Abstract
Background: AZA is a DNA methyltransferase (DNMT) inhibitor with a modest response rate (20-25%) and duration (Å4 months) in MF. Ruxolitinib and azacytidine may target distinct clinical and pathological manifestations of myelofibrosis.
Aim:To determinethe efficacy and safety of the combination in pts with MF requiring therapy(ClinicalTrials.gov Identifier: NCT01787487).
Methods:A sequential approach with single-agent RUX 15 mg orally twice daily (if platelets 100-200) or 20 mg twice daily (if platelets >200) continuously in 28-day cycles for the first 3 months followed by the addition of AZA 25 mg/m2 on days 1-5 of each 28-day cycle starting cycle 4 was adopted. The AZA dosage could be gradually increased to a maximum of 75 mg/m2. Pts would be treated on study for 15 months followed by continuation of the combination off-study at the discretion of the treating physician.
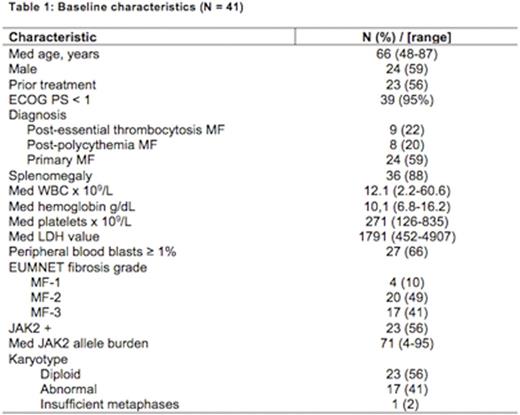
Results: 41 pts were enrolled between March 1, 2013 and June 30, 2016. Baseline characteristics are shown in table 1. 23 (56%) had received a median of 2 (range, 1-3) prior therapies for MF. 27 pts remain alive after a med follow-up of 20.4+ months (range, 0.5-37.3+).
39 pts were enrolled before January 1 2016 and are evaluable for response. International Working Group for Myelofibrosis Research and Treatment 2013 (IWG-MRT)objective responses were noted in 27 (69%), including PR in 2 (5%), CI for spleen and total symptom score (TSS) in 7 (18%), CI for TSS and hemoglobin in 2 (5%), CI for spleen only in 7 (13%), and CI for TSS only in 9 (21%). Ten (26%) pts had no IWG response and 2 (5%) pts had progression to AML on therapy. Three ptshave achieved a complete cytogenetic remission (baseline cytogenetics were +8 in two and del 20q in one). Responses occurred in 14 of 23 (61%) previously treated patients and 13 of 16 (81%) untreated pts (P=0.57). Median time to all responses was 1.0 month (range, 0.7-19.3 months). Median time to CI in spleen size was 1.8 months (range, 0.9 - 16.8), to CI TSS was 1.8 months (range, 0.7-15.8), CI Hb was 5.8 months (range, 1.1-14.7), and cytogenetic remission was 5.5 months (5.4-5.5). 29 pts had a baseline spleen ³5 cm and 14 (48%) achieved >50% palpable spleen length reduction at 24 weeks and 23 (79%) achieved >50% palpable spleen reduction at any time on study. 6/23 (26%) of the >50% palpable spleen reductions occurred after the addition of azacytidine. Among the 29 pts with baseline spleen ³5cm best spleen response included: 50-100% reduction in 23 (79%), 25-49% reduction in 4 (14%), no reduction in 2 (7%).
Serial JAK2 allele burden assessment was available in 15 of 17 JAK2 mutated responders. A reduction in the baseline JAK2V617F allele burden was noted in 13 of 15 (87%) serially evaluable responders with a median V617F allele burden reduction of 21% (range, 1-82%). Serial evaluation of bone marrow fibrosis was available in all 27 responders and revealed a reduction in EUMNET fibrosis score in 11 (41%) responders, including ³2 grade reduction in 2 pts and 1 grade reduction in BM fibrosis in 9pts with a median time to fibrosis improvement of 13.2 months (range, 5.1-13.2).
Three pts experienced grade 3/4 non-hematological toxicity including fatigue (n=2), nausea (n=1), and pneumonia (n=1). New onset grade 3/4 anemia and thrombocytopenia were seen in 25 pts [61%; of which 7 (17%) had a 2+ grade change] and 11 (27%) pts, respectively. The med overall survival is 38.7+ mos.
The AZA was administered in 34 (83%)pts: as planned in cycle 4 in 28pts(63%), earlierdueto increased blasts in 1pt, and later in 5pts[cycle 5 (n=2), cycle 7 (n=2), cycle 9 (n=1)]. Twoptsprogressed before starting AZA and 3 never started AZA due to prohibitive cytopenia. Twoptsare too early to start AZA. Six of 34 (18%)ptswho started AZA have required discontinuation of AZA: low platelets (n=3), fatigue (n=2), and low ANC (n=1).
Twenty fourpts remain on study. Reasons for discontinuation in 17 pts included stem cell transplant (n=6), lack of response (n=3), AML (n=2), toxicity (n=3), death (n=1), patient preference (n=2). Fourteen pts have died: AML (n=4), pneumonia (n=3), unknown cause (n=3), sepsis (n=2), post SCT complications (n=1), and other cancer (n=1).
Conclusion: Concomitant RUX with AZA was feasible and resulted ina IWG-MRT response rate of 69%. The >50% spleen length reduction at 24 weeks and at any time on study were superior to single agent RUX and 26% of the spleen reductions occurred after addition of AZA. This combination warrants further evaluation on a large scale.
Daver:Karyopharm: Honoraria, Research Funding; Otsuka: Consultancy, Honoraria; Ariad: Research Funding; Sunesis: Consultancy, Research Funding; BMS: Research Funding; Kiromic: Research Funding; Pfizer: Consultancy, Research Funding. Cortes:ARIAD: Consultancy, Research Funding; BMS: Consultancy, Research Funding; Novartis: Consultancy, Research Funding; Pfizer: Consultancy, Research Funding; Teva: Research Funding. Jabbour:ARIAD: Consultancy, Research Funding; Pfizer: Consultancy, Research Funding; Novartis: Research Funding; BMS: Consultancy. Verstovsek:Celgene: Research Funding; CTI BioPharma Corp: Research Funding; Galena BioPharma: Research Funding; Seattle Genetics: Research Funding; Roche: Research Funding; Geron: Research Funding; NS Pharma: Research Funding; Bristol-Myers Squibb: Research Funding; Gilead: Research Funding; Lilly Oncology: Research Funding; Promedior: Research Funding; AstraZeneca: Research Funding; Incyte Corporation: Membership on an entity's Board of Directors or advisory committees, Research Funding; Pfizer: Research Funding; Genentech: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.