.
Quality of the test samples
A cDNA equivalent of 2 × 105 cells was included in each PCR assay performed in duplicate. There was a variation between 1.3 × 104 to 3.1 × 107ABL copies per analysis with a median of 6.9 × 105ABL copies. These variations were most likely due to differences in the quality of the cell samples caused by different aspiration quality, stabilization, shipment (1 hour to 3 days), and seasonal variations. The assay for ABL quantification had a log linear range of 7 orders of magnitude. In addition, for all 3 AML subtypes ABL values were analyzed for the fusion gene/ABL ratio. No significances for a positive correlation were found (AML1-ETO: R2 = 0.003, P = .336; CBFBMYH11: R2 = 0.004, P = .306; PML-RARA: R2 = 0.004, P = .357), further confirming that the ABL value has no influence on the ratio.
Patients
In total, 349 patients were analyzed at diagnosis and showed PML-RARA (n = 121), AML1-ETO (n = 106), or CBFB-MYH11 fusion transcripts (n = 122; Table 4). In detail, we were able to detect CBFB-MYH11 fusion type A in 101 cases (82.8%) and rare fusion types in 21 cases (17.2%; type C: n = 1; type D: n = 14; type E: n = 5; type G: n = 1). Of the 121 samples with PMLRARA, 56 (46.2%) cases had bcr1 (L-transcript) and 65 (53.8%) cases had bcr3 fusion types (S-transcript).
The median patient age was 47.5 years (range, 19.2-82.2 years) for PML-RARA+, 51.8 years (range, 15.8-89.1 years) for AML1-ETO+, and 48.9 years (range, 19.0-77.8 years) for CBFB-MYH11+ AML. This study also included patients with therapy-related leukemia: 1 with PML-RARA, 7 with AML1-ETO, and 13 with CBFB-MYH11.
Transcript ratios at diagnosis
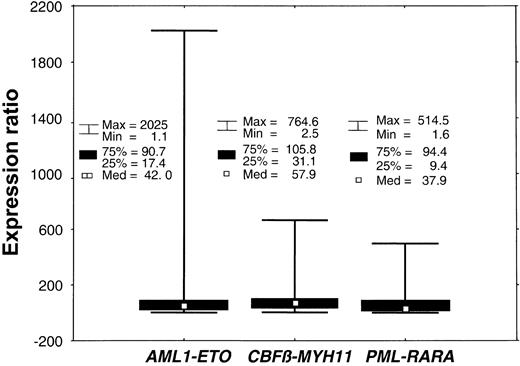
The expression ratios measured in 349 samples at diagnosis varied substantially in all subtypes (Table 5; Figure 3). With a range of 1.12 to 2025 these variations were most prominent in AML1-ETO+ cases. The percentage of blast cells within the bone marrow amounted to 15% to 90% (median, 70%). The percentage of cells type I and II within the bone marrow carrying the respective fusion genes as determined by FISH ranged from 52% to 99% (median, 90%). Thus, these latter variances cannot account for the high variation of expression levels, which can only be explained by high differences in expression activity of the malignant cells between individual patients.
Prognostic impact of transcript ratios at diagnosis
Data on clinical outcome were available in 269 of the 349 analyzed patients (77%). The median follow-up was 457 days for AML1-ETO, 531 days for CBFB-MYH11, and 323 for PML-RARA patients, respectively. The expression ratios of fusion genes (normalized to cABL as control gene) were used as a continuous variable in Cox regression analysis and in all 3 AML subtypes had a significant impact on OS as well as on EFS (Table 6). In contrast, the initial ratio had no impact on CR rates.
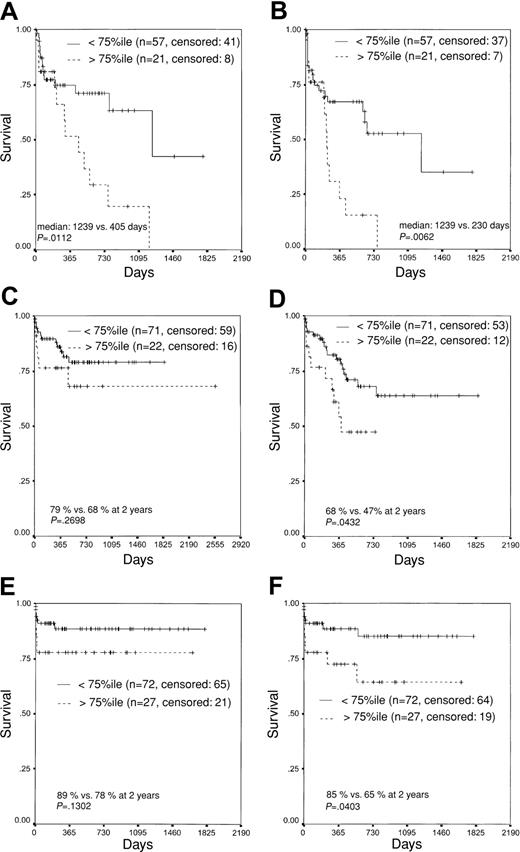
To further analyze the prognostic impact of the transcript ratios at diagnosis on EFS and OS, patients were separated into 2 groups, respectively, based on both the median (data not shown) and the 75th percentile of the initial transcript ratio. In all AML subtypes this resulted in significant differences with regard to both end points (Figure 4A-F).
Plots for OS and EFS. Kaplan-Meier plots for OS (A,C,E) and EFS (B,D,F) in patients with AML1-ETO (A-B), CBFB-MYH11 (C-D), and PML-RARA (E-F) fusion transcripts show that 2 prognostically different groups can be defined by a threshold of more (worse group) or less (better group) than the 75th percentile of the mean initial transcription ratio.
Plots for OS and EFS. Kaplan-Meier plots for OS (A,C,E) and EFS (B,D,F) in patients with AML1-ETO (A-B), CBFB-MYH11 (C-D), and PML-RARA (E-F) fusion transcripts show that 2 prognostically different groups can be defined by a threshold of more (worse group) or less (better group) than the 75th percentile of the mean initial transcription ratio.
Different fusion types in CBFB-MYH11+ and PML-RARA+ AML
Within the group of patients with inv(16) there was no impact of the type of fusion transcript (type A versus rare fusion types) on OS, but the transcript type had an unfavorable impact on EFS. Patients with PML-RARA had a slightly worse outcome in the presence of bcr3 as compared to those with PML-RARA and bcr1 (P = .057 for OS; P = .037 for EFS). The expression ratio was higher in cases with bcr3 (mean, 92.06 versus 40.5). Furthermore, the presence of bcr3 was significantly correlated (P = .002) with a diagnosis of AML M3v (Table 7). Accordingly, OS (P = .037) and EFS (P = .050) were slightly worse in patients with AML M3v as compared to those with AML M3.
Multiparameter analysis
Further parameters (age, blast percentage, absolute blast percentage, leukocyte counts, platelet counts, FISH positivity, secondary etiology, M3/M3v, fusion type) were assessed for correlations with OS and EFS (Tables 8, 9, 10). To prove the independent prognostic impact of the transcript ratio at diagnosis, multivariate analyses were performed within each of the respective AML subgroups. The results demonstrate that in all AML subgroups the initial transcript ratio was the only independent parameter (Table 11).
Transcription reduction during therapy and its impact on prognosis
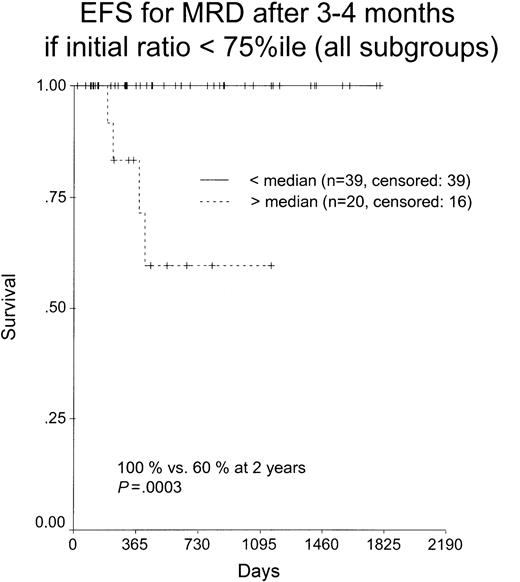
In AML1-ETO+, CBFB-MYH11+, and PML-RARA+ cases a total of 211, 303, and 345 samples, respectively, were analyzed during follow-up (median/range, 4/3-17, 4/3-15, and 5/3-19 samples per patient, respectively) at various time points (range, 3-63 months; Table 4). The kinetics of reduction of the transcript ratio after the first and second induction therapy as well as after the consolidation treatment had no impact on OS or EFS (Table 12). In the subgroup of patients with transcription ratios at diagnosis of less than the 75th percentile (n = 59), the transcript level after consolidation treatment had impact on EFS and OS (Figure 5). Due to limited case numbers at this time point, for this analysis all 3 groups were pooled. Thus, cases with an MRD level higher than the median level at this time point had an inferior outcome (P = .0003). Calculation of the hazard ratio revealed a 27.02-fold risk for relapse in these patients (P = .002).
Transcript levels after consolidation treatment. Kaplan-Meier plot for patients with ratios at less than the 75th percentile where a follow-up at 3 to 4 months (after consolidation therapy) was available.
Transcript levels after consolidation treatment. Kaplan-Meier plot for patients with ratios at less than the 75th percentile where a follow-up at 3 to 4 months (after consolidation therapy) was available.
New score for risk stratification of AML
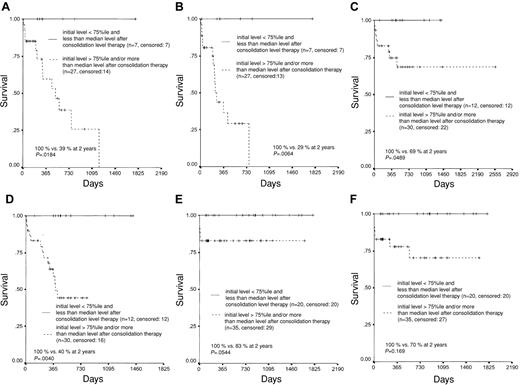
To combine the prognostic power of transcription rates at diagnosis and of MRD level after consolidation treatment, 131 patients (34 with AML1-ETO, 42 with CBFB-MYH11, and 55 with PML-RARA) in which either the transcript levels at diagnosis were more than the 75th percentile (n = 72, ie, 26.8% of 269 patients with clinical follow-up data) or in which an MRD assessment has been performed at the postconsolidation checkpoint and clinical follow-up data were available (n = 59) were evaluated. In all 3 AML subtypes a good risk group was identified by a transcription level at diagnosis of less than the 75th percentile and an MRD level of less than the median in which no case with treatment failure was observed. In contrast, patients with either a higher transcription level at diagnosis or a higher MRD level after consolidation therapy did significantly worse (Figure 6A-F). The median transcription ratio at that point was 0.003, 0.014, and 0.000, respectively.
Prognostic scores for OS and EFS. Kaplan-Meier plots depicting a prognostic score for OS (A,C,E) and EFS (B,D,F) in patients with AML1-ETO (A-B), CBFB-MYH11 (C-D), and PML-RARA (E-F) fusion transcripts considering the 75th percentile of the mean initial transcription ratio and the median transcription ratio after 3 to 4 months as thresholds. Two prognostically different groups can be defined with 100% survival for the better group.
Prognostic scores for OS and EFS. Kaplan-Meier plots depicting a prognostic score for OS (A,C,E) and EFS (B,D,F) in patients with AML1-ETO (A-B), CBFB-MYH11 (C-D), and PML-RARA (E-F) fusion transcripts considering the 75th percentile of the mean initial transcription ratio and the median transcription ratio after 3 to 4 months as thresholds. Two prognostically different groups can be defined with 100% survival for the better group.
This score resulted in even greater differences when AML1-ETO and CBFB-MYH11 cases were pooled together as CBF leukemias (Figure 7A-B) and when also the PML-RARA cases were added (Figure 7C-D).
New prognostic score. New prognostic score considering the 75th percentile of the mean initial transcription ratio and the mean transcription ratio after 3 to 4 months as thresholds for and the CBF leukemias (A-B) and all 3 groups pooled together (C-D).
New prognostic score. New prognostic score considering the 75th percentile of the mean initial transcription ratio and the mean transcription ratio after 3 to 4 months as thresholds for and the CBF leukemias (A-B) and all 3 groups pooled together (C-D).
For statistical analysis it was not possible to build up a third group with high initial levels and low MRD after 3 to 4 months because only 8 cases with this combination were available (1 with AML1-ETO, 1 with CBFB-MYH11, and 6 with PML-RARA) in none of which has an event yet been observed.
Early detection of relapses
For early detection of relapse we were able to analyze a subset of 142 patients at 3 or more different time points during and after therapy. Fifteen of these cases (10.6%) had a relapse. In 8 patients increasing transcript ratios indicated molecular relapses 1 to 5 months before hematologic and cytogenetic relapses occurred. Using a χ2 correlation it was shown that an increase even from the undetectable to a marginal transcription level significantly predicts relapse (P < .0001; Table 13).
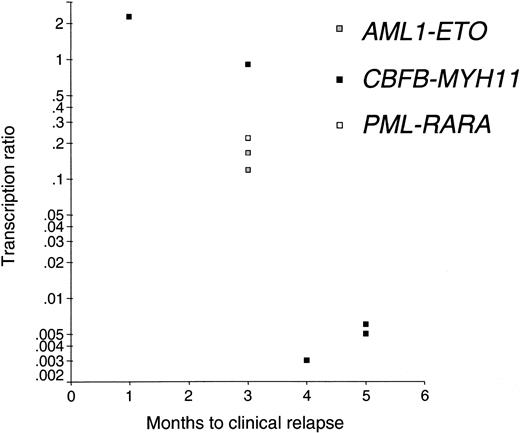
Stressing the importance of small follow-up intervals, in all 7 relapsing patients with follow-up intervals longer than 6 months, relapses were not detected beforehand by PCR. As in all 8 cases with follow-up intervals of 3 months or less a clinical relapse was predictable by increasing PCR levels. Thus, follow-up sampling at least every 3 months is mandatory to predict relapses at the molecular basis. In addition, it could be shown that higher levels at the time of increase during follow-up are correlated with a more rapid manifestation of the clinical relapse (Spearman correlation P = .007; Figure 8).
Early detection of relapses. The higher the transcription ratio after previous negativity the faster the relapse; y-axis, transcription ratio after previous PCR negativity; x-axis, time from first PCR-positivity after negativity to clinical relapse. Spearman rank correlation, P = .007.
Early detection of relapses. The higher the transcription ratio after previous negativity the faster the relapse; y-axis, transcription ratio after previous PCR negativity; x-axis, time from first PCR-positivity after negativity to clinical relapse. Spearman rank correlation, P = .007.
Discussion
AMLs with t(15;17), inv(16)/t(16;16), and t(8;21) are associated with a favorable prognosis.1,2,14 However, 10% to 30% of these patients are prone to treatment failure.1,6-10 Early identification of high-risk patients is important to adjust treatment strategies.6,9 We developed quantitative and highly sensitive real-time RT-PCR assays to detect and to monitor the above-mentioned fusion transcript types.
The present study for the first time showed in large numbers of AML1-ETO+, CBFB-MYH11+, and PML-RARA+ patients that transcription levels at diagnosis are strongly correlated with clinical outcome. Furthermore, a prognostic score was established by combining the transcript ratio at diagnosis and MRD level after consolidation therapy after 3 to 4 months of start of therapy. Thus, quantitative PCR can accurately identify prognostically unfavorable subgroups in these overall relatively favorable AML subgroups.
Varying transcription ratios at diagnosis already have been reported for AML1-ETO and CBFB-MYH11, but without demonstrating a prognostic impact.40,41 However, it was suggested that high transcript rates in CBFB-MYH11+ AML correlate with high blast count and shorter survival, although only 14 patients had been analyzed.30 Thus, the present study is the first one to demonstrate a significant impact of the initial transcript levels on OS and EFS in a large cohort of patients of all 3 AML subtypes analyzed. It has to be stressed that in the Cox regression model OS is significantly related to the transcript levels at diagnosis as continuous variables in all 3 analyzed AML subtypes, whereas the separation of the patients into 2 groups, respectively, resulted in significant differences in the OS in the AML1-ETO group only but not in the CBFB-MYH11 and PML-RARAgroups. This is most probably due to effective salvage therapies in the latter 2 groups in particular because the differences in EFS were greater than in OS and significant in all subgroups. In multivariate analysis the transcript levels were shown to be the only significant parameter for OS and EFS. A further large study,54 which included an analysis of PML-RARA ratios at diagnosis with respect to outcome, did not find an impact of the initial transcription ratio on prognosis. It has to be pointed out that these controversial findings may be based on different treatment strategies or the use of different housekeeping genes (ABL versus GAPDH) that may be differently expressed and may have an influence on the ratio. In a previous preclinical study using different leukemias, healthy blood, and sorted bone marrow cells, we have shown that ABL analyzed versus porphobilinogen deaminase (PBGD), β2-microglobulin, and glucose-6-phosphate dehydrogenase (G6PDH) was the most stably expressed gene in hematopoietic cells, which was the basis for selecting ABL in the present study.52 Response rates were not affected by initial transcript levels due to the overall high CR rates in these 3 subtypes of AML. The only other parameter carrying prognostic importance was the kind of transcript in PML-RARA and CBFB-MYH11. Like in the Intergroup 0129 study54 in PML-RARA cases bcr3 was correlated with a higher initial ratio than bcr1 at diagnosis. In addition bcr3 was associated with a worse prognosis than bcr1. However, we could show that this association was not independent but rather reflected the correlation between M3v and high leukocyte counts. Similarly, in M4eo the rare CBFBMYH11 fusion type was correlated to secondary etiology of the disease but not independently to a worse prognosis.
In contrast to childhood ALL where the levels of MRD at 5 weeks after induction therapy39 and even at day 1538 were highly correlated with risk of relapse, there is no evidence that MRD levels at early time points after first and second induction therapy are correlated with outcome in AML1-ETO+, CBFB-MYH11+, and PML-RARA+ AML patients. Interestingly, whereas in AML, in general, early response to treatment as assessed by cytomorphology is of major prognostic impact this is not true forAML with AML1-ETO and CBFB-MYH11.10,55 This is in line with data of the present study and may indicate that early assessment of MRD in these AML subtypes may not have prognostic impact due to the overall very good response to therapy and to a rather slow decline of the leukemia cell mass. This is in accordance with a recent paper that shows the significance of transcription levels at later time points in patients who achieved CR in CBFB-MYH11+ AML.43 After consolidation therapy, which was also the second checkpoint in an MRD study on PML-RARA,54 the unfavorable subgroup had a 4.1-fold risk for relapse compared with a 27-fold risk for relapse in our mixed cohort, suggesting that this is the most important time point for MRD monitoring in these 3 AML subgroups. In this PML-RARA study the cutoff for the higher risk group was MRD 10–5. In recent studies on inv(16)/CBFB-MYH11+ AML critical values in follow-up samples indicative for high-risk patients were suggested. Whereas in one study 10 CBFB-MYH11 copies per PCR after completion of chemotherapy were suggested,30 others suggested a critical transcript ratio of 0.25% at any time in remission to be predictive for relapse.42 A similar study described a threshold of 1% after induction and 0.1% after consolidation to identify patients at high risk of relapse.43 The cutoff levels in the present study that identify those high-risk cases that have not already been recognized by initial high ratios were 0.003% for AML1-ETO, 0.014% for CBFB-MYH11, and greater than 0.000 for PML-RARA. In addition, in the presented study ratios even lower than 0.1% have been detected in 3 patients (0.036%, 0.006%, 0.005%) after previous negativity in good-quality samples; these patients all subsequently had a relapse within 3, 5, and 5 months, respectively. Therefore, the limit of the previously suggested values may be too insensitive. In addition, due to the high interpatient variability of transcript ratios at diagnosis, which may persist in individual cells in remission, general cut-off levels may not be appropriate. We did not find interspersed positive samples in CCR samples as did others,54 which might be due to different sensitivity levels of the assays used in the different studies. By using fusion gene-specific cDNA priming the sensitivity can be extended compared with random priming of cDNA synthesis, but this may have a disadvantageous effect on the predictive relevance of these values. Due to the different treatment strategies, different AML cohorts, and methodologies for quantitative monitoring, it is not yet possible to compare critical cutoff levels given in different recent studies, that is, to establish definite levels useful for overall therapeutic suggestions. Based on our data it may be more informative to monitor the kinetics in single patients at least every 3 months and use an increase of transcript levels as predictor for relapse as has previously been suggested for PML-RARA.20 Furthermore, follow-up assessments every 3 months are indicated because in all patients monitored at 3-month intervals an increasing MRD level was detected before the occurrence of clinical relapse. It is our hope that, in the future, follow-up assessment may be done more easily and frequently using peripheral blood. However, although it has been shown that peripheral blood may be used for follow-up studies in ETV6-AML1+ ALL,56 little and controversial data have been reported for peripheral blood samples in AML.22,54 So far, monitoring of MRD in peripheral blood cannot be recommended in general for patients with AML.22 However, data reported by Gallagher et al54 indicate comparable sensitivities for the analysis of peripheral blood and bone marrow in APL. Still, further investigations are necessary to clarify this issue.
By combining one checkpoint at diagnosis with a second checkpoint after consolidation therapy, in the present study for the first time a score based on quantitative PCR was established that identifies some of the poor-risk patients by the initial transcription ratio and some by the MRD level at checkpoint 2. It is obvious that in the present data set there are differences in the EFS between the 3 AML subgroups with AML1-ETO cases having the worst overall outcome. However, despite these differences the application of the newly defined prognostic score revealed a significant prognostic impact with regard to EFS within all of these 3 subtypes. Therefore, combined data have also been evaluated and the score has been shown to be highly predictive of the outcome. In addition, there are differences between the subgroups with regard to the time of treatment failure. In most PML-RARA cases failures occurred before checkpoint 2, that is, after consolidation. However, because the score is composed from both initial transcription ratios and MRD levels at checkpoint 2, the identification even of these early failure cases can be accomplished by the score.
In summary, the present study revealed the following important aspects: (1) fusion transcript levels at diagnosis have significant impact on OS and EFS but not on CR rate in AML with AML1-ETO, CBFB-MYH11, and PML-RARA fusion transcripts; (2) the 75th percentile of the initial transcript ratio can be defined as a threshold that separates 2 prognostically different groups; (3) combining this parameter with the median MRD level after consolidation therapy can help define a group with a most favorable EFS. These data indicate that AML-specific fusion transcript levels can be assessed validly, specifi-cally, and sensitively by quantitative real-time PCR. Thus, this technique allows the identification of poor-risk patients within this otherwise prognostically favorable subgroup of AML. The new score that is based on quantification of fusion transcripts at 2 different time points and that was established from a retrospective analysis has to be validated in prospective studies.
These data may serve as the basis for future studies to evaluate alternative treatment strategies in patients within the high-risk groups who might benefit from early therapeutic interventions.
Prepublished online as Blood First Edition Paper, July 3, 2003; DOI 10.1182/blood-2003-03-0880.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.
We greatly acknowledge all of more than 300 physicians for their confidence in our laboratory, for sending the patient samples, and for providing us with information on the clinical courses of the patients. We thank Jürgen Krauter for providing CBFB-MYH11 and AML1-ETO plasmids and Andreas Hochhaus for providing an ABL plasmid. Olfert Landt (TIBMOLBIOL, Berlin, Germany) is acknowledged for help with designing LightCycler hybridization probes. In addition, we thank Gudrun Mellert, Sabine Koneberg, Claudia Tschulik, Nina Herrmann, Tanja Skorupinski, Karin Hecht, and Rita Lapping for their excellent technical assistance.