All mice harboring the X-linked Gata1low mutation in a predominantly CD1 background are born anemic and thrombocytopenic. They recover from anemia at 1 month of age but remain thrombocytopenic all their life and develop myelofibrosis, a syndrome similar to human idiopathic myelofibrosis, at 12 months. The effects of the genetic background on the myelofibrosis developed by Gata1low mice was assessed by introducing the mutation, by standard genetic approaches, in the C57BL/6 and DBA/2 backgrounds and by analyzing the phenotype of the different mutants at 12 to 13 (by histology) and 16 to 20 (by cytofluorimetry) months of age. Although all the Gata1low mice developed fibrosis at 12 to 13 months, variegations were observed in the severity of the phenotype expressed by mutants of different backgrounds. In C57BL/6 mice, the mutation was no longer inherited in a Mendelian fashion, and fibrosis was associated with massive osteosclerosis. Instead, DBA/2 mutants, although severely anemic, expressed limited fibrosis and osteosclerosis and did not present tear-drop poikilocytes in blood or extramedullary hemopoiesis in liver up to 20 months of age. We propose that the variegation in myelofibrosis expressed by Gata1low mutants of different strains might represent a model to study the variability of the clinical picture of the human disease.
Introduction
Gata1 is the member of the GATA transcription factor family1 essential for appropriate erythroid,2 megakaryocytic,3,4 eosinophilic,5 and mast6 cell differentiation. Because of the highly conserved structure shared by the GATA proteins,7 the functions of GATA-1 are highly dependent on appropriate lineage expression.8,9 The importance of the region of the X chromosome upstream to Gata1, which includes the DNA hypersensitive site I, for appropriate gene expression in erythroid10,11 and megakaryocytic (Mk)12,13 cells was demonstrated by its targeted deletion in 6-SV 295 embryonic stem cells that were used to generate GataneoδHS (or Gata1low) mutants, subsequently maintained by crossing with C57BL/6 mice.10 Gata1low mutants are anemic10,11 and thrombocytopenic,12,13 and more than 95% of them die at birth. Anemia is due to the fact that the mutation reduces Gata1 expression in erythroblasts, increasing (up to 30%) their apoptotic rate,10,11 while thrombocytopenia is caused by lack of Gata1 expression in Mks that remain immature and release abnormal platelets, the megathrombocytes.12,13 These Mk alterations have been implicated in the massive bone formation observed in the mutants that survive up to 5 months of age.14
When the mutation was transferred in the CD1 background, pups were still born thrombocytopenic and anemic, but a higher number (> 50%) of them survived to adulthood (Vannucchi et al15 and Figure 1). These mutants recovered from anemia at 1 month, thanks to the homeostatic mechanism that recruits the spleen as the hemopoietic site and increases the erythroid output precisely as necessary to compensate for the erythroblast apoptosis induced by the mutation.15 This mechanism, unique to mice, also is involved in the recovery from anemia induced by hemolytic agents,16 radiation,17 and deletions of other erythroid genes, such as Stat518 and Lyn,19 but does not restore megakaryocytopoiesis. Therefore, GATA-1low mutants remain thrombocytopenic all their life in spite of the numerous Mks present in their tissues.20 The abnormalities of Gata1low Mks include increased P-selectin localization on the demarcation membrane system (DMS) and pathologic neutrophil emperipolesis,21 2 defects also expressed by Mks from patients with idiopathic myelofibrosis (IM),22,23 a clonal myeloproliferative disorder characterized by thrombocytopenia and/or anemia, marrow fibrosis, increased stem/progenitor cell trafficking, and extramedullary hemopoiesis that may be associated with osteosclerosis and eventually evolve to leukemia.24-26 Indeed, Gata1low mice developed myelofibrosis, a syndrome similar to IM that manifested itself at 10 to 12 months of age with marrow and spleen fibrosis and, from 15 months on, with tear-drop poikilocytes in blood, increased stem/progenitor cell trafficking, and extramedullary hemopoiesis in liver.20 In these mutants, marrow fibrosis was associated with osteosclerosis, but bone formation never reached the levels described for the original C57BL/6/6-SV 295 mutants,14,20 suggesting that the genetic background might affect the complexity of the phenotype induced by the mutation.
Stem cells from C57BL/6 and DBA/2 mice are known for the differences in how their cycling properties change with age.27,28 These differences have been evidenced by many experimental models, the most clear of which is probably represented by chimeric DBA/2-C57BL/6 animals.29 In these mice, hemopoiesis is primarily from DBA/2 stem cells in young animals but derives mainly from C57BL/6 stem cells as the mice grow old.29 This is due to the fact that DBA/2 stem cells proliferate faster and initially overcome the C57BL/6 ones but, being also extinguished faster, are later on competed out by C57BL/6 cells.27,28 These stem cell differences are due to polymorphisms in loci termed quantitative trait loci (QTL).30,31 Since Gata1low mice developed myelofibrosis after 12 months of age and the disease involved massive amplification of the progenitor cell compartments,6,20 QTL might cooperate with the mutation in determining the complexity of the phenotypic trait. To test this hypothesis, the mutation was introduced in the C57BL/6 and DBA/2 strains and the phenotype expressed by Gata1low mutants of different strains compared. Indeed, myelofibrosis was developed by all the Gata1low mutants, but marked strain variegations were observed. In the C57BL/6 background, the mutation ceased to be inherited in Mendelian fashion, and the mutants developed fibrosis quickly and with massive osteosclerosis. In contrast, DBA/2 mutants, although severely anemic, expressed limited fibrosis and osteosclerosis and did not present tear-drop poikilocytes in blood or extramedullary hemopoiesis in liver up to 20 months of age. This variegation suggests the existence of gene modifier(s), possibly represented by QTL, that might enhance/suppress those traits pleiotropic to the Gata1low mutation itself, and might represent a model with which to study the variability of the clinical picture of IM in humans.
Summary of breeding GATA-1low mutants with normal DBA/2, C57BL/6, and CD1 mice. For the purpose of this study, the contribution of each parental background to the genotype of the offspring was calculated under the assumption that each parent contributed 50% of the alleles and that the effect of crossing-over events to the allelic transmission was negligible. Crossings that do not follow Mendelian inheritance are indicated in gray. The results represent the summary of at least 20 to 30 matings for each crossing.
Summary of breeding GATA-1low mutants with normal DBA/2, C57BL/6, and CD1 mice. For the purpose of this study, the contribution of each parental background to the genotype of the offspring was calculated under the assumption that each parent contributed 50% of the alleles and that the effect of crossing-over events to the allelic transmission was negligible. Crossings that do not follow Mendelian inheritance are indicated in gray. The results represent the summary of at least 20 to 30 matings for each crossing.
Materials and methods
Mice
The Gata1low colony was started by mating a genetically modified male10 (of mixed C57BL/6 and 6-SV 295 backgrounds, kindly provided by Dr S. Orkin) with CD1 females (Charles River, Calco, Italy). Their F1 offspring was crossed again until a line of homozygous mutants, with a genotype 62.5% CD1 and 37.5% C57BL/6 plus 6-SV 295 in unknown proportion, was obtained.15 The colony was maintained thereafter at the homozygous stage. To transfer the mutation in different backgrounds, CD1, DBA/2 (DBA/2NCrIBR), and C57BL/6J (C57BL/6JCrIBR) females (Charles River) were mated with third-to-fifth generation GATA-1low/0 males as described in Figure 1. Littermates were genotyped at birth by polymerase chain reaction (PCR)6,10 and housed for up to 2 years under good animal care practice conditions in the animal facilities of Istituto Superiore Sanità. Littermates negative for the Gata1low mutation—that is, wild-type at the Gata1 locus—were analyzed as normal controls. All the experiments were performed with sex-matched and age-matched mice under protocols approved by the Institutional Animal Care Committee.
Hematologic parameters
Blood was collected from the retro-orbital plexus into ethylen-diamino-tetracetic acid-coated microcapillary tubes (20-40 μL/sampling). Hematocrit (Hct), white blood cells (WBCs), and platelet (ptl) counts were determined manually.
Histology
Liver, spleen, and femurs were fixed in 10% (vol/vol) phosphate-buffered formalin (Sigma, St Louis, MO), paraffin embedded, and cut into 2.5- to 3-μM sections that were stained with hematoxylin-eosin, Gomori-silver (MicroStain MicroKit, Diapath, Bologna, Italy), or the osteoclast-specific tartrate-resistant acid phosphatase (387-A Kit, Sigma) staining. Microscopic evaluations were performed using an Axioskop 40 microscope (Carl Zeiss, Oberkochen, Germany) equipped with 10 ×/0.30 numerical aperture (NA) air, 20 ×/0.50 air, or 100 ×/1.3 NA oil immersion objectives. Images were acquired with CoolSNAP cf digital CCD camera (Photometrics, Huntington Beach, CA) and elaborated with the MetaMorph Imaging System (Molecular Devices, Union City, CA). The number of Mks was determined at 40 × original magnification in randomly chosen multiple sections to cover a total area of 33.5 mm.2 Fibrosis was quantified on images of Gomori-stained sections according to Beham-Schmid et al32 and modified by Vannucchi et al.20 Mean (± SD) numbers of fiber intersections were calculated from values obtained on at least 3 different sections/organ/mouse.
Flow cytometry and cell sorting
Mononuclear cells obtained from liver, spleen, and marrow were suspended in Ca++ Mg++-free phosphate-buffered saline supplemented with 1% (vol/vol) bovine serum albumin, 2 mM EDTA (ethylenediaminetetraacetic acid), 0.1% NaN3, and incubated for 30 minutes on ice with 1 μg/106 cells of phycoerythrin (PE)-conjugated CD117 (anti-c-kit) and TER119, and fluorescein isothiocyanate (FITC)-conjugated anti-CD34, anti-Sca-1, and CD71 (all from Pharmingen, San Diego, CA). Cell fluorescence was analyzed with a Beckman Coulter Epix Elite ESP (Beckman Coulter, Miami, FL). Frequencies of stem/progenitor cells in spleen were analyzed on light density (ρ < 1.080) cells separated over Ficoll (Sigma). Blood mononuclear cells were enriched by hypotonic lysis (0.87% NH4Cl for 15 minutes on ice). Dead cells and nonspecific signals were excluded by propidium iodide staining (5 μg/mL, Sigma) and appropriate isotype controls (Pharmingen). In selected experiments, cells in the prospectic stem/progenitor cell gates were sorted (80%-90% pure upon reanalysis) with a cell sorter FACS ARIA (Becton Dickinson, Franklin Lakes, NJ).
Progenitor cell counts
The frequency of progenitor cells in purified marrow and spleen cells from representative normal and GATA-1low littermates was determined by plating 100 cells/mL in standard methylcellulose cultures (0.9% wt/vol) containing fetal bovine serum (30% vol/vol, Sigma) and recombinant growth factors (rat stem cell factor [SCF, 100 ng/mL], mouse interleukin-3 [IL-3, 10 ng/mL], granulocyte-colony stimulating factor [G-CSF], and granulocyte-macrophage colony stimulating factor [GM-CSF] [50 ng/mL each] [all from Sigma] and human erythropoietin [EPO, 2 U/mL; Boehringer Mannheim, Mannheim, Germany]).33 The cultures were incubated at 37°C in a humidified incubator containing 5% CO2 in air and colonies derived from more or less mature progenitor cells were scored at 8 and 15 days.
RNA isolation and quantitative RT-PCR analysis
RNA was prepared by lysing bone marrow with Trizol (Gibco BRL, Paisley, United Kingdom) and reverse transcribed with 2.5 μM random hexamers using the superscript kit (InVitrogen, Milan, Italy). Transforming growth factor-β (TGF-β1) cDNA was quantified using a TaqMan reverse transcriptase-polymerase chain reaction (RT-PCR) assay (catalog no. Mm00441724_m1, Applied Biosystems, Foster City, CA), while osteocalcin cDNA was quantified by SYBR-green technology (SYBR green PCR master mix, Applied Biosystems).34 Glyceraldehyde phosphate dehydrogenase (GAPDH) cDNA was concurrently amplified as a control. Reactions were performed in an ABI PRISM 7300 Sequence Detection System (Applied Biosystems). Data were analyzed and the cycle threshold (Ct) calculated with the Sequence Detection System (SDS) software. cDNA levels were expressed as ΔCt (ΔCt = target gene Ct - GAPDH Ct).
Statistical analysis
Statistical analysis was performed by analysis of variance (Anova test) using Origin 3.5 software for Windows (Microcal Software, Northampton, MA).
Results
Mendelian inheritance of the GATA-1low mutation in DBA/2 but not in CD1 or C57BL/6 backgrounds
To introduce the Gata1low mutation in different backgrounds, normal C57BL/6, DBA/2, and CD1 females were crossed with hemizygous Gata1low/0 males (from the same litter) and their F1 heterozygous female offspring backcrossed with normal males (C57BL/6, DBA/2, or CD1, as appropriate) to obtain mutant F2 mice (75% C57BL/6 or DBA/2 and 90.6% CD1, respectively). The mating of normal females with the original Gata1low/0 males had a low (14%-25%) success rate that was not reflected by deviations in the expected Mendelian inheritance ratio in the F1 offspring (Figure 1). In fact, wild-type males and heterozygous females represented the expected 50:50 offspring in both DBA/2 and C57BL/6 F1 litters. An exception was represented by CD1 F1 litters, in which normal males prevailed over heterozygous females (77% vs 23%). In contrast, in the F2 litters the expected 25% Mendelian rate was observed only among the DBA/2 offspring. In CD1 F2 litters, Gata1low/0 males and Gata1low/+ females represented only 10% of the total offspring, and Gata1low/0 males were never detected in C57BL/6 F2 litters. These results indicate that the Gata1low mutation is lethal in C57BL/6 but not in DBA/2 mice.
Changes with age in blood values and femur and spleen cellularity in GATA-1low mutants of different backgrounds
Blood values and marrow and spleen cellularity of 12- to 13- and 16- to 20-month-old Gata1low mice and normal littermates are presented in Table 1. As expected, 12- to 13-month-old wild-type mice of different strains expressed similar levels of Hct (.44-.49 [44%-49%]), ptl (900-1300 × 109/L [0.9-1.3 × 106/μL]), and WBC (0.008-0.011 × 109/L [8-11 × 103/μL]) counts. These values did not change with age, since the small Hct reduction observed in normal DBA/2 and CD1 mice at 16 to 20 months was not statistically significant (Table 1).
All of the mutant mice expressed a number of blood thrombocytes significantly lower than normal (Table 1). Ptl counts were not affected by genetic background and age but were sensitive to gene dose, since ptl counts of heterozygous F1 + F2 females were 3 times higher than those of hemizygous F2 males (approximately 420 × 109/L vs 170 × 109/L [0.42 × 106/μL vs 0.17 × 106/μL], respectively) (Table 1). This is probably due to the fact that, because of X inactivation, in heterozygous females, hemopoiesis derives both from normal and Gata1low stem cells. Clusters of normal platelets, likely derived from the normal stem cells, are presented in Figure 2C.
The observation that human females carrying missense mutations in the zinc finger domains of Gata1 have normal platelet counts had suggested that stem cells expressing Gata1 from the normal allele have a proliferation advantage over those expressing the mutant one.35 In contrast, the platelet counts in the heterozygous female mice did not increase with age (Table 1). This difference might be due either to specie-specific Gata1 function or to the fact that alterations in levels of expression or in the structure of Gata1 have different consequences at the stem/progenitor cell level.
At 12 to 13 months of age, all of the mutant mice expressed Hct similar to controls, with the exception of hemizygous DBA/2 males that were clearly anemic (Table 1). At 16 to 20 months of age, the Hct became significantly lower than normal in DBA/2 mutants (both heterozygous females and hemizygous males) and in CD1 hemizygous males. Low levels of Hct were not necessarily associated with presence of tear-drop poikilocytes in blood. In fact, numerous tear-drop poikilocytes were already present on blood smears from C57BL/6 mutants that had normal Hct at 12 months of age (Figure 2B) but were undetectable on smears from DBA/2 mutants, in spite of their anemia, up to 16 to 20 months (Figure 2C and results not shown). As expected,20 tear-drop poikilocytes appeared on blood smears from CD1 mutants at 16 to 20 months of age (not shown).
Although Gata1 should not affect myeloid differentiation,1 most of the mutants had WBC counts significantly lower than normal (0.0039-0.0055 × 109/L vs 0.008-0.011 × 109/L [3.9-5.5 × 103/μL vs 8-11 × 103/μL], respectively, Table 1). This might be due to abnormalities in WBC mobilization due to splenomegaly and/or fibrosis. Unfortunately, this hypothesis cannot be supported by data on spleen weights that were not recorded.
As expected, femur and spleen cellularity of normal mice was greatly affected by genetic background. At 12 to 13 months, the average cell number of the femur from normal mice ranged from approximately 25 × 106 (C67BL/6) to approximately 13 × 106 (DBA/2), while that of the spleen ranged from 180 × 106 (CD1) to 70 × 106 (C57BL/6). In normal controls, the cell number per femur and spleen did not change with age, with the exception of a modest, but not significant, decrement observed for the femur of DBA/2 mice and the spleen of CD1 animals (Table 1).
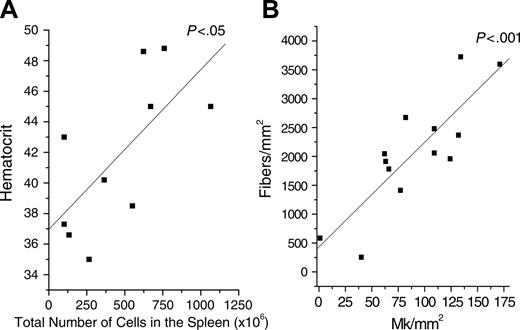
The cell number in the femur from 12- to 13-month-old mutants was 2- to 5-fold lower than normal in all of the backgrounds investigated (Table 1). This number did not change within heterozygous C57BL/6 females, increased by 5-fold in heterozygous DBA/2 females, and decreased by 2- to 3-fold in all of the hemizygous males. On the other hand, spleen cellularity strikingly increased in heterozygous females, independently from background, and in hemizygous males of CD1 background (Table 1). Of interest, the total cell number in the spleen from hemizygous DBA/2 males, the most severely anemic, was not statistically higher than normal. The spleen cellularity and the Hct of the different mutant mice were directly correlated (P < .05, Figure 3A).
Therefore, thrombocytopenia was homogenously expressed by all the mutant mice analyzed, while occurrence of anemia was greatly dependent on genetic background and inversely correlated with spleen cellularity.
Frequency of Mk and presence of osteosclerosis and fibrosis in hemopoietic tissues from 12- to 13-month GATA-1low mutants of different backgrounds
It has been reported that the Gata1low mutation increases the number of Mks present in the hemopoietic tissues.12,20,21 Such increase was no longer detectable at 12 to 13 months in all the mutants. In F1 + F2 heterozygous females, the frequency of Mks was similar to and 2-fold higher than normal in marrow and spleen, respectively. In hemizygous males, the frequency of Mks was 3-fold and 10-fold higher than normal in marrow and spleen of CD1 mutants, but was similar to and 5-fold higher than normal in marrow and spleen of DBA/2 mice (Figure 2 and Table 2). Interestingly, Mks, which were barely detectable in liver parenchyma from normal littermates and from F1-F2 heterozygous females of C57BL/6 and DBA/2 backgrounds, were consistently observed in liver from hemizygous DBA/2 males and from both heterozygous females and hemizygous males of CD1 background (Figure 2 and Table 2).
Limited tissue fibrosis is associated with the process of aging. Consistent with this, some fibrosis was detected in hemopoietic tissues from all 12- to 13-month-old normal mice analyzed (Table 2). Fibrosis, however, was significantly higher than normal in femur and spleen of all the mutants analyzed. The numbers of Mks and of fibers were not correlated in the marrow but were significantly (P < .001) and linearly correlated in the spleen (Figure 3B). Fibrosis also was detected in liver from DBA/2 and CD1 mutants (ie, in all those cases in which significant numbers of Mks were detectable in this organ) (Figure 2 and Table 2).
The lack of correlation between number of Mks and extent of fibrosis in the marrow at 12 months of age might be due to the fact that the process of new fiber formation is no longer active in this organ. We had previously shown that, in Gata1low mutants, fibrosis is associated with high levels of TGF-β expression.20 Therefore, to prove whether fibrosis was still active in the marrow of old mutants, we compared, by quantitative RT-PCR, the levels of TGF-β expressed in this organ from Gata1low mice of different backgrounds. Comparable levels of TGF-β were expressed by the bone marrow of old wild-type animals (Table 3). On the other hand, the bone marrow from the mutants expressed levels of TGF-β either similar to (C57BL/6) or lower (DBA/2 and CD1) than normal (Table 3). These results confirm that active fibrosis was reduced in the marrow from old Gata1low animals.
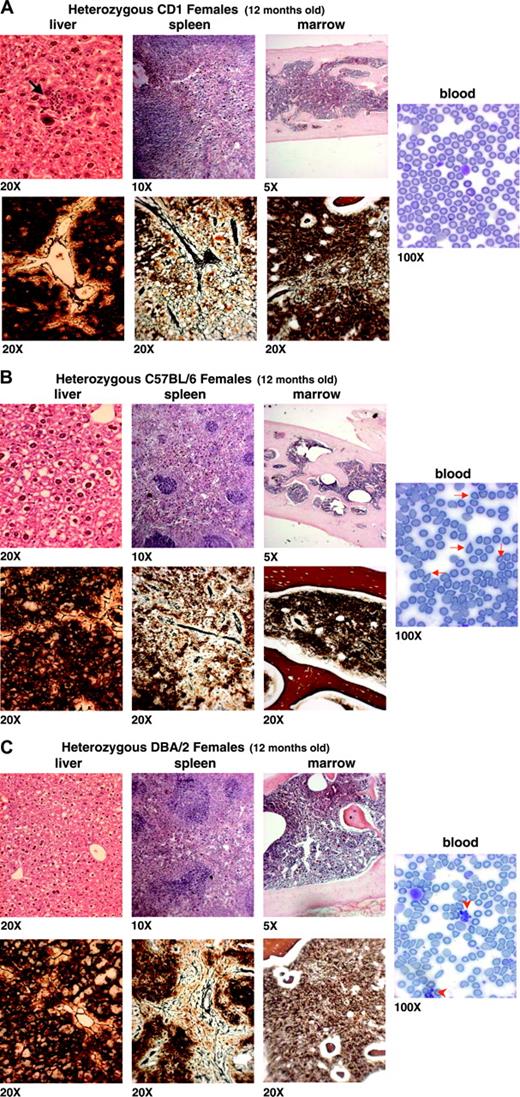
Histologic analysis of the myelofibrotic trait in 12-month-old heterozygous F1 Gata1low females carrying the mutation in a predominantly CD1, C57BL/6, or DBA/2 background. Blood smears are shown on the right panels, while hematoxilin-eosin and Gomori staining of liver, spleen, and marrow are shown on the top and bottom panels, as indicated. The genetic background of the mice analyzed is specified in Figure 1, while the quantification of Mk frequency and of the fibrosis in each animal group is presented in Table 2. The arrow in the liver section in panel A indicates an erythroid nest within the parenchyma, already detectable at 12 months in CD1 mutants, while the arrowheads on the blood smears in panel C indicate clusters of normal platelets, that is, deriving from the stem cell population that had inactivated the X chromosome carrying the Gata1low allele. These clusters were detected at high frequency, mainly on blood smears of heterozygous DBA/2 mutants. Similar results were obtained in at least 3 to 6 animals for experimental points. Original magnifications are indicated at the bottom of each panel.
Histologic analysis of the myelofibrotic trait in 12-month-old heterozygous F1 Gata1low females carrying the mutation in a predominantly CD1, C57BL/6, or DBA/2 background. Blood smears are shown on the right panels, while hematoxilin-eosin and Gomori staining of liver, spleen, and marrow are shown on the top and bottom panels, as indicated. The genetic background of the mice analyzed is specified in Figure 1, while the quantification of Mk frequency and of the fibrosis in each animal group is presented in Table 2. The arrow in the liver section in panel A indicates an erythroid nest within the parenchyma, already detectable at 12 months in CD1 mutants, while the arrowheads on the blood smears in panel C indicate clusters of normal platelets, that is, deriving from the stem cell population that had inactivated the X chromosome carrying the Gata1low allele. These clusters were detected at high frequency, mainly on blood smears of heterozygous DBA/2 mutants. Similar results were obtained in at least 3 to 6 animals for experimental points. Original magnifications are indicated at the bottom of each panel.
The total number of cells in the spleen from Gata1low mice is positively correlated with the hematocrit, and the frequency of MK in the spleen is correlated with the fibrosis in the spleen. Each dot represents an individual mutant mouse. For the purpose of this analysis, results obtained in heterozygous or hemizygous mice of different backgrounds were pooled together. The best-fitting linear regression and the corresponding significance value are indicated.
The total number of cells in the spleen from Gata1low mice is positively correlated with the hematocrit, and the frequency of MK in the spleen is correlated with the fibrosis in the spleen. Each dot represents an individual mutant mouse. For the purpose of this analysis, results obtained in heterozygous or hemizygous mice of different backgrounds were pooled together. The best-fitting linear regression and the corresponding significance value are indicated.
As shown in Figure 2, heterozygous females of different backgrounds differed greatly in the extent of osteosclerosis in femur. As expected,14 C57BL/6 mutants expressed massive bone formation, while osteosclerosis was limited, or barely detectable, in CD1 or DBA/2 mutants, respectively (Figure 2). There was no association between frequency of Mks in marrow and extent of bone formation (Figure 2 and Table 1).
Bone formation is the result of the balance between osteoclast and osteoblast activity. To address whether differences in bone formation in different strains were due to differences in osteoclast/osteoblast activity, we determined osteoclast frequency (on the basis of tartrate-resistant acid phosphatase staining) and osteoblast proliferation activity (indirectly, as levels of osteocalcin expression) in the femur of normal and mutant mice of different strains. Although the complex structure of the bone precludes a precise quantification of the osteoclasts, no differences were appreciated in the frequency of these cells in bones from the mutant and the corresponding wild-type animals. However, bones from CD1 mice (both Gata1low and wild-type) contained twice as many osteoclasts than those from mice of the other strains (Figure 4). On the other hand, similar low levels of osteocalcin were expressed by wild-type mice of all backgrounds, but mutant mice expressed at least 1-log more osteocalcin than the corresponding wild-type animals (Table 3). Differences in osteocalcin levels expressed by mutants of different strains were observed but were not statistically significant by Anova analysis.
Therefore, all the mutants analyzed, independently from background, developed fibrosis at 12 to 13 months of age. The number of Mks was not associated with either fibrosis or osteosclerosis in marrow, but a direct linear relationship between number of mutant Mks and number of fibers was present in spleen.
Expression of stem (CD117pos/Sca-1pos) and progenitor (CD117pos/CD34pos) cell antigens, as well as of erythroblast (TER119pos/CD71pos) markers, on marrow cells from normal and mutant mice of different backgrounds
It is known that in mice the expression of hemopoietic-specific antigens is profoundly affected by genotype36 and age.37 Therefore, in preliminary experiments, we compared the expression profile of antigens that define stem (CD117pos/Sca-1pos) and progenitor (CD117pos/CD34pos) cells, as well as of those that mark erythroblasts at different stages of maturation (TER119pos/CD71pos), in marrow cells from normal littermates of different backgrounds (Figure 5 and Table 4). The cells also were functionally characterized by clonogenic (progenitor assay) and morphologic (May Grunwald-Giemsa staining) criteria.
Marrow cells from different strains expressed slightly different CD117 staining patterns, but the differences did not involve the CD117pos/Sca-1pos population whose frequency was roughly equivalent in all 3 strains analyzed (Figure 5 and Table 4). As control, CD117pos/Sca-1pos cells were sorted and their cloning efficiency determined in semisolid assay. In all the cases, 10% of CD117pos/Sca-1pos cells, but none of the CD117pos/Sca-1neg ones, formed colonies at day 15. Sca-1 recognizes Ly-6A/E,38 an antigen that in Ly-6b mice, such as C57BL/636 and DBA/2,39 marks 99% of the marrow repopulating activity of the CD117pos cells. Therefore, in the rest of the study, the frequency of CD117pos/Sca-1pos cells was taken as indication of the number of stem cells present in a tissue.
The expression pattern of CD34 on marrow cells from mice of different backgrounds was heterogeneous. In C57BL/6 mice, most (96%) of the CD117pos cells were CD34high, and these cells contained all the colony-forming activity of the tissue (approximately 90 colonies/100 cells plated). In contrast, the CD117pos fraction of marrow from CD1 and DBA/2 mice expressed a continuum pattern of CD34 staining (from dim to high) (Figure 5). Both CD117posCD34dim and CD34high cells, independently sorted and plated in standard methyl-cellulose culture, gave rise to more than 75 colonies (mostly day 15 or 8, respectively) per 100 plated cells. In mice, the CD34 antigen is expressed by all progenitor cells but only by fetal-neonatal stem cells.37 Adult stem cells, however, retain the ability to express CD34 when induced to cycle.40 The results on the cloning efficiency and these considerations suggest that the CD34dim fraction of CD117pos cells represents an immature stem/progenitor cell population evident only in some strains. Therefore, for the purpose of this study, the frequency of all the CD117pos cells (CD34high in C57BL/6 mice and CD34dim-high in CD1 and DBA/2 mice, all indicated as CD34pos for clarity) was considered as a measure of the progenitor cells present in a tissue.
Bone marrow cells from normal mice of the 3 backgrounds analyzed contained significantly different numbers of erythroid cells. In fact, TER119posCD71pos cells represented 18% or 30% of the total marrow cells from C57BL/6 and CD1 mice, respectively. Furthermore, the changes in TER119 and CD71 expression pattern supposedly associated with the process of erythroblast maturation were found to be extremely sensitive to genetic background. In DBA/2 mice, CD71 and TER119 expression divided the erythroid cells into the 4 classes (TER119medCD71high and TER119high CD71high, CD71med and TER119highCD71low), corresponding to pro-erythroblasts and basophilic, chromatophilic, and orthocromatophilic erythroblasts, described by Socolovsky et al18 (Figure 5 and not-shown morphologic analysis of sorted cells). Such distinction was conserved in the CD1 background (Figure 5) but not in C57BL/6 mice, in which the levels of CD71 expression remained relatively constant with maturation, while that of TER119 became markedly increased (Figure 5), as described for this same mouse strain by Hall et al.41 Because of these strain differences, no attempt was made in the rest of the study to quantify the proportion of different erythroid precursors, and the frequency of the entire TER119posCD71pos cell population was taken as an indication of active erythropoiesis in a tissue.
Tartrate-resistant acid phosphatase staining of representative bone marrow sections from 12-month-old heterozygous F1 Gata1low females carrying the mutation in a predominantly C57BL/6 and in hemizygous F2 males of CD1 and DBA/2 backgrounds, as indicated. Staining of the corresponding sections from wild-type littermates also are indicated. Results are representative of those obtained in 3 animals per experimental group. The presence of osteoblasts is indicated by arrows. Original magnification, × 20.
Tartrate-resistant acid phosphatase staining of representative bone marrow sections from 12-month-old heterozygous F1 Gata1low females carrying the mutation in a predominantly C57BL/6 and in hemizygous F2 males of CD1 and DBA/2 backgrounds, as indicated. Staining of the corresponding sections from wild-type littermates also are indicated. Results are representative of those obtained in 3 animals per experimental group. The presence of osteoblasts is indicated by arrows. Original magnification, × 20.
In the marrow, the frequency of CD117posSca-1pos and CD117posCD34pos cells was significantly lower than normal only in hemizygous CD1 males, while that of TER119posCD71pos cells was decreased only in hemizygous DBA/2 males (Figure 6 and Table 4). However, since marrow cellularity of all the mutants (independently from their strain; Table 1) was greatly lower than normal, the number of hemopoietic cells of all types (stem/progenitor cells and erythroblasts) was greatly reduced in marrow from all 16- to 20-month-old mutants (Figure 7).
In contrast, in the spleen, the frequency of CD117posSca-1pos cells was significantly higher than normal in hemizygous DBA/2 males, while that of CD117posCD34pos cells was significantly increased in all the DBA/2 and CD1 mutants (Table 4). Overall, because of the great increase in cellularity detected in this organ in most of the mutants (Table 1), the number of stem/progenitor cells increased in spleens from all mutants (Figure 7). On the other hand, the numbers of maturing erythroblasts (both as frequency, Figure 6 and Table 4, and as total number, Figure 7) were higher than normal only in spleens from hemizygous CD1 mutants.
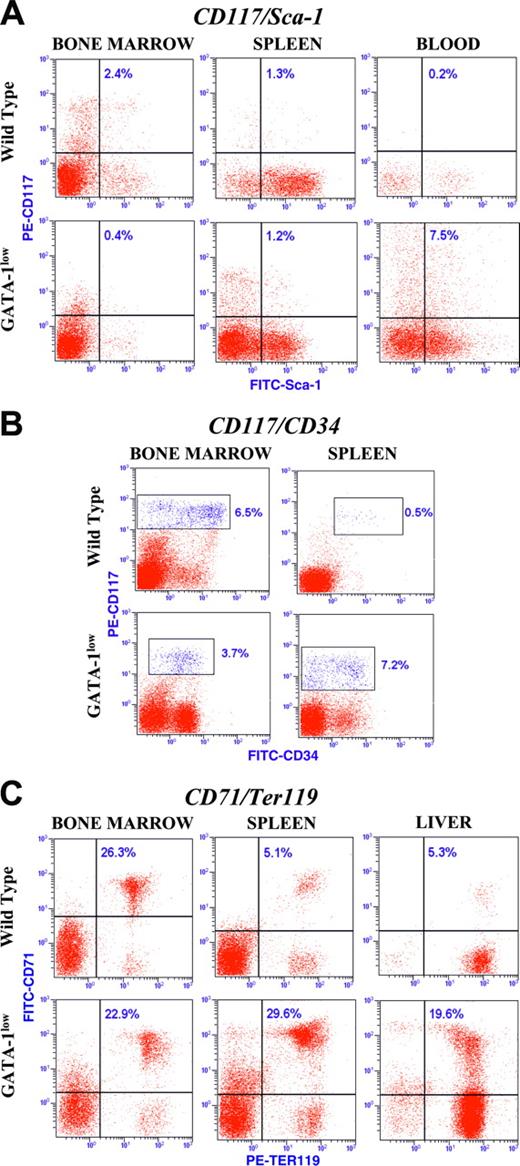
Dot plot analysis for the expression of the CD117/Sca-1, CD117/CD34, and CD71/TER119 antigens in marrow cells from 16- to 20-month-old F1-F2 normal littermates with predominant C57BL/6, DBA/2, or CD1 backgrounds, as indicated. The gates defining stem and progenitor cells as well as those corresponding to maturing erythroblasts are CD117pos/Sca-1pos, CD117pos/CD34dim to high, and CD71pos/Ter119pos, respectively, as indicated. The number in each quadrant indicates the frequency of marrow cells in that gate. Similar results were obtained in at least 3 mice for each strain, and mean (± SD) of results obtained in different mice is presented in Table 4.
Dot plot analysis for the expression of the CD117/Sca-1, CD117/CD34, and CD71/TER119 antigens in marrow cells from 16- to 20-month-old F1-F2 normal littermates with predominant C57BL/6, DBA/2, or CD1 backgrounds, as indicated. The gates defining stem and progenitor cells as well as those corresponding to maturing erythroblasts are CD117pos/Sca-1pos, CD117pos/CD34dim to high, and CD71pos/Ter119pos, respectively, as indicated. The number in each quadrant indicates the frequency of marrow cells in that gate. Similar results were obtained in at least 3 mice for each strain, and mean (± SD) of results obtained in different mice is presented in Table 4.
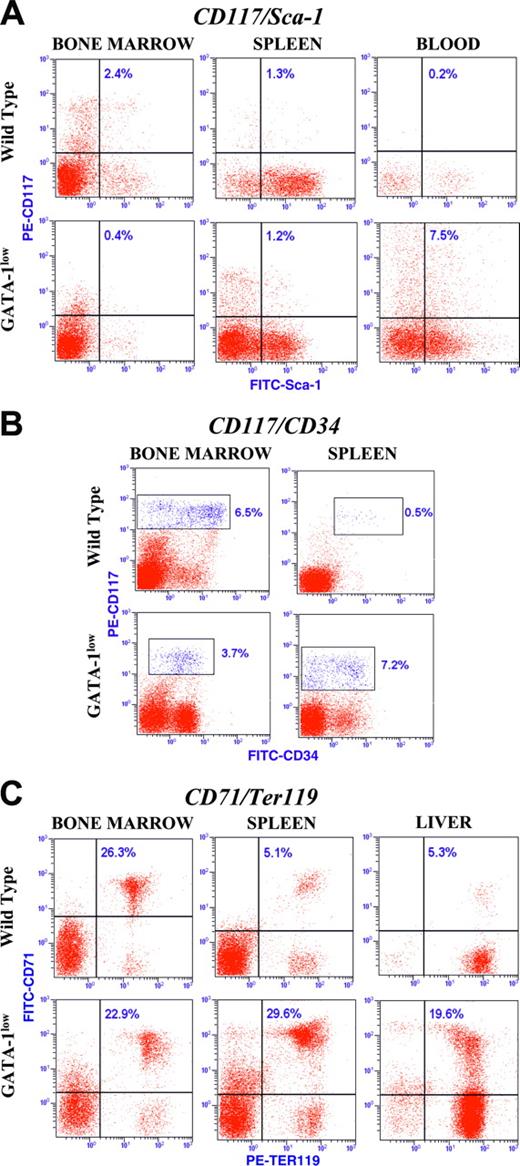
Of note, stem/progenitor cells were detectable in blood mainly from CD1 mutants, where the frequency of CD117posCD34pos and Sca-1pos cells reached 2.5% and 7.5% of the total mononucleated cells, respectively (Figure 6A and data not shown). CD1 mutants also were the only ones in which significant numbers of progenitor cells and maturing erythroblasts were detected in the liver at this age (Figure 6C and Table 4).
Therefore, at 16 to 20 months of age, the marrow was a poor hemopoietic site in all of the mutants analyzed, while the spleen itself was still an active erythropoietic organ mainly in CD1 mutants. These mutants also were the only ones to express significant extramedullary hemopoiesis in liver.
Dot plot analysis for the expression of the CD117/Sca-1, CD117/CD34, and CD71/Ter119 antigens in cells from marrow, spleen, blood, and liver from 16- to 20-month-old Gata1low mutants and normal littermates of CD1 background, as indicated. The number in each quadrant indicates the frequency of the cells in that gate. Similar results were obtained in at least 3 mice for each strain, and mean (± SD) of results obtained in different mice is presented in Table 4.
Dot plot analysis for the expression of the CD117/Sca-1, CD117/CD34, and CD71/Ter119 antigens in cells from marrow, spleen, blood, and liver from 16- to 20-month-old Gata1low mutants and normal littermates of CD1 background, as indicated. The number in each quadrant indicates the frequency of the cells in that gate. Similar results were obtained in at least 3 mice for each strain, and mean (± SD) of results obtained in different mice is presented in Table 4.
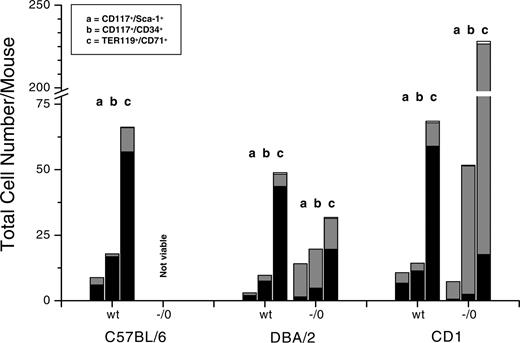
Total number of stem and progenitor cells and of maturing erythroblasts in normal as well as in hemizygous Gata1low mutants of different background, as indicated. C57BL/6 mutants were not analyzed because they were never born. The total number of each cell population was calculated by multiplying its frequency (Table 4) per the total number of cells per organ (Table 1), taking into account that the cells in a femur correspond to 8% of the total marrow population of a mouse.74 The relative contribution of the marrow, spleen, and liver to each population is indicated in black, gray, and white, respectively.
Total number of stem and progenitor cells and of maturing erythroblasts in normal as well as in hemizygous Gata1low mutants of different background, as indicated. C57BL/6 mutants were not analyzed because they were never born. The total number of each cell population was calculated by multiplying its frequency (Table 4) per the total number of cells per organ (Table 1), taking into account that the cells in a femur correspond to 8% of the total marrow population of a mouse.74 The relative contribution of the marrow, spleen, and liver to each population is indicated in black, gray, and white, respectively.
Discussion
Thrombocytopenia, fibrosis, and reduced hemopoiesis in the marrow were the only traits expressed by Gata1low mutants independently from their genetic background (Figure 2 and Table 2). Strain variegations were instead observed in perinatal mortality, occurrence of anemia, and complexity (tear-drop poikilocytes in blood, osteosclerosis, extramedullary hemopoiesis) of the myelofibrosis developed by these animals (Table 5).
The strain-independent expression of thrombocytopenia and fibrosis is in agreement with the notion that Mks are direct targets of the Gata1low mutation.12,13 In fact, low GATA-1 in Mks hampers expression of Mk-specific genes, assembling of α-granules, and formation of platelet territories.12,13,21 On the other hand, in spleen, fibers and Mks numbers were directly correlated (Figure 3), in agreement with the assumption that fibrosis results from pathologic P-selectin localization on the DMS of the Mks that triggers neutrophil emperipolesis, para-apoptosis of Mk, and TGF-β release in the micro-environment.21 High TGF-β levels in the microenvironment would, then, cause fibrosis by stimulating fibroblasts to produce reticulinic and collagen fibers.42
Mutant Mks might also be responsible for the osteosclerosis observed in Gata1low mice by stimulating osteoblast proliferation in vivo, as they do in vitro under cell-cell contact conditions.14 However, strain differences in bone formation were not directly related to the numbers of Mks present within the marrow. The observation that normal mice receiving transplants of stem cells infected with a thrombopoietin (TPO)-expressing retrovirus develop both myelofibrosis and osteosclerosis, while mice deficient for the osteoclast inhibitor osteo-protegerin received transplants of the same TPO-overproducing stem cells develop only myelofibrosis,43 suggests that unbalanced osteoblast/osteoclast activity might contribute to strain differences in bone formation. In this regard, the high levels of osteocalcin in the bones indicate that all the mutant mice experience high osteoblast activity (Kacena et al14 and Vannucchi et al20 ; Table 3). Statistically significant (P < .05 by t test) differences in osteocalcin expression were observed in mice of different strains, but it is still unclear whether these differences represent strain or individual variability. On the other hand, the number of osteoclasts in the bone from CD1 mutants, although normal for their strain, was higher than that of C57BL/6 and DBA/2 mutants (Figure 4). Therefore, strain-specific balances in osteoblast/osteoclast activity might be responsible, at least in part, for the differences in osteosclerosis of these mutants (Table 5).
Osteoblasts are key elements of the hemopoietic stem cell niche.44 Genetically engineered mutations that increase the number of osteoblasts increase also the number of stem cells in the marrow,45,46 while genetic ablation of the osteoblast pool results in extramedullary hemopoiesis in spleen and liver.47 In contrast, in Gata1low mice, the stem cell frequency in the marrow appeared to be inversely related to osteoblast activity (lower in CD1 than in DBA/2 mice [Table 4], with a bone mass lower in DBA/2 than in CD1 mutants [Figure 2]). Since osteoblast proliferation is probably triggered by cell-cell interaction with mutated Mks, we suggest that establishment of this pathologic interaction might dislodge stem cells from their niches. This dislodgment, however, is not sufficient to cause extramedullary hematopoiesis, since hematopoietic cells were almost exclusively observed in livers from CD1 mutants (both at 12 to 13 and 16 to 20 months), that is, in the strain in which stem cells remain functional with age. It is possible that establishment of extramedullary hemopoiesis is the result of both extrinsic (dislodgment from the niche) and intrinsic (retention of stem cell properties) mechanisms (Table 5).
Also, increased erythroblast apoptosis is a direct consequence of the Gata1low mutation,10,11 but adult mutants express normal Hct by recruiting the spleen as hemopoietic site at 1 month,15,48 and a direct correlation between total number of spleen cells and Hct was still present in mutants of 12 to 20 months of age (Figures 3, 7; Table 1). Activation of spleen erythropoiesis in response to stress is controlled by the locus Fv2, which encodes the Stk receptor and confers susceptibility to the Friend Leukemia Virus (FLV).49 Strains susceptible to FLV (DBA/2 and CD1) express the Fv2s allele, which encodes a truncated Stk, sf-Stk, transcribed from an alternative promoter containing a functional GATA binding site. This site is instead deleted in Fv2r, the allele carried by strains resistant to FLV (C57BL/6).50 When expressed, sf-Stk forms a complex with the EPO receptor required for the first step in FLV leukemogenesis51 (ie, constitutive activation of the receptor by the viral gp5552 ) and to recover from erythroid stresses (ie, phenylhydrazine-induced hemolytic anemia50 ) that involve spleen erythropoiesis. It is possible that Fv2 might also favor compensation of the defective erythropoiesis induced by the Gata1low mutation (Table 5). In fact, the Gata1low mutation does not necessarily affect sf-Stk expression, since CD1 mutants (carrying Fv2s) retain the capacity to recover from phenyl-hydrazine-induced anemia.15 At birth, the mutation was progressively less lethal as the background increased from C57BL/6 (Fv2r) to CD1 and DBA/2 (both Fv2s), suggesting that Fv2s protects from anemia at the end of fetal development. On the other hand, the observation that 16- to 20-month-old DBA/2 and CD1 mutants differed greatly in the number of TER119pos/CD71pos cells present in spleen and in occurrence of anemia (Tables 1, 4; Figure 7) indicates that Fv2s alone is not sufficient to sustain spleen erythropoiesis in old mice.
Discrepancies between genotype and function have been described for many inherited hematologic diseases53 and complicate the analysis of genetically modified mice.54 In an increasing number of cases, discrepancies are ascribable to polymorphisms in loci different from those determining the primary defect defined “gene modifiers.” The complex trait variegation expressed by Gata1low mutants of different backgrounds suggests that their phenotype might be affected by one (or more) gene modifier(s). Strain variegation was already manifested at the first backcrossing, when allelic heterozygosity was only 50% to 75%. Therefore, eventual Gata1 gene modifier(s), as all the QTL identified so far,55 must have low meiotic recombination rates. The infrequency of the chromosome regions with low recombination rates suggests that QTL and Gata1 gene modifiers might be the same genes. The QTL identified up to now include a cluster on chromosome 7, and 3 clusters on chromosome 11.55 The chromosome 11 clusters have been identified on the basis of 2 experimental approaches: genetic segregation and differential expression profiling for genes highly expressed in DBA/2 stem cells.55 One of these clusters contains STAT-5a, b, and is only 30 cM distant from the region synthenic with the cytokine cluster on human chromosome 5q. Another one is synthenic with human 17q21 that contains BCRA1.55 The cluster on chromosome 7 and those on chromosome 11 are linked by the observation that chromosome 7 contains a Stat5 gene modifier that restores the ability of STAT-5null stem cells to engraft in competitive repopulation assays.56 It is possible that either STAT-5a, b, the chromosome 7 cluster, or any other QTL gene might affect the phenotype, including development of extramedullary hemopoiesis, of Gata1low mutants (Table 5).
Also, human IM is a heterogeneous disease that manifests itself with extremely variable morbidity and mortality.24-26 Familiar predisposition toward development of this disease is only now starting to be appreciated.57,58 It is possible that, as perinatal mortality would have prevented the recognition of genetically transmitted myelofibrosis in C57BL/6 mutants, familiar predisposition in IM is masked by in utero mortality.
The primary molecular defect leading to IM, the IM locus, has not been identified as yet. Although it is unlikely that this locus is represented by Gata1, Mks from these patients express the same morphologic (ie, pathologic neutrophil emperipolesis)23 and biochemical (reduced GATA-1 content)59 abnormalities of Gata1low Mks. Many cytogenetic defects (mostly del(13q), del(20q), or partial trisomy 1q) have been associated with IM, but none of them is unique to the disease.60,61 Other genes that also have been implicated in IM are classic tumor suppressor genes, such as the retinoblastoma, Tp53, p16, and the RAS family of proto-oncogenes.62-64 Since IM stem cells present either elevated expression65 or point mutations66,67 of c-kit, this gene also has been implicated with IM development. Furthermore, elements of the b-fibroblast growth factor pathway have been proposed as candidates for the IM locus.68 Interestingly, elements of this pathway represent QTL in mice.69 More recently, a V617F mutation in the Jak2 gene was discovered in 30% to 50% of IM patients.59,70-73 However, it is still unclear how mutations in any of these genes can possibly induce the specific Mk abnormalities observed in IM. Our results suggest that although none of these genes might represent the IM locus, any of them might represent a gene modifier(s) of Gata1 and contribute to the overall phenotype of the Gata1low mice. Conversely, Gata1 gene modifiers represent likely candidates for the IM locus.
In conclusion, the variegation of the phenotype expressed by Gata1low mutants in different backgrounds suggests that this complex trait is the result of the interaction between the primary genetic defect, the Gata1low mutation, with one (more) gene modifier(s). Although most of the variegation observed remains unexplained, on the basis of the known genetic differences among the strains investigated, possible candidates for Gata1 gene modifiers were identified. It is possible that polymorphisms in these genes might underlay the variability of the clinical picture of human IM as well.
Prepublished online as Blood First Edition Paper, August 18, 2005; DOI 10.1182/blood-2005-03-1060.
Supported by Ministero per la Ricerca Scientifica e Tecnologica, grant RBNE0189JJ_003, RBNE015P72_003, and DM193, 2003; the Ministero Università Ricerca Scientifica Tecnologica (MURST) (Progetti di Ricerca di Interesse Nazionale [PRIN] 2000, grant MM06103241; 2003, grant 06488803); Programma di Ricerca Cellule Staminali, from the Ministry of Health, Associazione Italiana Ricerca Cancro, and institutional funds from Istituto Superiore Sanità, Rome, Italy. A.P. was the recipient of an Associazione Italiana Leucemie (AIL) fellowship, Florence, Italy.
F.M. performed cytofluorimetric analysis. B.G. performed cell purification and culture. B.P. organized the data and performed statistical analysis. E.A. conducted all of the mating and kept a registry of the mouse colony. V.G. performed mice genotyping. A.P. performed histologic analysis and quantitative RT-PCR determinations. C.B. prepared hemopoietic tissues for analysis. A.M.V., F.P., G.M., and A.R.M. designed research, analyzed the data, and wrote the paper.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.
The authors are grateful to Len Zon and Gary Van Zant for helpful discussions. This study would not have been possible without the enthusiastic cooperation of all the staff of the animal facility of the Istituto Superiore di Sanità.