Abstract
Regulators of apoptosis in acute myeloid leukemia (AML) have been extensively studied and are considered excellent therapeutic targets. Apoptosis repressor with caspase recruitment domain (ARC), an antiapoptotic protein originally found to be involved in apoptosis of cardiac cells, was recently demonstrated to be overexpressed in several solid tumors. To assess its importance in AML, we profiled ARC expression in 511 newly diagnosed AML patients using a validated robust reverse-phase protein array and correlated ARC levels with clinical outcomes. ARC was variably expressed in samples from patients with AML. ARC level was not associated with cytogenetic groups or with FLT-3 mutation status. However, patients with low or medium ARC protein levels had significantly better outcomes than those with high ARC levels: longer overall survival (median, 53.9 or 61.6 vs 38.9 weeks, P = .0015) and longer remission duration (median, 97.6 or 44.7 vs 31.1 weeks, P = .0007). Multivariate analysis indicated that ARC was a statistically significant independent predictor of survival in AML (P = .00013). Inhibition of ARC promoted apoptosis and sensitized cytosine arabinoside-induced apoptosis in OCI-AML3 cells. These results suggest that ARC expression levels are highly prognostic in AML and that ARC is a potential therapeutic target in AML.
Introduction
Chemoresistance, the major obstacle to successful therapy for patients with acute myeloid leukemia (AML), is often attributed to deregulated apoptosis1 of AML cells. Therefore, identifying targets that can be used to induce apoptosis and sensitize leukemic cells to chemotherapy could be therapeutically useful for this disease. Apoptosis repressor with caspase recruitment domain (ARC), an antiapoptotic protein, is present in normal heart,2,3 skeletal muscles,4 and brain tissues5 and protects them from apoptosis. ARC inhibits both the extrinsic and intrinsic apoptosis pathways by inhibiting Fas-FADD binding and assembly of the death-inducing signaling complex, by suppressing the activity of caspase-2 and caspase-8, and by blocking Bax activation.4,6-9 Increased ARC expression was recently demonstrated to occur in various cancer cells,10 including all stages of renal cell carcinomas,6 primary human colon adenocarcinomas,11 and malignant gliomas12 ; ARC levels increase with grade and are correlated with poor prognosis. ARC overexpression confers both chemotherapy and radiation resistance,13,14 consistent with its role as an antiapoptotic protein. ARC was also reported to contribute to chemotherapy resistance by abolishing mitochondrial fission mediated by dynamin-related protein-1.13,14 ARC was reported to exhibit bidirectional interactions with p53: whereas p53 inhibits ARC transcription15 and promotes ARC degradation through the p53-induced ubiquitin E3 ligase MDM2,16 ARC negatively regulates p53 by inhibiting its tetramerization and triggering its cytoplasmic localization.17 Expression of ARC was also found to be regulated by oxidative stress in cancer cells.18 In glioma cells, platelet-derived growth factor receptor inhibition with imatinib led to decreased ARC levels; this effect may play a role in triggering apoptosis and sensitizing cells to the inhibition of inhibitor of apoptosis protein (IAP),12 suggesting that ARC can be targeted therapeutically. ARC expression and function in AML have not been investigated.
Reverse-phase protein array (RPPA) is a robust and reproducible high-throughput proteomics system that can quantitatively determine protein expression levels in large sample sets and requires only small amounts of protein. Our group has validated the RPPA assay and demonstrated that it is a valuable tool for the functional profiling of protein expression in AML.19,20 In an effort to search for new clinically relevant therapeutic targets in AML, we profiled ARC expression levels in samples obtained from 511 newly diagnosed patients with AML using this novel technology,19,20 correlated ARC levels with clinical outcomes, and found a major prognostic impact that was independent of other known prognostic variables in AML.
Methods
Patient population
Peripheral blood (PB) and bone marrow (BM) specimens were collected from 511 patients with newly diagnosed AML and 21 with newly diagnosed acute promyelocytic leukemia (APL) evaluated at the University of Texas M. D. Anderson Cancer Center (MDACC) from September 1999 to July 2010. A total of 387 BM and 283 PB samples were studied from the newly diagnosed patients; for 140 patients, both were available. A paired relapse sample was available for 47 of the AML and one of the APL patients. All but one of the relapse samples were from BM. The outcomes analysis reported here is restricted to the newly diagnosed patients. The demographics and clinical characteristics of the newly diagnosed patients are described in Table 1. This patient population is typical of the MDACC referral pattern: a high percentage of patients with unfavorable cytogenetics (49%) and a very high percentage with an antecedent hematologic disorder (40%). Cancer and Leukemia Group B system was adopted for cytogenetic risk group classification as described.21
Demographics and clinical characteristics of the newly diagnosed AML patients in the study set
| Treated . | Category . | All cases . | All treated cases . | Lowest third . | Middle third . | Highest third . | P . |
|---|---|---|---|---|---|---|---|
| N | 511 | 415 | 147 | 131 | 137 | ||
| Male/female | 291:220 | 218:197 | 73:56 | 71:70 | 75:69 | .79 | |
| Age, y | Minimum | 15.8 | 15.8 | 17.4 | 17.3 | 15.8 | |
| Maximum | 87.23 | 87.4 | 87.4 | 83.4 | 85.8 | .33 | |
| Median | 65.7 | 64.3 | 61.9 | 64.5 | 65.5 | ||
| FAB | M0 | 5.7% | 5.1% | 8.2% | 3.8% | 2.9% | .09 |
| M1 | 10.8% | 11.6% | 10.2% | 16.0% | 8.8% | .14 | |
| M2 | 33.1% | 36.9% | 47.6% | 34.4% | 27.7% | .002 | |
| M4 | 22.7% | 23.4% | 12.2% | 21.4% | 37.2% | .000003 | |
| M5 | 10.0% | 9.9% | 10.2% | 9.2% | 10.2% | .95 | |
| M6 | 5.3% | 5.1% | 4.1% | 7.6% | 3.6% | .24 | |
| M7 | 2.0% | 1.9% | 3.4% | 0.8% | 1.5% | .25 | |
| Unknown | 7.2% | 5.5% | 4.1% | 6.1% | 6.6% | .62 | |
| RAEBT | 3.3% | 0.7% | 0.0% | 0.8% | 1.5% | ||
| Cytogenetics | Favorable | 6.7% | 8.0% | 11.6% | 6.1% | 5.8% | |
| Intermediate | 44.0% | 47.0% | 48.3% | 41.2% | 51.1% | .11 | |
| Unfavorable | 49.3% | 45.1% | 40.1% | 52.7% | 43.1% | ||
| FLT-3 | ITD | 14.9% | 17.8% | 17.7% | 20.6% | 15.3% | .74 |
| D835 | 3.1% | 5.8% | 6.8% | 5.3% | 5.1% | .96 | |
| Both | 1.6% | 1.9% | 2.0% | 1.5% | 2.2% | .8 | |
| Zubrod PS | 3 or 4 | 3.3% | 3.1% | 2.0% | 1.5% | 5.8% | .15 |
| AHD | ≥ 2 months | 39.9% | 37.1% | 32.7% | 38.2% | 40.9% | .39 |
| Infection | Yes | 19.8% | 22.2% | 23.1% | 21.4% | 21.9% | .94 |
| WBCs | Median | 8.8 | 9.9 | 13.3 | 9.9 | 8.8 | .48 |
| Platelets | Median | 56 | 55.5 | 49 | 65 | 56 | .26 |
| Hemoglobin | Median | 9.6 | 9.6 | 9.8 | 9.7 | 9.2 | .23 |
| % BM blast | Median | 46 | 50 | 58 | 45 | 47 | .06 |
| % Blood blast | Median | 18 | 20.5 | 35 | 16 | 12 | .000002 |
| Response | CR | 57.1% | 55.1% | 65.6% | 51.1% | .045 | |
| Resistant | 32.8% | 36.1% | 23.7% | 38.0% | .026 | ||
| Fail | 10.1% | 8.8% | 10.7% | 10.9% | .81 | ||
| Relapse | 61.6% | 45.7% | 67.4% | 72.9% | .001 | ||
| Alive | 25.8% | 34.0% | 28.2% | 14.6% | .001 | ||
| Overall survival, wk (median) | 49.14 | 53.9 | 61.6 | 38.9 | .0015 | ||
| Remission duration, wk (median) | 45.86 | 97.6 | 44.7 | 31.1 | .0007 | ||
| Treated . | Category . | All cases . | All treated cases . | Lowest third . | Middle third . | Highest third . | P . |
|---|---|---|---|---|---|---|---|
| N | 511 | 415 | 147 | 131 | 137 | ||
| Male/female | 291:220 | 218:197 | 73:56 | 71:70 | 75:69 | .79 | |
| Age, y | Minimum | 15.8 | 15.8 | 17.4 | 17.3 | 15.8 | |
| Maximum | 87.23 | 87.4 | 87.4 | 83.4 | 85.8 | .33 | |
| Median | 65.7 | 64.3 | 61.9 | 64.5 | 65.5 | ||
| FAB | M0 | 5.7% | 5.1% | 8.2% | 3.8% | 2.9% | .09 |
| M1 | 10.8% | 11.6% | 10.2% | 16.0% | 8.8% | .14 | |
| M2 | 33.1% | 36.9% | 47.6% | 34.4% | 27.7% | .002 | |
| M4 | 22.7% | 23.4% | 12.2% | 21.4% | 37.2% | .000003 | |
| M5 | 10.0% | 9.9% | 10.2% | 9.2% | 10.2% | .95 | |
| M6 | 5.3% | 5.1% | 4.1% | 7.6% | 3.6% | .24 | |
| M7 | 2.0% | 1.9% | 3.4% | 0.8% | 1.5% | .25 | |
| Unknown | 7.2% | 5.5% | 4.1% | 6.1% | 6.6% | .62 | |
| RAEBT | 3.3% | 0.7% | 0.0% | 0.8% | 1.5% | ||
| Cytogenetics | Favorable | 6.7% | 8.0% | 11.6% | 6.1% | 5.8% | |
| Intermediate | 44.0% | 47.0% | 48.3% | 41.2% | 51.1% | .11 | |
| Unfavorable | 49.3% | 45.1% | 40.1% | 52.7% | 43.1% | ||
| FLT-3 | ITD | 14.9% | 17.8% | 17.7% | 20.6% | 15.3% | .74 |
| D835 | 3.1% | 5.8% | 6.8% | 5.3% | 5.1% | .96 | |
| Both | 1.6% | 1.9% | 2.0% | 1.5% | 2.2% | .8 | |
| Zubrod PS | 3 or 4 | 3.3% | 3.1% | 2.0% | 1.5% | 5.8% | .15 |
| AHD | ≥ 2 months | 39.9% | 37.1% | 32.7% | 38.2% | 40.9% | .39 |
| Infection | Yes | 19.8% | 22.2% | 23.1% | 21.4% | 21.9% | .94 |
| WBCs | Median | 8.8 | 9.9 | 13.3 | 9.9 | 8.8 | .48 |
| Platelets | Median | 56 | 55.5 | 49 | 65 | 56 | .26 |
| Hemoglobin | Median | 9.6 | 9.6 | 9.8 | 9.7 | 9.2 | .23 |
| % BM blast | Median | 46 | 50 | 58 | 45 | 47 | .06 |
| % Blood blast | Median | 18 | 20.5 | 35 | 16 | 12 | .000002 |
| Response | CR | 57.1% | 55.1% | 65.6% | 51.1% | .045 | |
| Resistant | 32.8% | 36.1% | 23.7% | 38.0% | .026 | ||
| Fail | 10.1% | 8.8% | 10.7% | 10.9% | .81 | ||
| Relapse | 61.6% | 45.7% | 67.4% | 72.9% | .001 | ||
| Alive | 25.8% | 34.0% | 28.2% | 14.6% | .001 | ||
| Overall survival, wk (median) | 49.14 | 53.9 | 61.6 | 38.9 | .0015 | ||
| Remission duration, wk (median) | 45.86 | 97.6 | 44.7 | 31.1 | .0007 | ||
RAEBT indicates refractory anemia with excess of blasts in transformation; PS, Performance Status; AHD, antecedent hematologic disorder; and WBCs, white blood cells.
Of the 511 AML patients, 415 were treated at MDACC and were evaluable for outcome. Among the treated patients, 277 received regimens that contained high-dose cytosine arabinoside (Ara-C), 35 received standard-dose Ara-C, and 8 received low-dose Ara-C. Most of the Ara-C-treated patients also received other treatments. A variety of regimens were given to the rest of the patients. Of the 21 APL patients, 15 received all-trans retinoic acid plus arsenic trioxide, 5 received all-trans retinoic acid plus other drugs, and one received liposomal all-trans retinoic acid. Outcome evaluation was performed during the study period (September 1999 to July 2010). To assess overall survival and remission duration, the vital status of each patient was ascertained as of July 2010.
Sample collection, sample preparation, and ARC RPPA
Proteomic profiling was performed on samples from patients with AML. Samples had been acquired during routine diagnostic assessments in accordance with the regulations and a protocol approved by the MDACC Investigational Review Board. Informed consent had been obtained in accordance with the Declaration of Helsinki. Samples were analyzed under an Investigational Review Board-approved laboratory protocol. Samples were enriched for leukemic cells by performing Ficoll-Hypaque (Sigma-Aldrich) density-gradient separation to yield a mononuclear fraction, followed by CD3/CD19 depletion to remove contaminating T and B cells if they were calculated to be more than 5% on the basis of the differential. After this procedure, the blast purity reached approximately 98% as determined in approximately 20 samples by flow cytometry. The samples were normalized to a concentration of 1 × 104 cells/μL and a whole-cell lysate prepared. RPPA was carried out following the methodology and validation of the technique described fully in previous publications.19,20 Briefly, patient samples were printed in 5 serial dilutions onto slides along with normalization and expression controls. Slides were probed with a strictly validated primary antibody against ARC (Imgenex) and a secondary antibody to amplify the signal, and finally a stable dye22 was precipitated. The stained slides were analyzed using MicroVigene software (Vigene Tech) to produce quantified data.
Cell culture and treatment
OCI-AML3 cells (a cell line harboring an NPM1 type A mutation), kindly provided by Dr M. Minden (Ontario Cancer Institute, Toronto, ON) were cultured in RPMI 1640 medium supplemented with 10% heat-inactivated fetal calf serum, 2mM l-glutamine, 100 U/mL penicillin, and 100 μg/mL streptomycin. Exponentially growing OCI-AML3 cells were electroporated as described previously23 with ARC antisense oligonucleotide (ASO, ISIS454415: TGCCAAGTGGCTCACCAGGG) or a control oligonucleotide (NSO, ISIS141923: CCTTCCCTGAAGGTTCCTCC) (ISIS Pharmaceuticals) using Nucleofector solution T and program X-001 according to the manufacturer's instructions (Amaxa Biosystems). For the combination of ARC ASO and Ara-C, OCI-AML3 cells were electroporated with ARC ASO, cultured for 24 hours, and then treated with Ara-C for an additional 48 hours.
Cell viability
Cell viability was determined by trypan blue exclusion using a Vi-Cell XR Cell Counter (Beckman Coulter). Apoptosis was estimated by flow cytometry measurements of phosphatidylserine externalization24 with annexin V-Cy5 (BD Biosciences) using a FACSArray Bioanalyzer (BD Biosciences). Membrane integrity was assessed simultaneously by 7-amino-actinomycin D exclusion in the annexin V-stained cells.
Western blot analysis
ARC protein levels were determined by Western blot analysis. Signals were detected using Odyssey Infrared Imaging System (LI-COR Biosciences) and quantitated using Odyssey Software Version 3.0 (LI-COR Biosciences). β-actin was included as a loading control.
Statistical analysis
Supercurve algorithms were used to generate a single value from the 5 serial dilutions.25 Loading control26 and topographic normalization procedures accounted for protein concentration and background staining variations. Patients were divided into thirds based on ARC expression levels (low, medium, and high cohorts). Unbiased clustering, perturbation bootstrap clustering, and principal component analyses were performed as described previously.19,20 Comparison of protein levels between paired samples was done using the paired t test. Normalization procedures were performed using the R software program (Version 2.8.0). For correlation of ARC expression with the expression of several other proteins participating in apoptotic mechanisms, we accounted for multiple testing using a Bonferroni correction and thus accepted any proteins with Pearson correlation R > whatever corresponding to a nominal P value: P = .05/176 = .00028. Associations between ARC expression level and categorical clinical variables were assessed in R using standard t tests, linear regression, or mixed-effects linear models. Associations between ARC level and continuous variables were assessed using Pearson and Spearman correlation and linear regression analysis. Bonferroni corrections were performed to account for multiple statistical parameters for calculating statistical significance. The Kaplan-Meier method was used to generate survival curves. Univariate and multivariate Cox proportional hazards modeling was performed to investigate the association between ARC level and survival as categorized variables. Twelve variables were initially used that were univariate predictors in this dataset or that had traditionally been prognostic. A stepwise analysis was conducted until only significant (P < .05) variables remained. Then, the previously removed variables were sequentially added back, one by one, until a final model with only significant variables remained. Outcome analysis and multivariate analysis were carried out using Statistica Version 9 software (StatSoft).
For studies with cell lines, the experiments were conducted 3 times or more and results were expressed as mean plus or minus SEM. Paired t test was used to analyze the significance of ARC knockdown on cell survival, and analysis of variance was used to assess the significance of ARC knockdown on cell sensitivity to Ara-C. P values of less than .05 were considered to be statistically significant.
Results
ARC expression and correlation with levels of other proteins
We determined ARC levels by RPPA in the 511 samples obtained from newly diagnosed AML patients. ARC was found to be variably expressed in these samples independent of patient age and gender (Table 1). ARC levels in 140 paired BM and PB samples were compared to assess whether the sample source affected ARC level, but no differences were observed (P = .628). Therefore, ARC levels in BM samples were used in the main analysis if both were available. Among the 47 cases with a paired diagnosis and relapse sample, there was no consistent pattern of change (P = .495; Figure 1). In most cases, the paired levels were within 2-fold of each other, but ARC levels were twice as likely to be more than or equal to 2-fold higher at relapse (n = 12) than at diagnosis (n = 7, Figure 1).
Comparison of ARC levels in paired newly diagnosed and relapse samples.
In addition to ARC, this same set of samples was also probed with 174 other antibodies, enabling us to correlate ARC expression levels with the levels of other proteins. Notably, ARC levels in the AML samples had significant negative correlations with levels of Bad (R = −0.29, P < .00001), cleaved caspase-9 (R = −0.24, P < .00001), cIAP1 (R = −0.23, P < .00001), caspase-9 (R = −0.22, P < .00001), XIAP (R = −0.21, P < .00001), and Bcl-2 (R = −0.20, P = .00001), and a positive correlation with the level of MDM2 (R = 0.30, P = .00003). The significance of these findings and the full analysis of results for all the other proteins will be reported separately.
Correlation of ARC expression with clinical characteristics of patients
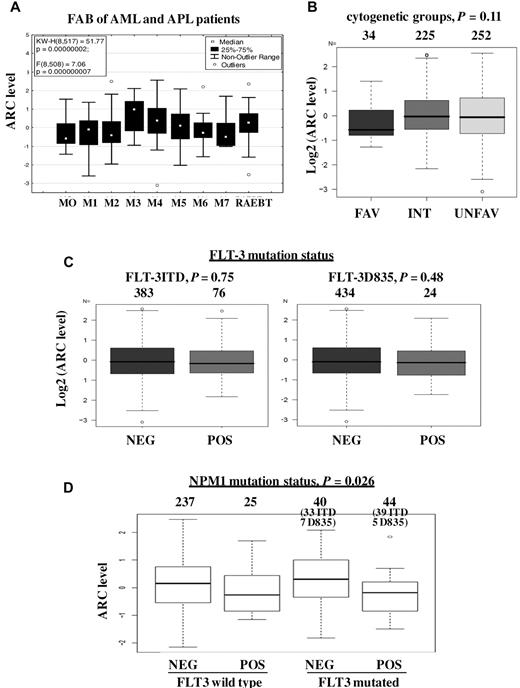
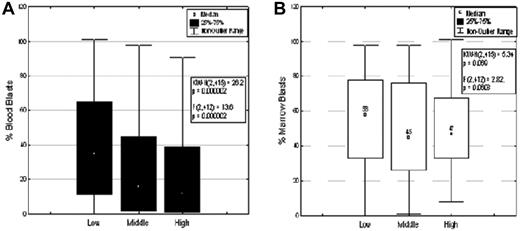
The correlations of ARC expression level with clinical characteristics were determined (Figure 2; Table 1). ARC expression was found to be significantly different among French-American-British (FAB) classes (P < .0001); ARC level was higher in FAB types M4 (P < .0001) and M3 (P = .012) and lower in type M2 (P = .002) (Figure 2A). Although ARC expression differed significantly among specific cytogenetic abnormalities (P = .0061), ARC level was not associated with the broader classification into favorable, intermediate, and unfavorable cytogenetics (P = .11; Figure 2B). The high ARC level observed in karyotype t(15;17) is consistent with the high ARC expression in samples from FAB type M3 patients. No correlation was observed between ARC level and FLT-3 mutation status (Figure 2C). ARC level was significantly lower in patients with mutated NPM1, in both FLT-3 wild-type group and FLT-3-mutated group (P = .026; Figure 2D). ARC level showed a significantly negative correlation with PB blast count (P < .0001; Figure 3A) but did not correlate with BM blast counts (P = .0608; Figure 3B). In addition, no significant relationship was found between ARC level and any of the following variables: patient performance status, antecedent hematologic disorder status, infection status, white blood cell count, platelet count, or hemoglobin (Table 1). ARC level also did not correlate with whether the patients had a history of prior malignancy, chemotherapy, or radiation therapy. Furthermore, the same relationships between ARC level and BM blast count, PB blast count, white blood cell count, platelet count, and hemoglobin levels were also observed when only those patients with intermediate cytogenetics were analyzed (data not shown).
ARC expression and clinical characteristics of AML samples. (A) ARC level and FAB types of AML and APL patients (P < .0001). (B) ARC level and cytogenetic groups (P = .11). (C) ARC level and FLT-3 mutation status: ITD (left, P = .75) and D835 (right, P = .48). (D) ARC level and NPM1 mutation status.
ARC expression and clinical characteristics of AML samples. (A) ARC level and FAB types of AML and APL patients (P < .0001). (B) ARC level and cytogenetic groups (P = .11). (C) ARC level and FLT-3 mutation status: ITD (left, P = .75) and D835 (right, P = .48). (D) ARC level and NPM1 mutation status.
ARC expression and blast counts of AML samples. (A) ARC level and PB blast count (P < .0001). (B) ARC level and BM blast count (P = .0608).
ARC expression and blast counts of AML samples. (A) ARC level and PB blast count (P < .0001). (B) ARC level and BM blast count (P = .0608).
ARC expression is prognostic in AML and correlates with overall survival and remission durations
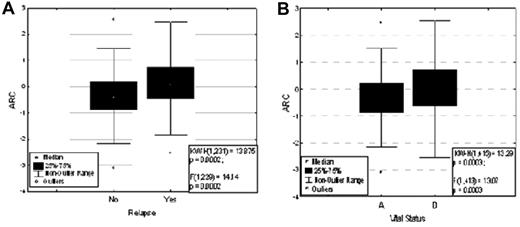
The Lowess smoothed curve for the Martingale residual plot of ARC levels showed a relatively straight line (supplemental Figure 1, available on the Blood Web site; see the Supplemental Materials link at the top of the online article): the linear trend suggested a continuous variable. Therefore, for outcome analysis, the range of ARC levels in patients was arbitrarily divided into thirds, and the treated patients were split into 3 groups by ARC level (low-, medium-, and high-ARC cohorts). This created unbalanced cohort size among the treated patients. Patients with low ARC levels were slightly more likely (55.1%) and those with medium ARC levels were significantly more likely (65.1%) to achieve complete remission (CR) than patients with high ARC levels (51.1%) (P = .045, Table 1). Among the patients who achieved CR (n = 237), those with lower ARC levels were more likely to stay in CR (CCR [continuous complete remission]) and those with higher ARC levels were more likely to relapse (P = .0002, Figure 4A). Furthermore, when patients were identified by their vital status, ARC levels at diagnosis were significantly higher in those patients who had died (n = 308) compared with those who were still alive (n = 107; P = .0003; Figure 4B).
ARC expression and response of AML patients. (A) ARC level and treatment response (P = .0002). (B) ARC level and vital status (P = .0003).
ARC expression and response of AML patients. (A) ARC level and treatment response (P = .0002). (B) ARC level and vital status (P = .0003).
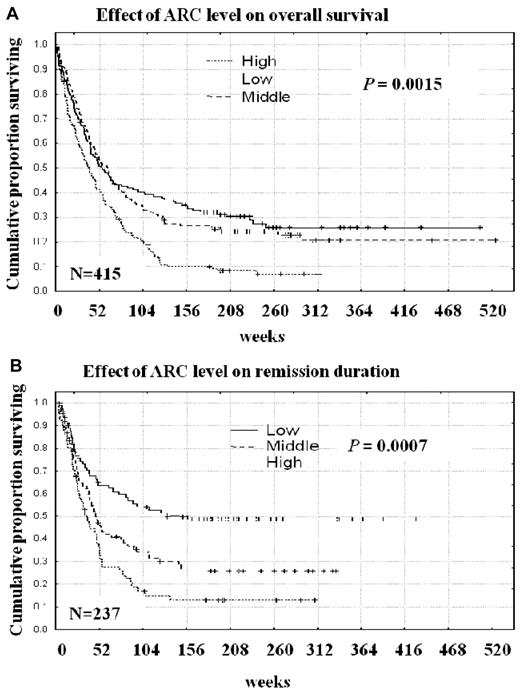
The lengths of overall survival and remission durations were compared among the 3 ARC expression groups. Duration of overall survival was significantly longer for patients with low or medium ARC levels than for those with high ARC levels (median, 53.9 or 61.6 vs 38.9 weeks, P = .0015, Table 1; Figure 5A). Interestingly, among those patients who underwent transplantation, high ARC levels also were associated with poor outcomes. Among the transplantation patients, 10 of 25 with low ARC and 7 of 10 with medium ARC were still alive, compared with 2 of 21 with high ARC levels. Similarly, remission duration was significantly longer for patients with low or medium ARC levels than for patients with high levels of ARC (median, 97.6 or 44.7 vs 31.1 weeks, P = .0007, Table 1; Figure 5B). The prognostic significance of ARC expression was also found to apply to patients with intermediate cytogenetics: patients with high ARC levels had significantly shorter overall survival (P = .014, supplemental Figure 2A) and remission duration (P = .015, supplemental Figure 2B) than patients with low or medium ARC levels. The prognostic impact of ARC expression remained in patients with normal cytogenetics (P = .003, supplemental Figure 3A). Within this group, patients with low ARC levels lived significantly longer than patients with high ARC levels when they had NPM1 mutation (P = .0015, supplemental Figure 3B), although longer but not significant when they had wild-type NPM1 (P = .36, supplemental Figure 3C). We also analyzed the prognostic impact of ARC levels in terms of NPM1 mutation status in patients with any cytogenetics and found similar results: patients with low ARC levels lived significantly longer than patients with high ARC levels when they had the NPM1 mutation (P = .003, supplemental Figure 4A) although longer but not significant when they had wild-type NPM1 (P = .12, supplemental Figure 4B). The prognostic impact was also found to apply to a more homogeneous patient group of adult AML patients younger than 60 years with normal karyotype (P = .010, supplemental Figure 5). Further, we analyzed ARC levels and clinical outcome in patients treated with anthracycline plus histone deacetylase inhibitor, the treatment group enrolled the highest numbers of patients (n = 191) and found that again patients with low ARC levels survived significantly longer than patients with high ARC levels in this uniformly treated group of patients (P = .04, supplemental Figure 6). ARC levels maintained the same clinical significance when patients were divided at the median or into sixths instead of thirds according to ARC levels.
ARC expression and clinical outcome of AML patients. (A) ARC level and overall survival (P = .0015). (B) ARC level and remission duration (P = .0007).
ARC expression and clinical outcome of AML patients. (A) ARC level and overall survival (P = .0015). (B) ARC level and remission duration (P = .0007).
ARC expression is an independent predictor of overall survival in multivariate analysis
Cox proportional hazards modeling was performed to evaluate for factors that were independent predictors of overall survival. Multivariate analysis using ARC level as a continuous variable indicated that ARC is a statistically significant independent predictor of overall survival in AML. The final model included 6 variables, as shown in Table 2: ARC level (P = .00013), age at diagnosis (P < .00001), cytogenetic group (favorable, intermediate, or unfavorable; P < .00001), FLT-3 mutation status (P = .01068), white blood cell count (P < .00001), and albumin level (P = .00014). The risk remains constant over time for high expressers of ARC as the smoothed estimated β parameter is flat over the entire time range, determined by the plot of Schoenfeld residuals (not shown). Similar results were obtained when the values of ARC levels were divided into thirds. ARC remained prognostically significant when its expression was used as a continuous variable in the univariable analysis (P = .00027). Treatment is known to be a significant predictor in univariate analysis. Although the 415 patients used for outcome evaluation received different forms of treatment depending on age and cytogenetics, treatment was a nonsignificant factor in multivariate analysis when age and cytogenetics were included. The prognostic value of ARC was further assessed in adult patients younger than 60 years (n = 174) by multivariate analysis. ARC level was still a significant prognostic factor (P = .023). The final model included the same 6 variables (supplemental Table 1). However, although ARC is predictive individually in AML, it is not a significant predictor of CR attainment in multivariate analysis.
Multivariate analysis final model (n = 415)
| Variable . | β . | SE . | t . | Exponent β . | Wald statistic . | P . |
|---|---|---|---|---|---|---|
| ARC | 0.240 | 0.063 | 3.833 | 1.271 | 14.695 | .00013 |
| Age at diagnosis | 0.039 | 0.005 | 8.242 | 1.040 | 67.931 | .00000 |
| Cytogenetic group | −0.692 | 0.109 | −6.344 | 0.501 | 40.247 | .00000 |
| FLT-3 mutant | −0.365 | 0.143 | −2.553 | 0.694 | 6.518 | .01068 |
| WBCs | 0.006 | 0.001 | 4.931 | 1.006 | 24.315 | .00000 |
| Albumin | −0.313 | 0.082 | −3.814 | 0.731 | 14.544 | .00014 |
| Variable . | β . | SE . | t . | Exponent β . | Wald statistic . | P . |
|---|---|---|---|---|---|---|
| ARC | 0.240 | 0.063 | 3.833 | 1.271 | 14.695 | .00013 |
| Age at diagnosis | 0.039 | 0.005 | 8.242 | 1.040 | 67.931 | .00000 |
| Cytogenetic group | −0.692 | 0.109 | −6.344 | 0.501 | 40.247 | .00000 |
| FLT-3 mutant | −0.365 | 0.143 | −2.553 | 0.694 | 6.518 | .01068 |
| WBCs | 0.006 | 0.001 | 4.931 | 1.006 | 24.315 | .00000 |
| Albumin | −0.313 | 0.082 | −3.814 | 0.731 | 14.544 | .00014 |
WBC indicates white blood cells.
Inhibition of ARC promotes apoptosis and sensitizes Ara-C-induced cell death in OCI-AML3 cells
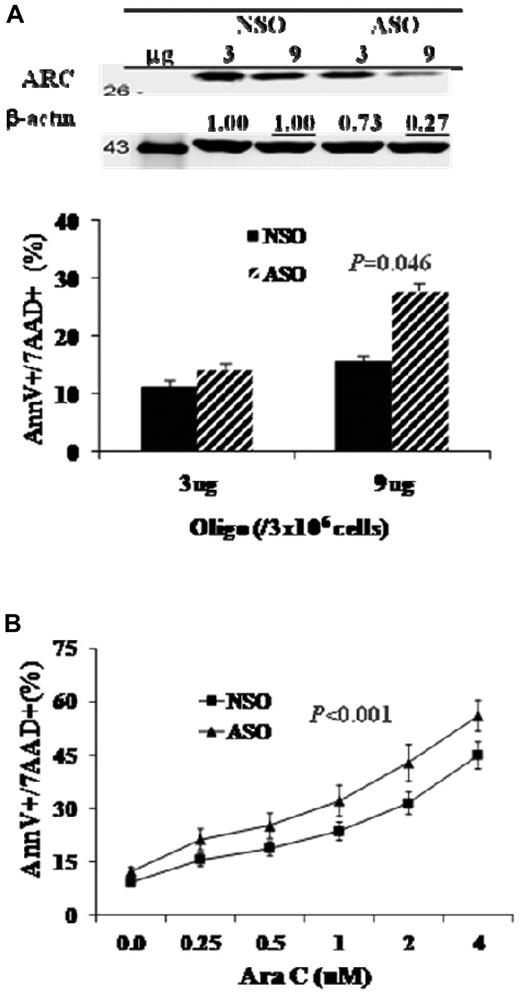
To assess the biologic function of ARC in AML, we treated OCI-AML3 cells with ARC ASO and determined the effects of ARC inhibition on cell viability and chemosensitization. As shown in Figure 6A, ARC ASO inhibited ARC expression and induced dose-dependent apoptosis. Ara-C, the most commonly used therapeutic agent for AML therapy, induced apoptosis in OCI-AML3 cells, and the apoptosis induction was enhanced when ARC expression was inhibited (Figure 6B).
Inhibition of ARC promotes apoptosis and sensitizes Ara-C-induced cell death in AML cells. (A) OCI-AML3 cells were treated with ARC ASO by electroporation for 72 hours. ARC protein levels were determined at 48 hours by Western blot analysis. (B) OCI-AML3 cells were treated with ARC ASO (1 μg/million cells) for 24 hours, then with Ara-C for an additional 48 hours. Cell death was determined by annexin V (AnnV) staining in the presence of 7-amino-actinomycin D (7AAD).
Inhibition of ARC promotes apoptosis and sensitizes Ara-C-induced cell death in AML cells. (A) OCI-AML3 cells were treated with ARC ASO by electroporation for 72 hours. ARC protein levels were determined at 48 hours by Western blot analysis. (B) OCI-AML3 cells were treated with ARC ASO (1 μg/million cells) for 24 hours, then with Ara-C for an additional 48 hours. Cell death was determined by annexin V (AnnV) staining in the presence of 7-amino-actinomycin D (7AAD).
Discussion
In this study, we profiled the expression of ARC using a novel and robust RPPA method in samples obtained from 511 well-characterized AML patients and assessed the clinical significance of this protein. The results demonstrated that ARC was variably expressed in newly diagnosed AML samples. Importantly, we found that ARC expression was highly prognostic in AML. A high ARC level was a strong adverse prognostic factor for overall survival and remission durations. Furthermore, ARC level was an independent predictor of outcome in AML. An independent confirmation of this important finding is needed.
ARC levels did not change in a consistent direction between diagnosis and relapse, but ARC levels were found twice as likely to be more than or equal to 2-fold higher at relapse (n = 12) than at diagnosis (n = 7), suggesting that a larger paired sample size is needed to draw a definitive conclusion. It is worth noting that, among the patients who achieved CR (n = 237), those with higher ARC levels were more likely to relapse (n = 146, P = .0002). Therefore, for the paired samples, ARC levels were relatively high, even at diagnosis, compared with the diagnosis samples from patients who did not relapse.
We observed that ARC expression is correlated with the levels of several apoptosis-related proteins. In particular, ARC was found to be negatively correlated with levels of the antiapoptotic proteins cIAP1, XIAP, and Bcl-2. p53 was previously reported to negatively regulate ARC levels, and ARC was found to antagonize p53 function and alter its cellular localization.15,17 However, we did not find a negative correlation between ARC and p53 levels, but we found a positive correlation between ARC and MDM2 by RPPA. This is not surprising because p53 signaling and regulation are complex. p53 is negatively regulated by MDM2 via proteasomal degradation. p53 is normally expressed at low or undetectable levels in untreated cells. When activated, p53 not only regulates the expression of multiple genes involved in cell growth and survival but also induces MDM2 expression, which in turn keeps p53 levels in check. In malignant cells, the loss of p53 function can be caused either by p53 gene deletion and MDM2 overexpression, which result in low or undetectable levels of p53, or by p53 gene mutations, which lead to high levels of p53 because MDM2-mediated proteasomal degradation is lacking. The RPPA set we used can neither distinguish between wild-type and mutated p53 nor ascertain whether low or undetectable expression of p53 is the result of low basal levels of p53, deletion of the p53 gene, or overexpression of MDM2. We sequenced the p53 gene in 42 samples from this cohort of patients. Although we observed mutations in 17 of 32 samples with very high p53 levels and none of 10 samples with levels below the median, p53 mutation status did not correlate with ARC levels. Furthermore, it needs to be pointed out that cellular localization, which contributes to p53 function and signaling, cannot be assessed by RPPA. The lack of a correlation between ARC and p53 levels does not rule out the interaction of these 2 proteins. How ARC is regulated by p53 and the relationship between ARC and p53 signaling in AML are topics for future studies.
We found that ARC expression correlated with the FAB types M2, M3, and M4 in AML patients. This correlation probably does not suggest a relationship with cellular differentiation stage because it was not found for other FAB types. Furthermore, we found that ARC levels were negatively correlated with PB blast count (P < .00001). This correlation does not lend itself to an obvious explanation at this time.
ARC expression was highly prognostic in AML. A high ARC level was a strong adverse prognostic factor for overall survival and remission duration. It is important to emphasize that AML is a stem cell disease and that the ineffectiveness of chemotherapy in eradicating leukemic stem cells contributes at least in part to the inevitable relapse of AML. The fact that a high ARC level was a strong adverse prognostic factor for remission duration may suggest that high ARC expression in AML stem cells protects them from chemotherapy-induced cell death. We will determine the expression of ARC in the CD34+38− compartment of AML in a future study using RPPA. The fact that ARC expression level was an independent predictor of outcome in AML makes it potentially useful as a prognostic factor for AML. Importantly, because ARC is an antiapoptotic protein and is associated with a poor clinical outcome in AML, ARC may serve as a molecular target in AML therapy. Indeed, we found that inhibition of ARC promoted apoptosis and sensitized Ara-C-induced cell death in AML cells (Figure 6).
In the past few years, new molecular diagnostic and prognostic markers for AML have emerged, such as the expressions of ERG, BAALC, EVI1, and CEBPA. Many of these markers were discovered after this patient cohort was diagnosed and treated, so the data are not available from these patients. It will be interesting to include these data and establish a possible relationship of ARC with these markers in the future.
The online version of this article contains a data supplement.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Acknowledgments
The authors thank Bradley S. Tadlock and Karen R. Muller for assisting with the manuscript preparation.
This work was supported in part by the Leukemia & Lymphoma Society (S.M.K.) and the National Institutes of Health (P01 CA55164; M.A.).
National Institutes of Health
Authorship
Contribution: B.Z.C. conceptualized the study and wrote the paper; Y.H.Q. and D.H.M. performed experiments; N.Z. and K.R.C. analyzed the data; D.A.T., F.R., and H.M.K. helped with patient data and patient samples; E.K. provided agents for the study; M.A. supported the study and edited the paper; and S.M.K. analyzed the data, supported the study, and wrote the paper.
Conflict-of-interest disclosure: E.K. works for Isis. The remaining authors declare no competing financial interests.
Correspondence: Steven M. Kornblau, Section of Molecular Hematology and Therapy, Department of Leukemia, Unit 448, University of Texas M. D. Anderson Cancer Center, 1515 Holcombe Blvd, Houston, TX 77030; e-mail: skornblau@mdanderson.org; and Bing Z. Carter, Section of Molecular Hematology and Therapy, Department of Leukemia, Unit 448, University of Texas M. D. Anderson Cancer Center, 1515 Holcombe Blvd, Houston, TX 77030; e-mail: bicarter@mdanderson.org.


This feature is available to Subscribers Only
Sign In or Create an Account Close Modal