Abstract
Presence of internal tandem duplication in the FMS-like tyrosine kinase 3 gene(s) (FLT3-ITD) is associated with poor outcome among patients with cytogenetically normal acute myelogenous leukemia (CN-AML). Patients carrying a higher FLT3-ITD allelic burden have worse outcome. Allogeneic stem cell transplantation (SCT) in first remission (CR1) is reported to improve survival in this poor risk patients. We investigated the impact of FLT3-ITD allelic burden, number of FLT3-ITD mutations and SCT with a matched related or unrelated donor in CR1 in newly diagnosed FLT3-ITD mutated intermediate cytogenetic risk AML patients diagnosed between July 2000 and November 2013, who achieved CR1 with induction therapy. In order to reduce the selection bias, only patients who were alive and free of AML for at least 4 months were included in the analysis (median time to SCT was 4.5 months).
The study group had 169 patients and 48 of 169 had SCT in CR1. Patients in the transplant group were younger than those in the non-transplant group (median age 55 v 62 years, p=0.001). As presented in Table1, there was no significant difference in the distribution of gender, white blood cell and platelet counts, bone marrow (BM) blast number, the proportion with CN, the level of FLT3-ITD allele level and number of FLT3-ITD mutations at diagnosis between the two groups. The median follow-up of survivors in the transplant and no transplant groups were similar (30.7 vs. 34.5 months).
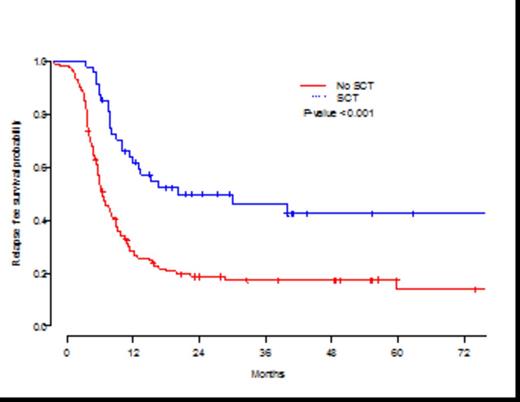
Overall, SCT in CR1 was associated with longer relapse-free survival (RFS) compared to no-transplant approach. The 3-year RFS from CR1 for transplant patients was 46.3% (95% confidence interval (CI),33.3%-64.2%) compared to 17.5% (95% CI,11.5%-26.4%) for non-transplant patients (P<0.001) (Figure 1). This was largely due to a higher cumulative relapse incidence (RI) of 68% in the non-transplant patients compared to 41% who received SCT (P<0.001). Overall survival (OS) from CR1 was also different between the groups (P=0.0003), with 3-year estimates of 53.5% (95% CI,39.6%-72.3%) for transplant patients compared to 24.3% (95% CI, 17.3%-34.2%) for no-transplant patients. The non-relapse mortality rate of 13% observed with SCT was similar to 15% observed in non-transplant patients (P=0.86).
Higher WBC and FLT3-ITD alleleic burden at diagnosis were poor prognostic factors associated with decreased RFS and OS in univariate analyses. Older age decreased OS but not RFS. For multivariate regression, we excluded WBC count at diagnosis from the analysis as high WBC was associated with FLT3-ITD allelic burden at diagnosis. Multivariate regression models showed similar results with SCT in CR1 leading to improved RFS and OS while older age and higher FLT3-ITD allelic burden lost their prognostic significance as shown in Table 2.
Our results indicate that SCT in CR1 AML FLT3-ITDmut patients is associated with a longer RFS, largely due to a reduction in the RR. The main reason of treatment failure remains to be relapse and strategies incorporating FLT3-ITD inhibitors before and after SCT may lead to a significant improvement in RFS of this high risk group.
Patient and disease characteristics
| . | All patients . | Transplant group . | Non-transplant group . | p . |
|---|---|---|---|---|
| Median age, years (IQR) | 59 (50-68) | 55 (47-62) | 62 (51-70) | 0.001 |
| Age <60 | 85 (50.3%) | 30 (62.5%) | 55 (45.5%) | 0.06 |
| Female | 82 (48.5%) | 25 (52.1%) | 57 (47.1%) | 0.6 |
| Diagnosis after 2008 | 106 (62.7%) | 38 (79.2%) | 68 (56.2%) | 0.01 |
| Median WBC at diagnosis (IQR) | 11.6 (3.8-29.7) | 9 (3-40) | 12 (4-28) | 0.98 |
| Median platelets at diagnosis, (IQR) | 45 (25-74) | 45 (22-84) | 45 (25-71) | 0.99 |
| Median BM blast count at diagnosis, (IQR) | 43(12-74) | 32 (6-64) | 46 (13-75) | 0.2 |
| Diagnostic cytogenetics | ||||
| Diploid | 128 (75.7%) | 33 (68.8%) | 95 (78.5%) | |
| Other abnormalities | 36 (21.3%) | 12 (25%) | 24 (19.8%) | 0.4 |
| Unknown | 5 (2.9%) | 3 (6.3%) | 2 (0.2%) | |
| Median FLT3/ITD allelelic burden, (IQR) | 0.34 (0.1-0.47) | 0.30 (0.04-0.47) | 0.35 (0.12-0.49) | 0.3 |
| FLT3/ITD allele ratio <0.3 | 75 (44.4%) | 24 (50%) | 51 (42.1%) | 0.5 |
| Number of FLT3/ID mutations | ||||
| 1 | 130 (76.9%) | 35 (72.9%) | 95 (78.5%) | |
| 2 | 21 (12.4%) | 9 (18.8%) | 12 (9.9%) | |
| 3 | 12 (7.1%) | 3 (6.3%) | 9 (7.4%) | 0.4 |
| Unknown | 6 (3.6%) | 1 (2.1%) | 5 (4.1%) |
| . | All patients . | Transplant group . | Non-transplant group . | p . |
|---|---|---|---|---|
| Median age, years (IQR) | 59 (50-68) | 55 (47-62) | 62 (51-70) | 0.001 |
| Age <60 | 85 (50.3%) | 30 (62.5%) | 55 (45.5%) | 0.06 |
| Female | 82 (48.5%) | 25 (52.1%) | 57 (47.1%) | 0.6 |
| Diagnosis after 2008 | 106 (62.7%) | 38 (79.2%) | 68 (56.2%) | 0.01 |
| Median WBC at diagnosis (IQR) | 11.6 (3.8-29.7) | 9 (3-40) | 12 (4-28) | 0.98 |
| Median platelets at diagnosis, (IQR) | 45 (25-74) | 45 (22-84) | 45 (25-71) | 0.99 |
| Median BM blast count at diagnosis, (IQR) | 43(12-74) | 32 (6-64) | 46 (13-75) | 0.2 |
| Diagnostic cytogenetics | ||||
| Diploid | 128 (75.7%) | 33 (68.8%) | 95 (78.5%) | |
| Other abnormalities | 36 (21.3%) | 12 (25%) | 24 (19.8%) | 0.4 |
| Unknown | 5 (2.9%) | 3 (6.3%) | 2 (0.2%) | |
| Median FLT3/ITD allelelic burden, (IQR) | 0.34 (0.1-0.47) | 0.30 (0.04-0.47) | 0.35 (0.12-0.49) | 0.3 |
| FLT3/ITD allele ratio <0.3 | 75 (44.4%) | 24 (50%) | 51 (42.1%) | 0.5 |
| Number of FLT3/ID mutations | ||||
| 1 | 130 (76.9%) | 35 (72.9%) | 95 (78.5%) | |
| 2 | 21 (12.4%) | 9 (18.8%) | 12 (9.9%) | |
| 3 | 12 (7.1%) | 3 (6.3%) | 9 (7.4%) | 0.4 |
| Unknown | 6 (3.6%) | 1 (2.1%) | 5 (4.1%) |
Multivariate regression models for RFS and OS
| Variables . | RFS . | OS . | ||
|---|---|---|---|---|
| HR (95%CI) | p | HR (95%CI) | p | |
| Log (FLT3-ITD allele level at diagnosis) | 1.12(0.9-1.3) | 0.14 | 1.13 (0.95-1.33) | 0.16 |
| HSCT | ||||
| No | 1.0 | 1.0 | ||
| Yes | 0.38 (0.24-0.60) | <0.001 | 0.43 (0.26-0.70) | 0.001 |
| Variables . | RFS . | OS . | ||
|---|---|---|---|---|
| HR (95%CI) | p | HR (95%CI) | p | |
| Log (FLT3-ITD allele level at diagnosis) | 1.12(0.9-1.3) | 0.14 | 1.13 (0.95-1.33) | 0.16 |
| HSCT | ||||
| No | 1.0 | 1.0 | ||
| Yes | 0.38 (0.24-0.60) | <0.001 | 0.43 (0.26-0.70) | 0.001 |
Kaplan-Meier estimates of leukemia-free survival by transplant groups
Kantarjian:ARIAD, Pfizer, Amgen: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.