Abstract
Background: AYA patients with cancer comprise a unique and understudied population with higher treatment-related toxicity than younger children.
Objective: To describe the outcome of AYA patients with AML treated with intensive chemotherapy and GO.
Methods: AYA patients (ages 16-21) on Children’s Oncology Group (COG) trials AAML03P1 and AAML0531 were analyzed and compared to younger patients. Both trials included patients with newly diagnosed AML. Differences in disease characteristics, treatment response, treatment-related mortality (TRM) and relapse risk (RR) were compared. Patients received either 5 chemotherapy courses or 3 followed by stem cell transplant (SCT) dependent on donor availability alone in 03P1, and risk stratification and donor availability in 0531. On AAML0531, patients with t(8;21) or inv(16) were considered low risk and did not receive SCT. High risk patients had adverse cytogenetics (-7, -5/5q-), high FLT3-ITD HAR (>0.4) or slow response with >15% marrow disease after induction 1. High risk patients received SCT if any donor was available. All other patients were intermediate risk and received SCT if a matched family donor was available.
GO (3 mg/m2/dose) was administered to all patients on AAML03P1 while patients on AAML0531 were randomized to treatment with or without GO. Patients scheduled to receive GO received one dose on induction 1, day 6, and for patients that did not proceed to SCT, a second dose was given on intensification 2, day 7.
Results: A total of 1350 patients 21 years old and younger were studied. Significantly different characteristics of AYA compared to younger patients are presented in Table 1, whereas gender, race, ethnicity, t(8;21) and inv(16) were similar between age groups.
Significant differences by age
| . | Age < 16 Years (n=1144) . | Age 16-21 Years (n=206) . | p value . |
|---|---|---|---|
| Normal Cytogenetics | 225 (20.5%) | 70 (36.5%) | <0.001 |
| 11q23/MLL rearrangement | 256 (23.3%) | 22 (11.5%) | <0.001 |
| FLT3-ITD high allelic ratio | 146 (14.3%) | 37 (20%) | 0.047 |
| CEBPα mutation | 46 (4.8%) | 17 (9.4%) | 0.012 |
| NPM mutation | 58 (6%) | 23 (12.8%) | 0.001 |
| Death before remission | 25 (2.4%) | 5 (2.5%) | 0.803 |
| CR after Induction I | 826 (74.1%) | 165 (82.1%) | 0.015 |
| MRD negative after Induction I | 572 (68.3%) | 123 (76.4%) | 0.042 |
| . | Age < 16 Years (n=1144) . | Age 16-21 Years (n=206) . | p value . |
|---|---|---|---|
| Normal Cytogenetics | 225 (20.5%) | 70 (36.5%) | <0.001 |
| 11q23/MLL rearrangement | 256 (23.3%) | 22 (11.5%) | <0.001 |
| FLT3-ITD high allelic ratio | 146 (14.3%) | 37 (20%) | 0.047 |
| CEBPα mutation | 46 (4.8%) | 17 (9.4%) | 0.012 |
| NPM mutation | 58 (6%) | 23 (12.8%) | 0.001 |
| Death before remission | 25 (2.4%) | 5 (2.5%) | 0.803 |
| CR after Induction I | 826 (74.1%) | 165 (82.1%) | 0.015 |
| MRD negative after Induction I | 572 (68.3%) | 123 (76.4%) | 0.042 |
5 year EFS and OS from study entry was 44.2% and 60.0% for AYA patients compared to 50.2% and 64.8% for younger patients (p=NS). AYA patients had significantly higher TRM when compared to younger patients: 13.3% vs. 7.3% from study entry (p=0.005) primarily due to death in remission, 12.4% vs. 5.6% (p=0.004). Despite similar 5 yr relapse rates after achieving remission, 38.8% vs, 36.7%, 5 yr DFS (48.8% vs. 57.7%, p=0.058) and OS (63.7% vs. 70.9%, p=0.067) from end of induction 2 trended worse for AYA patients, reflective of the increased treatment-related death in remission.
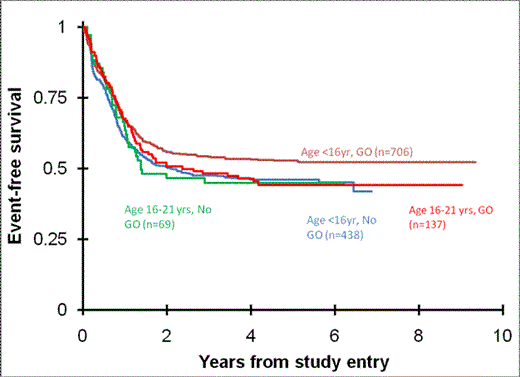
AYA patients that received GO combined with chemotherapy had similar EFS and OS compared to those treated without GO (Figure 1). TRM in AYA patients treated with GO was significantly higher compared to younger patients (15.5% vs. 8.6%, p=0.014). In younger patients, while GO added to chemotherapy did not result in improved OS, a significant improvement in EFS was seen. For these patients, the decrease in the relapse rate was not offset by an increased TRM as was seen in AYA patients (Table 2).
Differences in outcomes by use of GO
| . | GO . | No GO . | p value . |
|---|---|---|---|
| AYA patients (16-21 years) | n=137 | n=69 | |
| 5 year OS | 60.8% | 58.5% | 0.682 |
| 5 year EFS | 44.3% | 44.9% | 0.754 |
| 5 year TRM | 15.5% | 8.7% | 0.21 |
| 5 year RR* | 36.7% | 42.5% | 0.262 |
| Younger patients (<16 years) | n=706 | n=438 | |
| 5 year OS | 65.5% | 63.7% | 0.580 |
| 5 year EFS | 52.8% | 46.0% | 0.018 |
| 5 year TRM | 8.6% | 5.3% | 0.045 |
| 5 year RR* | 32.9% | 42.6% | <0.001 |
| . | GO . | No GO . | p value . |
|---|---|---|---|
| AYA patients (16-21 years) | n=137 | n=69 | |
| 5 year OS | 60.8% | 58.5% | 0.682 |
| 5 year EFS | 44.3% | 44.9% | 0.754 |
| 5 year TRM | 15.5% | 8.7% | 0.21 |
| 5 year RR* | 36.7% | 42.5% | 0.262 |
| Younger patients (<16 years) | n=706 | n=438 | |
| 5 year OS | 65.5% | 63.7% | 0.580 |
| 5 year EFS | 52.8% | 46.0% | 0.018 |
| 5 year TRM | 8.6% | 5.3% | 0.045 |
| 5 year RR* | 32.9% | 42.6% | <0.001 |
*In CR patients (AYA GO n=114; no GO n=59; <16y GO n=577, no GO 356)
SCT outcomes by age group revealed similar results with increased TRM among AYA SCT recipients (n=37) compared to younger SCT recipients (n=172) (18.9% v 7.8%, p=0.027) and resulted in a lower 5 yr DFS (50.1% v 62.7%, p=0.214) despite similar RR (31% v 29.5%, p=0.854).
Conclusion: AYA patients on these studies experienced lower OS and EFS when compared to younger patients. The favorable impacts of GO and SCT were abrogated in AYA patients due to higher TRM negating the improved RR with their use. In the AYA population, further focus upon reducing TRM is needed in order to achieve similar benefits.
A report from the Children’s Oncology Group
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal